Utilization of Prescription Opioids in Canada's Public Drug Plans, 2006/07 to 2012/13
April 2014
PDF version
ISBN: 978-1-100-23532-5
Cat No.: H82-17/2014E-PDF
About the PMPRB
The Patented Medicine Prices Review Board (PMPRB) is an independent quasi-judicial body established by Parliament in 1987.
The PMPRB has a dual role: to ensure that prices at which patentees sell their patented medicines in Canada are not excessive; and to report on pharmaceutical trends of all medicines and on R&D spending by patentees.
The PMPRB reports annually to Parliament, through the Minister of Health, on its activities, on pharmaceutical trends relating to all medicines, and on R&D spending by patentees.
The NPDUIS Initiative
The National Prescription Drug Utilization Information System (NPDUIS) provides critical analyses of drug price, utilization, and cost trends in Canada to support drug plan policy decision-making for participating federal, provincial, and territorial governments.
The NPDUIS initiative is a partnership between the PMPRB and the Canadian Institute for Health Information (CIHI). It was established in 2001 by the federal, provincial and territorial Ministers of Health.
Acknowledgements
This report was prepared by the Patented Medicine Prices Review Board (PMPRB) under the provisions of the National Prescription Drug Utilization Information System (NPDUIS).
The PMPRB would like to acknowledge the contributions of:
- The members of the NPDUIS Steering Committee, for their expert oversight and guidance in the preparation of this report.
- The PMPRB NPDUIS staff for their contribution to the analytical content of the report: Gary Warwick, Senior Economic Analyst; Elena Lungu, A/Manager NPDUIS; and the PMPRB scientific and editing groups.
Disclaimer
NPDUIS is a research initiative that operates independently of the regulatory activities of the Board of the PMPRB. The statements and opinions expressed in this NPDUIS report do not represent the official position of the PMPRB.
Parts of this material are based on data and information provided by the Canadian Institute for Health Information. However, the analyses, conclusions and/or statements expressed herein are not necessarily those of the Canadian Institute for Health Information.
Executive Summary
This study focuses on the use of opioids by beneficiaries of selected public drug plans in Canada†. It reports on trends in the number of claims, drug cost and morphine equivalence from fiscal year 2006/07 to 2012/13. Morphine equivalence, which measures the relative potency of a particular opioid, was used to standardize the impact of utilization across opioids. The report compares different claimant groups based on the quantity of opioids utilized. Special focus is given to codeine, oxycodone and hydromorphone given the importance of their use in the treatment of pain.
Overview of opioid use – Trends and demographics
An overview of the utilization and cost in Canada's public drug plans gives the following results:
- The frequency of opioid use among plan beneficiaries is relatively high: among the plans included in the analysis, between 12.3% and 20.0% of active beneficiaries had at least one claim for an opioid in a given year.
- Conversely, opioids represent between 2.4% and 5.8% of all claims and between 1.5% and 3.6% of associated drug costs. This is because the average number of claims for opioids per beneficiary per year across the plans is typically less than 5 claims per year compared with between 18 and 42 claims for all other drugs.
- While the number of claims for opioids increased significantly in many of the drug plans between 2006/07 and 2012/13, much of the increase was in line with the percentage growth for all drugs. The exceptions were Saskatchewan (23.6% increase for all drugs vs. 42.5% for opioids); Nova Scotia (38.0% increase for all drugs vs. 81.1% for opioids); and Prince Edward Island (69.3% increase for all drugs vs. 86.2% for opioids)
- Three major factors contributed to the overall growth in claims:
- Population effect – the change in the overall active beneficiary population. This factor had an impact in Nova Scotia and Prince Edward Island.
- Plan concentration – the change in the share of opioid claimants relative to the total active beneficiary population. This factor had an impact in Saskatchewan and Prince Edward Island.
- Intensity of use – the change in the average number of claims per claimant. This factor had an impact in all of the drug plans analyzed.
- Differences in plan design make it difficult to draw conclusions based on a relative assessment between plans. Nonetheless, utilization rates for seniors, for which plan coverage is more consistent across the public drug plans, were fairly similar. A lot of variation was seen in non-senior utilization rates. Non-senior utilization rates tended to be lower in plans with the highest participation rates under 65 (i.e., Manitoba and Saskatchewan).
Weaker vs. stronger opioids
Eight drugs were analyzed: the weaker opioids – meperidine and codeine (alone and in combination with another analgesic); and the stronger opioids – morphine, oxycodone (alone and in combination with another analgesic), hydromorphone and fentanyl. Market shares were considered along with trends in use. The analysis revealed the following:
- Across the drug plans, weak opioids, such as codeine, accounted for 40.2% of all opioid claims, 12.8% of morphine equivalents and 9.1% of opioid drug costs in 2012/13. At the individual plan level, their use ranged from 31.7% of opioid claims in Ontario to 64.1% of opioid claims in the Non-Insured Health Benefits Program (NIHB).
- Strong opioid use was dominated by oxycodone and hydromorphone. Combined they accounted for 44.9% of opioid claims, 60.7% of morphine equivalents and 66.4% of opioid drug costs in 2012/13. At the individual plan level, their use ranged from 22.3% of opioid claims in the NIHB to 52.6% of opioid claims in Ontario.
- Much of the growth in opioid use in terms of claims and morphine equivalents resulted from an increased use of oxycodone and hydromorphone. Combined, these two drugs grew by 60.6% in terms of opioid claims per 1000 beneficiaries between 2006/07 and 2012/13.
Low- vs. high-use claimants of opioids
To better understand how individuals use opioids and how this use affects overall trends, opioid claimants were classified based on the number of claims made in a year. Two groups were identified as having the greatest impact: claimants making only 1 or 2 claims per year and those making more than 10 claims per year.
- The majority of opioid claimants were probably acute patients, with approximately 60% of claimants making only 1 or 2 claims in a year. Typically, weaker opioids were dispensed to this group, ranging from 47.8% of the total opioid claims in Nova Scotia to 79.9% in Manitoba in 2012/13.
- High-use opioid claimants, making more than 10 claims per year, represented between 10% and 19% of all opioid claimants. However, they were responsible for between 47.3% and 69.1% of all opioid claims and as much as 84.5% of opioid drug costs and 82.8% of morphine equivalents dispensed. On average, this group made three times as many claims for an opioid (24.7 compared to 7.7) and was dispensed more than three times the morphine equivalents (36,916/claimant compared to 9,056/claimant) in a year than the next group, which made between 6 and 10 claims per year.
- For high-use claimants, opioids represented only a part of their overall drug use. On average, members of this group made between 70 and 144 claims for drugs other than opioids in 2012/13. The average beneficiary made between 18 and 42 claims during the same period. The most frequently used drugs were used to treat problems related to pain or pain treatment. Other drugs were prescribed to treat general health problems, primarily high blood pressure and heart disease.
- Over time, the utilization of stronger opioids increased for all claimants regardless of the number of claims they made per year. For claimants making 1 or 2 claims per year, the share of claims for strong opioids increased from 34.8% in 2006/07 to 42.0% in 2012/13. For high-use claimants, the share of strong opioids grew from 66.7% to 73.7% over the same period.
† These include British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, New Brunswick, Nova Scotia, Prince Edward Island and Health Canada's Non-Insured Health Benefits Program.
Introduction
The prescribing and utilization of opioids is at the center of an ongoing public policy debate in Canada. On one hand, opioids are an important and effective treatment for pain. On the other hand, if used inappropriately, they are a source of addiction and a cause of death. As a result, Canada, along with most of the western world, has designated opioids as controlled substances and put laws in place controlling their prescription and dispensing to ensure that access is limited to only those patients in need.
In recent years Canada's experience with OxyContin has highlighted the dangers of opioid misuse. While the vast majority of patients have safely and successfully treated their pain with this drug, it has become associated with stories of abuse, addiction and even death. Its potency and purity have made it the street drug of choice for many addicts once they discovered ways to by-pass the drug's safety features. Even though OxyContin is no longer on the market1, the debate surrounding the prescription of opioids continues. Some groups have expressed concern about the perceived increase in the prescription rate of opioids, suggesting that physicians prescribe them too freely. Others suggest that the medical community is simply more aware of the need to treat pain and its underlying causes.
Regardless of the reason driving the growth in utilization, the fact is that more and more Canadians have been prescribed opioids to deal with their pain. In response, regulators have taken steps to strengthen and expanded their efforts to ensure appropriate prescribing. Now most Canadian jurisdictions combine tracking programs, including triplicate prescription programs, with education and support for patients and healthcare workers as ways of reducing the misuse and abuse of these drugs. For details of current provincial policies, see Appendix A.
This study focuses on opioid use among beneficiaries of selected public drug plans in Canada. It reports on trends in utilization in terms of claims, drug cost and morphine equivalence. The analysis groups patients based on intensity of use to gain further insight. Because of their individual importance in the treatment of pain, special focus is placed on codeine, oxycodone and hydromorphone.
Methodology
Data sources
Price and utilization data for this study came from the NPDUIS claims-level database housed within the Canadian Institute for Health Information (CIHI) and a DIN-level database maintained by the PMPRB. Both databases contain administrative data from various provincial drug plans, based on prescriptions dispensed and accepted as claims by drug programs, either for reimbursement or toward a plan deductible. Data was extracted for patients making claims for opioids for the fiscal years 2006/07 to 2012/13, based on the Anatomical Therapeutic Chemical (ATC) level 5 codes, established by the World Health Organization. Because beneficiary information in the NPDUIS database is limited for British Columbia, Ontario and Health Canada's Non-Insured Health Benefits (NIHB) Program over this period, some of the more in-depth analysis of drug utilization at this level was not possible. Unless otherwise specified, core analysis was performed on the public plans of Alberta, Saskatchewan, Manitoba, New Brunswick, Nova Scotia and Prince Edward Island.
Drugs covered in the study
The focus of the study is on eight drugs: the weaker opioids – meperidine and codeine (alone and in combination with another analgesic); and the stronger opioids – morphine, oxycodone (alone and in combination with another analgesic), hydromorphone and fentanyl. While there are other opioid treatments available on the market, they were not included either because they were not widely dispensed or were not included in provincial formularies. With the exception of fentanyl, all drugs studied are oral solids. Because the dosage strength of other forms, such as liquids or injectables, are not fixed, it is difficult to calculate how much a patient may be taking on a daily basis. The most common form of fentanyl is a transdermal patch. This was included because each patch is a fixed dose.
Morphine equivalence
Morphine equivalence (ME) measures the relative potency of a particular opioid and is used as a method of standardization. Converting each of the drugs into their respective equivalents greatly facilitates the tracking of prescribing trends. The Canadian Guideline for Safe and Effective Use of Opioids for Chronic Non-Cancer Pain (2010)2 was used as the primary source of conversion factors. These factors were corroborated using guidelines established in the UK and in a number of US states, as well as the University of Alberta. Because of the way fentanyl is metabolized by the body via the patch, morphine equivalence is generally reported with an upper and lower limit. For the purpose of the study, the mid-point of each band was used in the calculations.
Definitions
Drug plan active beneficiaries: The number of unique patients who have had at least part of at least one claim accepted by a plan/program for benefit.
Opioid claimant: A drug plan beneficiary with at least one claim for an opioid between 2006/07 and 2012/13.
High-use opioid claimant: An opioid claimant making more than 10 claims for an opioid per year.
Drug cost: The cost of the drug including the price of the product and wholesale upcharges, and excluding pharmacy markups.
NPDUIS National: Member drug plans supplying data to the NPDUIS initiative. This includes British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, New Brunswick, Nova Scotia and Prince Edward Island and Health Canada's Non-Insured Health Benefits (NIHB) Program.
Limitations
There are a number of limitations that should be considered when interpreting the results of this analysis.
Limitations pertaining to scope of the analysis
The public drug plan data analyzed represents only one component of the pharmaceutical market. Therefore, these results should not be extrapolated to other population groups, as the findings may be different for opioid use reimbursed under other publicly or privately funded plans or paid for out-of-pocket by patients.
Limitations pertaining to the administrative database
It is not known whether drugs were utilized as dispensed. Administrative drug plan data does not capture information on prescriptions dispensed but not claimed under the drug plan, or claimed but not approved. Nor does it contain any information about the indication for which a particular drug is prescribed.
Cross-jurisdictional comparison of opioid use
The study reports varying rates of opioid use across the public drug plans analyzed. Some of the variations may be explained by differences in the demographic profile of the population in each province, the public drug plan eligibility criteria and/or the cost-sharing structures. At a population level, there are differences in the demographic profiles across provinces (e.g., Alberta has a younger population), as well as varying prevalence rates for diseases. At a public drug plan level, each plan is uniquely designed to reimburse specific segments of the population. Furthermore, each public drug plan has a specific deductible-copayment structure, which can differ across the subgroups of the population reimbursed. Other factors that may explain variations across the plans include whether a particular drug is listed on the provincial drug formulary, whether reimbursement is granted on an exceptional basis (requiring special permission from the plan) and whether thxere are limits placed on prescription size (i.e., 30, 60 or 100 day).
More specifically, with respect to variations in the beneficiary population, the universal drug plans in British Columbia, Saskatchewan and Manitoba are income-based programs that provide broad-based coverage for all residents. Both Ontario and New Brunswick offer senior-based programs along with coverage for low-income families. In addition, Ontario offers an income-based program to cover catastrophic drug expenditures. Alberta offers a publicly funded supplementary drug benefit program that is premium-based for non-seniors and premium-free for seniors. Nova Scotia offers subscription-based programs with different fee structures for seniors and families, as well as income assistance recipients, persons with disabilities and children in the care of child welfare. In addition to a senior's plan, Prince Edward Island covers their non-senior population through a number of disease-specific plans, such as for multiple sclerosis. The NIHB drug plan offers broad drug coverage to the First Nations and Inuit populations. Appendix B provides further details on public drug plan design.
Note that the results for the senior population are more comparable across the public drug plans given the broad coverage of seniors, which results in a high degree of homogeneity in seniors across plans. On the other hand, the results for the non-senior population are less comparable, given the targeted demographics, which results in less homogeneity in the non-senior population across plans.
1 Overview of Opioid Use and Cost in Public Drug Plans
An overview of the use of opioids in public drug plans suggests that while a large proportion of drug plan beneficiaries use opioids in a given year, opioid use accounts for a small percentage of overall drug plan claims and cost. The results also suggest that there are variations across the provinces. These are further explored in the next section, which discusses demographic and plan design differences. Opioids are broadly utilized by public drug plan beneficiaries. Depending on the plan, as many as one-fifth of active beneficiaries (Ontario) made a claim for an opioid during 2012/13 (Figure 1.1). However, there are wide variations, with lower rates of use in Saskatchewan and Prince Edward Island (12.3% and 12.8%); medium rates in Nova Scotia and Manitoba (16.2% and 16.5%); and higher rates in New Brunswick and Alberta (19.4% and 19.0%).
Figure 1.1 Number and share of opioid claimants in the active beneficiary population, 2012/13

blank
|
AB |
SK |
MB |
ON |
NB |
NS |
PEI |
| Number of claimants |
98,103 |
82,777 |
130,459 |
648,569 |
22,985 |
21,912 |
4,061 |
| Total claims for opioids |
409,082 |
296,407 |
593,025 |
4,318,900 |
122,827 |
105,283 |
20,734 |
| Total drug cost for opioids |
$18,285,432 |
$10,665,037 |
$14,020,305 |
$123,248,213 |
$4,074,519 |
$3,648,784 |
$465,981 |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Despite the relatively large percentage of active beneficiaries with claims for opioids, typically these claims only represent between 2.4% and 4.3% of the public drug plan's overall claims in 2012/13, except for the NIHB, with 5.8% of drug plan claims being made for opioids (Figure 1.2). Similar findings are observed for the opioid-related drug costs, which ranged from 1.5% to 3.6% of the total drug plan costs.
Figure 1.2 Opioid claims and drug costs as a share of total drug plan claims and drug costs, by drug plan and NPDUIS National, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information; Public Drug Plan Aggregate Database, PMPRB.
The fact that this rather large beneficiary population has such a relatively small impact on the number of claims and drug costs may be explained by the generally acute nature of opioid treatment. This will be discussed in greater detail in Section 5.
Results in Figure 1.3 suggest that in most provinces, an average opioid claimant made about five or less claims for opioids in 2012/13. This amount is slightly higher in Ontario and among those covered by the NIHB, where the average is fewer than 7 claims. The average number of claims for all drugs per active beneficiary was markedly higher, ranging from 18 to 42 depending on the plan.
Figure 1.3 Average number of claims per active beneficiary, by select drug plan, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Similarly, Figure 1.4 suggests that the average cost per opioid claimant, which ranged between $83 and $190 in 2012/13, was significantly less than the average cost per active beneficiary for all drugs, which ranged between $544 and $1,392.
Note that the results reported for all active beneficiaries and all drugs reflect comorbidities and the use of multiple classes of drugs on a per patient basis. Also note that the differences in averages across plans reflect the demographic and drug plan design differences.
Figure 1.4 Average drug cost per active beneficiary, by select drug plan, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
2 Trends in Opioid Use in Public Drug Plans
Over the six-year period from 2006/07 to 2012/13, opioid use increased markedly in many of Canada's public drug plans. Depending on the public plan, the growth in use was driven by (i) a general growth in the active beneficiary population; (ii) an increase in the relative number of opioid claimants; and/or (iii) an increase in the number of claims made by each claimant.
An analysis of the indexed volume of claims for opioids from 2006/07 to 2012/13 suggests that the growth in use has been linear for the most part, with slower growth in Alberta, British Columbia and the NIHB (under 30%); with moderate rates in Ontario, Manitoba, New Brunswick and Saskatchewan (between 34.6% and 42.5%); and with faster growth in Nova Scotia and Prince Edward Island (81.1% and 86.2%, respectively) – see Figure 2.1.
Figure 2.1 Growth in approved claims for opioids, by drug plan, 2006/07–2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information; Public Drug Plan Aggregate Database, PMPRB.
To place this growth into context, with the exception of Saskatchewan, Nova Scotia and Prince Edward Island, the growth in opioid claims among the public plans was in line with, or lower than, the overall growth in all claims (Figure 2.2). The growth in claims for opioids greatly exceeds the growth in total claims in the three formerly listed public drug plans.
Figure 2.2 Growth in approved claims, opioids vs. all drugs, by drug plan, 2006/07–2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information; Public Drug Plan Aggregate Database, PMPRB.
To better understand the factors driving the growth in claims for opioids and the differences across the public drug plans, a driver of opioid use model was developed. This model captures three effects that impact the growth in claims for opioids:
1. Population effect – this effect captures the impact of the change in the size of the active beneficiary population.
2. Plan concentration – this effect captures the impact of the change in the share of opioid claimants relative to the active beneficiary population of the drug plan.
3. Intensity of use – this effect captures the impact of the change in the average number of opioid claims per claimants.
The model calculates the impact of each effect by isolating the change in the corresponding factor (e.g., population) over the 2006/07 to 2012/13 time period while holding the other two factors constant at 2006/07 values. A cross effect, resulting from the interaction between the three effects, is also reported.
This analysis is limited to the public drug plans with available beneficiary information in CIHI's NPDUIS Database. The findings provided in Table 2.1 suggest that an increase in the overall beneficiary population played an important role in the growth in claims for opioids in Nova Scotia. The growth in Saskatchewan was mainly driven by an increase in the number of opioid claimants relative to the overall beneficiary population. In Prince Edward Island, the growth appears to be driven by interplay of all three factors. Importantly, all drug plans saw significant growth – between 10% and 32% – in the number of opioid claims per claimant. The analysis of claim size within each of the provinces shows little change in the quantity dispensed per claim, eliminating the possibility that these increases could be caused by a change to smaller, more frequent prescription fills.
Table 2.1 Drivers of change in claims for opioids, by select public drug plan, 2006/07–2012/13
|
Change in beneficiary population |
Change in relative share of opioid claimants |
Change in number of claims per opioid claimant |
Cross effect |
Total growth in claims for opioids, 2006/07–2012/13 |
| Alberta |
9.6% |
-6.0% |
10.4% |
-0.3% |
13.8% |
| Saskatchewan |
9.8% |
16.3% |
11.5% |
4.8% |
42.5% |
| Manitoba |
8.7% |
-6.0% |
31.7% |
0.2% |
34.6% |
| New Brunswick |
6.9% |
1.7% |
25.5% |
12.7% |
36.4% |
| Nova Scotia |
40.6% |
4.4% |
23.3% |
12.7% |
81.1% |
| Prince Edward Island |
25.1% |
31.6% |
13.2% |
16.4% |
86.2% |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Further insight into the three drivers is provided below:
1. Population effect
Figure 2.3 reports the indexed increase in the active beneficiary population of public drug plans between 2006/07 and 2012/13. The results suggest that, while there was growth in all public drug plans, Nova Scotia and Prince Edward Island experienced the fastest growth in their active beneficiary population over this period. Thus, this was an important driver of opioid use in these public drug plans.
Figure 2.3 Change in active beneficiary population, by select public drug plan, 2006/07–2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
2. Share of opioid claimant effect
Figure 2.4 suggests that the share of opioid claimants in the active beneficiary population was quite stable between 2006/07 and 2012/13 for Alberta, Manitoba, New Brunswick and Nova Scotia. This translated into a modest impact on the growth in opioid use reported in Table 2.1. The opioid claimant's share of the active beneficiary population for these four public plans ranged between 15% and 20%. The shares of opioid claimants in Saskatchewan and Prince Edward Island increased markedly from 10% in 2006/07 to nearly 13% in 2012/13, contributing to the growth in opioid use in these two public plans.
Figure 2.4 Change in number of opioid claimants as a percentage of total active beneficiaries, by select public drug plan, 2006/07–2012/1

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
3. Individual opioid use effect
Lastly Figure 2.5 captures the change in the average number of claims made by opioid claimants for each public drug plan per fiscal year. The chart shows that all plans experienced a slight increase in the average number of claims for opioids. The largest increase occurred in Manitoba and New Brunswick, representing the equivalent of adding an extra claim per year per claimant.
Figure 2.5 Change in average number of claims per opioid claimant, by select public drug plan, 2006/07–2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
At an individual level, these increases appear to have had an impact on the amount of drugs dispensed. Figure 2.6 reports on trends in the average number of morphine equivalents (ME) per opioid claimant per year. The claimants in Manitoba seem to have had the lowest exposure to opioids (3,959 ME on average per person in 2012/13), whereas the claimants in Alberta, New Brunswick and Nova Scotia tend to have had the highest exposure to opioids (over 6,500 ME on average per person per year). While there has been an increasing trend in ME per claimant in many of the plans analyzed, Nova Scotia had the largest increase in exposure on a per claimant basis. This increase and the dip observed in Prince Edward Island are likely the result of plan design changes.
Figure 2.6 Change in morphine equivalents per opioid claimant, by select public drug plan, 2006/07–2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
3 Demographic Differences: Seniors vs. Non-Seniors
Some of the variations in opioid utilization across provinces may be explained by differences in the demographic profile of the population, the public drug plan eligibility criteria and/or the cost-sharing structures in each province. A more detailed discussion is provided in the Limitations section. Note that the analysis for Ontario and the NIHB provided in this section is limited to a single fiscal year, 2012/13. There is no trend analysis for these jurisdictions because the beneficiary information in CIHI's NPDUIS database is only partially available for this period.
The extent to which each drug plan covers their non-senior population is reflected in the relative distribution of these groups in the overall beneficiary population, as shown in Figure 3.1. With the exception of Saskatchewan, Manitoba and the NIHB, the three jurisdictions that offer universal coverage, seniors make up the majority of beneficiaries with claims for opioids, between 59% and 81% of the active beneficiary population.3
Figure 3.1 Share of total beneficiary population with claims for opioids non-seniors vs. seniors, by select public drug plan, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Despite the difference in demographic profiles across the jurisdictions, the relative share of opioid claimants within each drug plan is similar. Figure 3.2 reports the percentage of active beneficiaries in the two demographic groups with a claim for an opioid in 2012/13. In Alberta, Manitoba and Nova Scotia, the shares of opioid claimants in the senior versus the non-senior populations are very comparable, within two percentage points. In New Brunswick and Ontario, however, the difference in shares of opioid claimants in the senior/non-senior population are slightly higher, 3% more non-seniors in New Brunswick and 5% more seniors than non-seniors in Ontario. In Prince Edward Island the shares are the same.
Figure 3.2 Number and share of opioid claimants in the active beneficiary population, non-seniors vs. seniors, by select public drug plan, 2012/13
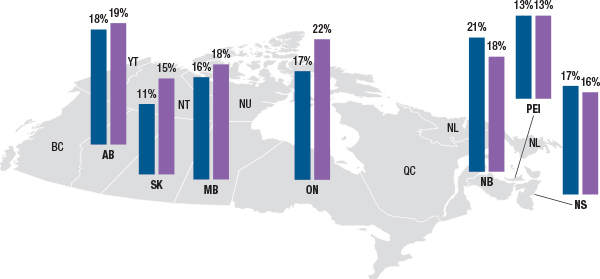
blank
|
AB |
SK |
MB |
ON |
NB |
NS |
PEI |
 Non-seniors Non-seniors |
21,850 |
59,973 |
99,834 |
224,201 |
9,938 |
4,330 |
1,205 |
 Seniors Seniors |
76,895 |
23,163 |
31,312 |
426,832 |
13,160 |
17,726 |
2,868 |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 3.3 reports the average number of claims made for opioids by seniors and non-seniors in 2012/13 by public drug plan. The results suggest that, with the exception of the NIHB (7.4 claims per beneficiary), Manitoba (6.1 claims/beneficiary) and Ontario (5.5 claims/beneficiary), the average annual opioid use among seniors is comparable across the public drug plans, between 4.0 and 4.5 claims per claimant.
In the non-senior group, however, there is more variation in the average number of opioid claims per claimant, ranging from a low of 3.2 claims per claimant in Saskatchewan to 8.8 in Ontario.
Figure 3.3 Average number of claims per opioid claimant, non-seniors vs. seniors, by select public drug plan, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The results for the senior population are relatively comparable across the public drug plans. The broad coverage of the senior population results in a large degree of homogeneity. Conversely, the results for the non-senior population are less comparable, given variations in plan coverage and design.
Figure 3.4 reports trends in the annual average opioid use for the non-senior and the senior populations from 2006/07 to 2012/13. For the senior population, the chart suggests that, with the exception of Manitoba, the average use of opioids remained relatively stable with a slight gradual increase over time, from a range of 3.3 to 4.1 in 2006/07 to a range of 4.0 to 4.5 in 2012/13, depending on the plan. The largest increase over this time period was in Manitoba, ranging from an average of 4.5 to 6.1 claims for opioids made by seniors.
In the case of non-seniors, however, the chart highlights more pronounced variations across the public drug plans. The large drop in the average number of claims in Nova Scotia in 2007/08 coincides with the launch of the Family Pharmacare program, which not only increased the number of eligible beneficiaries, but also likely changed both the disease profile and therapeutic mix of the beneficiaries represented in the administrative data.
Figure 3.4 Average number of claims per opioid claimant, non-seniors and seniors, by select public drug plan, 2006/07–2012/13


Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The individual use of opioids can also be expressed in terms of the average number of morphine equivalents (ME) per opioid claimant per year. This measure provides insight into the exposure to opioid treatment in a given population. The methodology for converting claims into morphine equivalents is explained in the Methodology section.
Figure 3.5 tracks the growth in the average number of morphine equivalents per opioid claimant between 2006/07 and 2012/13 for non-seniors and seniors. As with the average claims, the growth of morphine equivalents for seniors is fairly slow and stable, with comparable rates of utilization across the plans.
For the non-senior group, on the other hand, the variations are more pronounced, ranging in 2012/13 from a low of around 4,000 in Manitoba and Saskatchewan to a high of nearly 13,000 in Nova Scotia and Alberta. The average values for both the number of claims and morphine equivalents are lower for the two universal plans (Manitoba and Saskatchewan), which have a higher share of non-seniors (up to 77%), and higher for the two plans (Nova Scotia and Alberta) that have the lowest share of non-seniors (as low as 19%). The addition of the Family Pharmacare program in Nova Scotia has increased the share of non-seniors in the public drug plan and has resulted in a decrease in the average number of claims and morphine equivalents per claimant.
Figure 3.5 Average number of morphine equivalents per opioid claimant, non-seniors and seniors, by select public drug plan, 2006/07–2012/13


Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
4 Weaker vs. Stronger Opioids
Opioids can be grouped into two categories, weak or strong, based on their potency, which is measured in morphine equivalents (ME)4. Morphine equivalence measures the relative potency of a particular opioid and is used to standardize the impact of prescribing. The weaker opioids, such as meperidine (0.1 ME) and codeine (0.15 ME), are typically prescribed for mild to moderate pain. Stronger opioids are typically prescribed for moderate to severe pain, but could be prescribed for mild to moderate pain if a weaker opioid is found to be insufficient. The stronger opioids are morphine (1 ME), oxycodone (1.5 ME), hydromorphone (5 ME) and fentanyl (80–100 ME).
While weaker opioids are more commonly used by public drug plan beneficiaries, stronger opioids deliver the greatest exposure to opioid treatment and account for the largest share of the drug costs to the public plans.

4.1 Market Shares
In many of the public drug plans, weak opioids, like codeine (and combinations), play an important role in pain management, accounting for 40.2% of all claims for opioids. Figure 4.1.1 reports the distribution of opioids dispensed by drug plans and in NPDUIS National. The results suggest that the distribution of weaker and stronger opioid use varies significantly across plans, with weaker opioid use ranging from less than one third of claims in Ontario, New Brunswick and Prince Edward Island to close to or more than two thirds of all claims in Manitoba and the NIHB. Interestingly, oxycodone is not as widely dispensed in Saskatchewan as in other provinces. In fact, oxycodone combination drugs are not covered by the provincial plan.
Figure 4.1.1 Distribution of opioids by claims, by public drug plan and NPDUIS National, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information; Public Drug Plan Aggregate Database, PMPRB.
Given their potency, the strong opioids deliver much of the exposure to opioid treatment in public drug plans (87.2%), whereas the weak opioids account for the lowest share (12.8%). Figure 4.1.2 reports on the distribution of opioids by the total morphine equivalents of the opioids used in 2012/13.
Figure 4.1.2 Distribution of opioids by total volume of morphine equivalents, by public drug plan and NPDUIS National, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information; Public Drug Plan Aggregate Database, PMPRB.
Not only do the strong opioids deliver the most exposure to opioid treatment, but they also account for the majority of the drug costs associated with opioid use in public drug plans. Figure 4.1.3 reports on the distribution of individual drugs in the overall opioid drug costs in 2012/13. Simple oxycodone and hydromorphone accounted for 32.4% and 26.4%, respectively, of the opioid drug cost in NPDUIS National. Codeine, which was the most widely dispensed of the opioids in terms of claims, represented only 9.0% of this cost.
Figure 4.1.3 Distribution of opioids by drug cost, by public drug plan and NPDUIS National, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information; Public Drug Plan Aggregate Database, PMPRB.
The reason why opioids like oxycodone and hydromorphone represent such a large share of drug cost, despite representing only 6.8% and 19.4% of claims, is explained by their cost. For each opioid, Figure 4.1.4 reports on the average cost per claim, the average cost per opioid claimant and the average cost per unit. For all three measures, the average cost for oxycodone and hydromorphone is markedly higher than for codeine. For instance, the average price per unit for oxycodone and hydromorphone across all dosage forms and strengths was $1.74 and $0.70, respectively, whereas the price of codeine was $0.25 per unit. Only fentanyl was more expensive at an average of $8.30 per patch across the various strengths. A comparison is difficult to make, however, as a single patch is used over multiple days. In terms of morphine equivalents, the drugs are more comparable, with prices ranging from $0.02 to $0.04 per morphine equivalent.
Figure 4.1.4 Average cost of opioids, by claim and opioid claimant, for select public drug plans, 2012/13

blank
|
Meperidine |
Codeine |
Codeine Combo |
Morphine |
Oxycodone |
Oxycodone Combo |
Hydromorphone |
Fentanyl (patch) |
| Cost per unit |
$0.16 |
$0.25 |
$0.07 |
$0.47 |
$1.74 |
$0.13 |
$0.70 |
$8.30 |
| Morphine equivalents |
0.1 |
0.15 |
0.15 |
1 |
1.5 |
1.5 |
5 |
80–100 |
| Cost per morphine equivalent |
$0.03 |
$0.04 |
$0.02 |
$0.02 |
$0.04 |
$0.02 |
$0.02 |
$0.03 |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
4.2 Trends in Use
A review of the trends in opioid use in public drug plans in Section 2 reported that the average number of claims and morphine equivalents per beneficiary increased markedly between 2006/07 and 2012/13. One of the driving forces behind the growth in morphine equivalents was the increased use of more potent opioids.
Figure 4.2.1 reports on the overall increase in the average number of claims and morphine equivalents per 1000 beneficiaries between 2006/07 and 2012/13. This measure allows for the analysis of the mix of opioids used in a standardized population, as well as determining their contribution to the growth in use. The results show that, with the exception of Manitoba, the growth in morphine equivalents was greater than the increase in average claims.
Figure 4.2.1 Overall growth in claims and morphine equivalents per 1000 beneficiaries, by select public drug plan, 2006/07–2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
An analysis of how the use of individual opioids contributed to this growth indicates that the strong opioids, such as oxycodone, alone or combined with other analgesics, and hydromorphone accounted for the largest proportion of increases observed in Figure 4.2.1. Figure 4.2.2 captures the contribution that each of the opioids made to the overall growth in claims and morphine equivalents in each of the public drug plans. A result above (below) zero indicates a positive (negative) contribution to growth. The net effect adds up to 100% and refers to the increases reported in Figure 4.2.1.
In other words, Figure 4.2.2 shows that hydromorphone contributed 182% of the 11.5% increase in claims reported for Saskatchewan in Figure 4.2.1. The magnitude of the increase in claims for hydromorphone was balanced by a 48% decrease in claims for codeine and codeine combinations. Oxycodone shows little impact, as it is not as widely dispensed in Saskatchewan as in other provinces, either by itself or in combination with another analgesic.
The results suggest that between 2006/07 and 2012/13, hydromorphone, and to a lesser extent oxycodone, made the largest contribution to both the overall increase in claims and morphine equivalents across all of the drug plans.
Figure 4.2.2 Contribution to growth in average claims/morphine equivalents per 1000 beneficiaries by drug, by select public drug plan, 2006/07–2012/13

blank
|
AB |
SK |
MB |
NB |
NS |
PEI* |
| Absolute Growth (000) |
407 |
373 |
1,102 |
1,216 |
939 |
545 |
| Relative Growth |
11% |
11% |
32% |
28% |
24% |
12% |

blank
|
AB |
SK |
MB |
NB |
NS |
PEI* |
Absolute Growth
(millions) |
1,023 |
938 |
931 |
1,624 |
2,520 |
897 |
| Relative Growth |
19% |
23% |
31% |
31% |
58% |
19% |
*2009/10–2012/13
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 4.2.3 reports on the impact that each opioid had on the growth in claims per 1000 beneficiaries. The focus is on the four drugs that had the largest impact. For ease of reporting, the combination products are reported together with their respective opioid, oxycodone or codeine, while fentanyl and meperidine are combined in the “other” category.
In most of the drug plans analyzed, for a standard group of 1000 claimants, the growth in claims was driven by increases in the use of stronger opioids such as hydromorphone and oxycodone, and to a lesser extent, morphine. Except for Manitoba, the change in codeine and codeine combinations had a minimal impact on the increased number of claims per beneficiary.
Figure 4.2.3 Average number of claims per 1000 beneficiaries, by largest contributors to increase, by select public drug plan, 2006/07 vs. 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
A detailed look at oxycodone and hydromorphone shows the extent to which utilization increased over this period in selected public drug plans. Figure 4.2.4 reports on the number of claims per 1000 beneficiaries for oxycodone and hydromorphone in 2006/07 and 2012/13 along with the relative and the actual growth in claims.
The results should be interpreted as follows: in Alberta in 2006/07, a standard group of 1000 beneficiaries was dispensed 872 claims for oxycodone, whereas in 2012/13 a similar group was dispensed 1091 claims for the same drug. This represents an increase of 219 claims, translating into a 25% increase over the six-year period. Growth rates between 18% and 200% for these two drugs were observed across the provinces. The only exception occurred in utilization of oxycodone in Saskatchewan, where there was a 29% drop in the average number of claims for oxycodone per 1000 beneficiaries.
Figure 4.2.4 Growth in claims for oxycodone and hydromorphone per 1000 beneficiaries, by select public drug plan, 2006/07–2012/13


Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
4.3 Focus on Oxycodone and Hydromorphone
Oxycodone (and combinations) and hydromorphone are two of the most important contributors to the growth in opioid use in the pubic drug plans analyzed. Both are high-potency opioids used in the treatment of moderate to severe pain. Hydromorphone is also indicated for severe painful dry cough. Combined, the two drugs appear to play an important role both in the increased number of claims for opioids and in the overall growth in morphine equivalents dispensed.
Between 2006/07 and 2012/13, in most drug plans, the number of beneficiaries with claims for oxycodone products grew slowly but steadily. Figure 4.3.1 shows the growth of oxycodone claimants indexed to one in 2006/07. The attached table reports the increase in the actual number of claimants, their relative share of total opioid claimants and share of total plan beneficiaries.
Figure 4.3.1 Indexed growth in opioid claimants with claims for oxycodone by select public drug plan, 2006/07–2012/13

blank
|
Oxycodone claimants |
Number of claimants |
% of opioid claimants |
% of plan beneficiaries |
| 2006/07 |
2012/13 |
2006/07 |
2012/13 |
2006/07 |
2012/13 |
 |
AB |
17,684 |
19,416 |
19.0% |
20.2% |
3.8% |
3.8% |
 |
SK |
4,757 |
3,277 |
7.6% |
4.1% |
0.8% |
0.5% |
 |
MB |
11,790 |
13,110 |
9.3% |
10.1% |
1.6% |
1.7% |
 |
NB |
5,157 |
5,435 |
26.1% |
25.6% |
4.6% |
4.6% |
 |
NS |
1,666 |
3,079 |
11.9% |
14.9% |
1.7% |
2.3% |
 |
PEI |
656 |
1,192 |
27.2% |
29.9% |
2.6% |
3.8% |
| Total |
|
41,710 |
45,509 |
13.1% |
12.9% |
2.0% |
2.0% |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The results suggest that, with the exception of Nova Scotia and Prince Edward Island, the growth in the number of oxycodone claimants was steady until 2010/11 when the numbers began to decline, most notably in Manitoba and Saskatchewan. Generally, oxycodone, either by itself or in combination with another analgesic, is not as widely used in Saskatchewan as in other provinces. The growth in Nova Scotia and Prince Edward Island coincides with plan changes that expanded the overall beneficiary population. The growth in claimants slowed in 2011/12 in all of the provinces tracked, coinciding with the switch from OxyContin to OxyNEO that occurred at the beginning of 2012/13. This switch will be discussed in greater detail later in the section.
The number of hydromorphone claimants, on the other hand, increased at a steady high rate for all selected public drug plans. Figure 4.3.2 shows the growth of beneficiaries with claims for hydromorphone indexed to one in 2006/07. The attached table reports the increase in the actual number of claimants, their share of total opioid claimants and share of total plan beneficiaries.
Figure 4.3.2 Indexed growth in opioid claimants with claims for hydromorphone, by select public drug plan, 2006/07–2012/13

blank
|
Hydromorphone claimants |
Number of claimants |
% of opioid claimants |
% of plan beneficiaries |
| 2006/07 |
2012/13 |
2006/07 |
2012/13 |
2006/07 |
2012/13 |
 |
AB |
3,471 |
6,675 |
3.7% |
6.9% |
0.7% |
1.3% |
 |
SK |
11,731 |
26,832 |
18.9% |
33.4% |
1.9% |
4.0% |
 |
MB |
3,050 |
7,909 |
2.4% |
6.1% |
0.4% |
1.0% |
 |
NB |
2,545 |
3,950 |
12.9% |
18.6% |
2.3% |
3.3% |
 |
NS |
3,232 |
7,326 |
23.0% |
35.4% |
3.4% |
5.4% |
 |
PEI |
315 |
623 |
13.1% |
15.6% |
1.2% |
2.0% |
| Total |
|
24,344 |
53,315 |
7.7% |
15.2% |
1.2% |
2.4% |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The data suggests that the number of beneficiaries with claims for hydromorphone more than doubled in Manitoba, Saskatchewan and Nova Scotia between 2006/07 and 2012/13. These increases are also reflected in the share of beneficiaries with claims for hydromorphone relative to other opioids. In Saskatchewan and Nova Scotia, hydromorphone was dispensed to more than one third of opioid claimants in 2012/13.
Between 2006/07 and 2012/13, the drug costs related to oxycodone use grew slightly faster than the actual growth in claims. Figure 4.3.3 reports the trends in growth of oxycodone drug costs, while the attached table reports the actual oxycodone drug cost and the share of this drug cost in both the total opioid drug costs and all drug costs.
Figure 4.3.3 Indexed growth in drug costs for oxycodone, by select public drug plan, 2006/07–2010/11

blank
|
Oxycodone drug costs |
Amount (000) |
% of opioid drug costs |
% of plan drug costs |
| 2006/07 |
2012/13 |
2006/07 |
2012/13 |
2006/07 |
2012/13 |
 |
AB |
$6,812 |
$8,890 |
54.2% |
54.2% |
1.1% |
1.3% |
 |
SK |
$1,977 |
$1,973 |
34.3% |
22.2% |
0.7% |
0.5% |
 |
MB |
$3,959 |
$4,336 |
44.3% |
38.3% |
0.9% |
0.9% |
 |
NB |
$1,000 |
$1,298 |
39.1% |
36.1% |
0.7% |
0.8% |
 |
NS |
$185 |
$477 |
13.5% |
15.0% |
0.1% |
0.3% |
 |
PEI |
$52 |
$122 |
21.0% |
28.8% |
0.2% |
0.4% |
| Total |
|
$13,985 |
$17,096 |
44.5% |
39.0% |
0.9% |
0.9% |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The indexed growth cost for oxycodone for Nova Scotia and Prince Edward Island is markedly higher than the indexed growth in the respective claimants, due to the relatively higher cost for oxycodone compared to other opioids on the market. The dramatic dip in Manitoba's trend beginning in 2009/10 appears to be related to the increased use of oxycodone combination products, which are not as expensive as oxycodone alone. In Alberta, oxycodone's relative cost compared to other opioids remained unchanged.
Figure 4.3.4 shows that, as with oxycodone, the growth in drug costs for hydromorphone was generally higher than the growth in the claimants of this drug. The largest growth occurred in Saskatchewan and Nova Scotia, where hydromorphone costs represented 57% and 58% of the total opioid cost in 2012/13.
Figure 4.3.4 Indexed growth in drug costs for hydromorphone, by select public drug plan, 2006/07–2010/11

blank
|
Hydromorphone drug costs |
Amount (000) |
% of opioid drug costs |
% of plan drug costs |
| 2006/07 |
2012/13 |
2006/07 |
2012/13 |
2006/07 |
2012/13 |
 |
AB |
$1,965 |
$3,502 |
16% |
21% |
0.3% |
0.5% |
 |
SK |
$1,778 |
$5,073 |
31% |
57% |
0.6% |
1.4% |
 |
MB |
$1,546 |
$3,295 |
17% |
29% |
0.4% |
0.7% |
 |
NB |
$890 |
$1,510 |
35% |
42% |
0.6% |
0.9% |
 |
NS |
$668 |
$1,840 |
49% |
58% |
0.5% |
1.2% |
 |
PEI |
$51 |
$108 |
20% |
25% |
0.2% |
0.3% |
| Total |
|
$6,897 |
$15,327 |
22% |
35% |
0.4% |
0.8% |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The switch from OxyContin to OxyNEO
In February 2011, the manufacturer of OxyContin, the extended-release formulation of oxycodone, announced plans to remove the product from the Canadian market. To replace it, Purdue Pharma launched a newer version of the drug, OxyNEO, that boasted features designed to prevent drug abuse. The switch from OxyContin to OxyNEO had an important impact on the amount of oxycodone dispensed to beneficiaries of the public drug plans. All the provincial plans, except for Alberta, decided that OxyContin and OxyNEO would not be interchangeable and placed OxyNEO under their exceptional access programs, thus restricting the reimbursement of extended-release oxycodone. Typically, only patients that had already been prescribed OxyContin for at least 3 months continued to receive coverage for the new formulation.
Note that the following analysis was done for claims of the patented version of extended-release oxycodone only. Generic versions of the drug were authorized by Health Canada in November 2012, but were not included in the data.
Figure 4.3.5 reports on the morphine equivalents of extended-release oxycodone from the first quarter of 2010/11 to the fourth quarter of 2012/13. It shows that in the two years preceding the launch of OxyNEO, the amount of morphine equivalents dispensed grew steadily across the public plans. With the launch of OxyNEO, however, the volume dropped significantly to 54.4% of its high point the year earlier.
Figure 4.3.5 Change in morphine equivalents dispensed for extended-release oxycodone, 1st quarter 2010/11 to 4th quarter 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
While much of the overall drop was influenced by declining utilization rates in Ontario, the majority of provinces were affected. Figure 4.3.6 shows the use of oxycodone in terms of morphine equivalents across the selected provinces indexed to the first quarter of 2011/12. It shows that, with the exception of Saskatchewan and Prince Edward Island, the growth in claims for extended-release oxycodone was relatively flat in the year before OxyNEO was launched.
Figure 4.3.6 Change in number of morphine equivalents for extended-release oxycodone, by select public drug plan, 1st quarter 2011/12 to 4th quarter 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The decline in the use of extended-release oxycodone following the switch to OxyNEO is equally noticeable among high-use claimants (those with greater than 10 claims for opioids per year). Figure 4.3.7 shows that across all of the provinces studied, the number of morphine equivalents dispensed continued to grow at a steady pace until the introduction of OxyNEO, after which quantities dropped dramatically. This occurred despite the fact that many of these patients may have been eligible to switch to the new drug by virtue of having been treated with OxyContin for longer than 3 months. Initially in Alberta claims continue to grow, though at a much slower pace, before there too the volume began to decline. High-use claimants and their impact on utilization are discussed in greater detail in the next section.
Figure 4.3.7 Change in number of morphine equivalents for extended-release oxycodone by high-use claimants of opioids, by select public drug plan, 1st quarter 2011/12 to 4th quarter 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
5 Low- vs. High-use Claimants of Opioids
This section provides an assessment of the drug utilization patterns of low- versus high-use opioid claimants based on their number of claims per year. The analysis is limited to the following selected public drug plans with available claim-level data: Alberta, Saskatchewan, Manitoba, Ontario, New Brunswick, Nova Scotia and Prince Edward Island.
5.1 Patterns of Opioid Use
One of the primary reasons why opioid claimants, which represent up to one-fifth of the drug plan beneficiaries, account for a much smaller share of claims or cost is the generally acute nature of opioid treatment. An analysis of the data of selected NPDUIS public drug plans indicates that the majority of opioid claimants are treated for only a short period of time, in terms of their number of claims, and are typically prescribed weaker opioids.
The claims data from 2006/07 to 2012/13 indicates that the majority of patients are dispensed one or two opioids during a year. Figure 5.1.1 reports on the distribution of beneficiaries based on the number of claims for opioids they made in 2012/13. It shows that more than 60% of all beneficiaries made only one or two claims per year. This percentage is higher in Saskatchewan and Manitoba, with 73.2% and 69.4% of the total opioid claimants, respectively.
Figure 5.1.1 Distribution of opioid claimants by number of claims per year, by select public drug plan, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Note that the differences across plans may be explained by plan design and demographics, as the public drug plans in Saskatchewan and Manitoba, which have income-based coverage, have a higher percentage of claimants making only 1 or 2 claims. These plans also have the lowest average number of claims and lowest average morphine equivalents, as discussed in Section 3.
Beneficiaries that made more than 10 claims for opioids accounted for between 9.9% (Saskatchewan) and 19.9% (NIHB) of all opioid claimants in 2012/13. Despite being a relatively small group, these high-use claimants of opioids account for the majority of opioid claims, opioid costs and morphine equivalents dispensed. Table 5.1.1 reports on the relative importance that this beneficiary group plays in opioid utilization across the select public drug plans. In Saskatchewan, for example, despite representing only 9.9% of opioid claimants, high-use claimants account for 51.5% of opioid claims, 83.9% of drug costs and 82.0% of the morphine equivalents dispensed.
Table 5.1.1 Market share of high-use opioid claimants (>10 claims per year), by select public drug plan, 2012/13
|
Alberta |
Saskatchewan |
Manitoba |
Ontario |
New Brunswick |
Nova Scotia |
Prince Edward Island |
| Share of total opioid claimants |
10.1% |
9.9% |
11.5% |
17.7% |
16.7% |
13.7% |
15.1% |
| Share of total claims – opioids |
47.3% |
51.5% |
59.6% |
69.1% |
62.8% |
52.6% |
59.6% |
| Share of total drug cost – opioids |
65.9% |
83.9% |
80.6% |
84.0% |
84.5% |
70.1% |
81.0% |
| Share of morphine equivalents |
64.5% |
82.0% |
77.1% |
81.5% |
82.8% |
70.4% |
79.2% |
| Average claims per opioid claimant |
19.4% |
18.7% |
23.4% |
26.1% |
20.7% |
18.5% |
20.4% |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
High-use claimants are generally more likely to be dispensed higher potency opioids. Figure 5.1.2 shows how much of each of opioid is dispensed among the four groups, as a global figure for the select public plans. Codeine seems to be predominantly used by claimants making fewer claims, whereas higher potency opioids are more often dispensed to those with a higher treatment frequency.
Figure 5.1.2 Distribution of opioids by beneficiary group, by select public drug plan, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The opioid treatment intensity in the high-use claimant group is exacerbated by the potency of the opioids they are using. Figure 5.1.3 reports on the average number of morphine equivalents per claim, as well as the average number of morphine equivalents per opioid claimant by beneficiary group. Results are reported for the selected public drug plans. The results suggest the number of morphine equivalents increases significantly with an increase in the number of claims. When compared to other groups, high-use claimants, on average, made more than twice as many claims for an opioid (24.7 compared to 7.7) and were dispensed more than three times the morphine equivalents (36,916/claimant compared to 9,056/claimant) in a year than the group with the next largest utilization, those making between 6 and 10 claims per year.
Figure 5.1.3 Average number of morphine equivalents (ME), per claim and per opioid claimant, by beneficiary group and select public drug plan, 2012/13

blank
|
1-2 |
3-5 |
6-10 |
>10 |
| Average claims per claimant |
1.2 |
3.8 |
7.7 |
24.7 |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
5.2 Trends in Opioid Treatment Intensity
A review of the trends in opioid use in public drug plans in Section 2 reported that the average number of claims and morphine equivalents per beneficiary increased markedly between 2006/07 and 2012/13. This section provides insight into the role that treatment intensity has had in this growth.
Over the six-year period, the percentage of beneficiaries with more than 10 claims grew steadily. Figure 5.2.1 reports the percentage share that this group represents of the total beneficiaries with a claim for an opioid. The data to calculate the 2006/07 percentage for Ontario is not available. During this period, the share of high-use claimants of opioids increased between 1.7% in Saskatchewan and 4.5% in New Brunswick.
Figure 5.2.1 Share of opioid claimants with more than 10 claims, by year, by select public drug plan, 2006/07 vs. 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
In addition to the relative increase in high-use claimants, the share of higher potency drugs has generally increased for all beneficiary groups. The next four charts report the distribution of claims by individual drugs, corresponding to the four beneficiary categories.
For the low-use claimants, making only 1 or 2 opioid claims, the weak opioids dominated the treatment of pain. However, there appears to be a small increase in the share of stronger opioids in 2012/13 compared to 2006/07 (Figure 5.2.2). Across all of the public drug plans studied, this beneficiary group represents approximately 60% of all opioid claimants in 2012/13.
Figure 5.2.2 Distribution of claims by drug for opioid claimants with 1 or 2 claims, by select public drug plan, 2006/07 vs. 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
A similar pattern in utilization can be seen for opioid claimants making between 3 and 5 claims in a year. While weak opioids still dominate the drug-mix within this group, Figure 5.2.3 reveals the increasing importance of stronger opioids in the treatment of pain. Across the drug plans this beneficiary group represents approximately 15% of all opioid claimants.
Figure 5.2.3 Distribution of claims by drug for opioid claimants with 3 to 5 claims, by select public drug plan, 2006/07 vs. 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
As for the previous two beneficiary groups, Figure 5.2.4 shows that for claimants with 6 to 10 claims for opioids, stronger opioids play an increasing role in the treatment of pain. This is the smallest of the beneficiary groups, representing about 10% of the opioid claimants.
Figure 5.2.4 Distribution of claims by drug for opioid claimants with 6 to 10 claims, by select public drug plan, 2006/07 vs. 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Of the four beneficiary groups, the claimants that make more than 10 claims per year have the highest strong opioid use. Figure 5.2.5 captures the market share for each drug for these high-use claimants. This beneficiary group represents approximately 16% of all claimants.
For all four beneficiary groups, oxycodone combination drugs and hydromorphone appear to be the treatment of choice among the stronger opioids. Figure 5.2.5 Distribution of claims by drug for opioid claimants with more than 10 claims, by select public drug plan, 2006/07 vs. 2012/13
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 5.2.5 Distribution of claims by drug for opioid claimants with more than 10 claims, by select public drug plan, 2006/07 vs. 2012/13

The widespread increase in the use of stronger opioids across all beneficiary groups has led to an increase in the morphine equivalents used, on average, by each group. Figure 5.2.6 reports the change in the average number of morphine equivalents per opioid claimant by beneficiary group in 2006/07 versus 2012/13.
Figure 5.2.6 Percentage change in the average number of morphine equivalents per patient, by beneficiary group, by select public drug plan, 2006/07 vs. 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The results suggest that the morphine equivalents dispensed increased for most beneficiary groups and plans, with a low of 1.5% in Saskatchewan and a high of 40.5% in Nova Scotia. Much of Nova Scotia's increase was probably driven by the introduction of the Family Pharmacare plan in 2007. Both Saskatchewan and Prince Edward Island experienced declines in average morphine equivalents per patient in two of their beneficiary groups during this time.
5.3 Treatment Patterns of High-use Claimants of Opioids
High-use claimants are defined as public drug plan beneficiaries with more than 10 opioid claims per year. As the previous analysis suggests, although this group represents a small proportion of the total opioid claimants, they account for the majority of the claims and related costs. Moreover, these patients represent an increasing share of the opioid claimants. This section analyses how the treatment patterns of the high-use claimants has evolved over time.
For this analysis, two cohorts of patients were identified based on the same selection criteria but at two points in time: a 2008/09 cohort and a 2012/13 cohort. The patients were retained in each cohort if they were high-use claimants of opioids, namely, they made more than 10 claims in both 2008/09 and 2012/13. For this analysis, 2008/09 and 2012/13 are referred to as Year 0. The size of the cohort groups can be found in Figure 5.3.1.
Figure 5.3.1 Share of high-use claimants in the opioid claimant population, by select public drug plan, 2008/09 and 2012/13

blank
| Cohort size |
AB |
SK |
MB |
NB |
NS |
PEI |
| 2008/09 |
8,271 |
6,278 |
11,711 |
2,751 |
1,837 |
387 |
| 2012/13 |
9,733 |
7,880 |
14,794 |
3,566 |
2,844 |
596 |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 5.3.1 reports on the share of high-use claimants as a proportion of all opioid claimants, along with the cohort size in 2008/09 and 2012/13 for each of the selected public drug plans. The increased share in 2012/13 is in line with the increasing trend in high-use claimants reported in Section 5.2.
These high-use claimants were followed back for two years to 2006/07 and 2010/11, respectively (Year -2).
Only the new and the existing high-use claimants of opioids were retained for the analysis.
- Existing high-use claimants made 3 or more claims for opioids in Year -2.
- New high-use claimants made less than 3 claims for opioids (or no claims) in Year -1 (2007/08 or 2011/12).
The remaining claimants were not retained in the analysis.
Figure 5.3.2 reports the breakdown of the patients in each cohort by new and existing and by select public drug plan. The results suggest that the majority are existing high-use claimants.
The relative size of the existing population of each cohort is comparable across the selected public drug plans, in the 60%–70% range. Nova Scotia had a relatively lower share of existing claimants in 2008/09 (53%). This increased in 2012/13 (66%), probably as a result of the introduction of Family Pharmacare the year before.
Figure 5.3.2 Distribution of new and existing high-use claimants in the cohort, by select public drug plan, 2008/09 vs. 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
1. Existing high-use claimants
The results suggest that over time not only did these claimants make more claims per year, but they made claims for more potent drugs, especially oxycodone and hydromorphone.
Figure 5.3.3 follows the existing claimants in the 2012/13 cohort, tracking the average number of claims they made over a three-year period: 2010/11 (Year -2), 2011/12 (Year -1) and 2012/13 (Year 0).
In Year -2, the claimants in five of the six drug plans analysed made, on average, between 14 and 17 claims per year. Over the next two years, the average typically increased by 4 claims per year, ranging in 2012/13, between 19 and 24 claims per claimant per year.
Not only did the average number of claims per beneficiary increase in this population, but the share of higher potency drugs increased as well.
Figure 5.3.3 Average number of claims per existing high-use claimants of opioids, by select public drug plan, 2012/13 cohort

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 5.3.4 reports on the distribution of claims by drug for the existing high-use claimants of opioids in each of the years analyzed. With the exception of Prince Edward Island, the results show a steady increase in the share of high-potency drugs used by these claimants. This is especially the case for hydromorphone and oxycodone.
Figure 5.3.4 Distribution of drugs by claims made by the existing high-use claimants of opioids, by select public drug plan, 2012/13 cohort

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figures 5.3.5 to 5.3.7 report the percentage share of all claims made by existing claimants of oxycodone, hydromorphone and the two opioids combined for the 2008/09 and the 2012/13 cohorts. The results suggest that the combined share of oxycodone and hydromorphone is comparable across the selected public plans.
Figure 5.3.5 Oxycodone claims as a percentage of all opioid claims, existing high-use claimants, by select public drug plan, Year 0

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 5.3.6 Hydromorphone claims as a percentage of all opioid claims, existing high-use claimants, by select public drug plan, Year 0

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 5.3.7 Oxycodone and hydromorphone claims as a percentage of all opioid claims, existing high-use claimants, by select public drug plan, Year 0

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The main finding for the existing high-use claimants of opioids is that the 2012/13 cohort used more of these two potent drugs than the 2008/09 cohort (Figure 5.3.7).
Nevertheless, depending on the plan, the opioid treatment may be delivered through a varying mix of these two drugs. Comparing Figures 5.3.5 and 5.3.6, regardless the cohort, oxycodone use accounted for a larger share of the market in Alberta, New Brunswick and Prince Edward Island, and to a lesser extent Manitoba. On the other hand, hydromorphone use dominated the market in Saskatchewan and Nova Scotia. The two figures also show a significant switch from oxycodone to hydormorphone between the two cohorts in Saskatchewan and Prince Edward Island.
When the two treatments are combined, however, their market share of total claims is more comparable across the drug plans, representing around 50% or more of the total. In New Brunswick this percentage is a little higher at around 60% and lower in Manitoba at 40%. It also shows that there has been a slight increase in the use of these two drugs across all public drug plans. This suggests that the two drugs were possibly being prescribed interchangeably for moderate to severe pain.
2. New high-use claimants
The two cohorts of new high-use claimants made a similar number of claims in their first year. Figure 5.3.8 reports on the average number of claims made by the new patients of the two cohorts. The chart indicates that the opioid claimants in these groups made on average between 16 and 19 claims for opioids in their first year of high opioid use, depending on the plan.
Figure 5.3.8 Average number of claims per year, new high-use claimants, by select public drug plan, 2008/09 vs. 2011/12

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
An analysis of the two most widely used opioids in this group, oxycodone and hydromorphone, reveals some similarities with the existing high-use claimants. Figures 5.3.9 and 5.3.10 capture the share of total claims made for oxycodone and hydromorphone for the new high-use claimants.
Figure 5.3.9 Oxycodone claims as a percentage of all opioid claims, new high-use claimants, by select public drug plan, Year 0

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 5.3.10 Hydromorphone claims as a percentage of all opioid claims, new high-use claimants, by select public drug plan, Year 0

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The main finding for the new high-use claimants of opioids is that fewer claims for oxycodone were made by new patients in 2012/13 compared to their 2008/09 counterparts. While the switch from OxyContin to OxyNEO, which occurred at the beginning of 2012/13, may have played a role, a review of the data suggests that this trend began at least the year before.
3. Existing versus New High-use Claimants of Opioids
The previous two subsections suggest that the existing high-use claimants of opioids make, on average, more claims than the new high-use claimants. Nevertheless, when comparing the potency of the opioids used by the two groups, the existing high-use claimants did not always use the stronger opioids to as great an extent as their new counterparts. In fact, in Alberta, Saskatchewan and Nova Scotia, the new high-use claimants had a higher share of stronger opioids than the existing claimants, the difference being mainly driven by hydromorphone.
Figure 5.3.11 compares the distribution of opioids by claim for the new and existing patients in the 2012/13 cohort.
Figure 5.3.11 Distribution of opioids by claims, by province, new vs. existing patients, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The differences in the results may be explained by the disease profiles of the new versus the existing group. The next section provides insight into the concomitant drug use for the high-use claimants of opioids.
5.4 Focus on High-use Claimants of Opioids – Concomitant Drug Use
Generally speaking, the majority of high-use claimants of opioids have other non-opioid drug use, which may indicate other underlying health conditions. While diagnostic information is not available in the administrative data to point towards the potential cause of pain, the information does offer some insight into the other therapeutic treatment areas used by these patients.
Table 5.4.1 reports the average claims per claimant and drug cost per claimant for opioid and non-opioid drugs for high-use claimants of opioids by selected drug plan in 2012/13. The results suggest that the large majority of high-use claimants make many more claims for other drugs. This indicates that the number of claims and drug costs related to pain treatment represent only a part of the overall drug utilization of this group.
Table 5.4.1 Average claims per user and drug cost per user for opioid and non-opioid drugs for high-use claimants of opioids, by select public drug plan, 2012/13
|
AB |
SK |
MB |
ON |
NB |
NS |
PEI |
| Opioid drug use |
Average claims per claimant |
19 |
19 |
23 |
26 |
21 |
19 |
20 |
| Average cost per claimant |
$1,204 |
$1,100 |
$745 |
$912 |
$938 |
$867 |
$614 |
| Non-opioid drug use |
Average claims per claimant |
76 |
59 |
109 |
144 |
85 |
69 |
70 |
| Average cost per claimant |
$2,858 |
$1,966 |
$2,506 |
$2,508 |
$2,854 |
$2,427 |
$1,789 |
| Number of claimants with claims only for opioids |
29 of 9,762 |
119 of 7,880 |
158 of 14,794 |
585 of 111,758 |
21 of 3,566 |
14 of 2,844 |
N/A |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Table 5.4.2 lists the top 10 therapeutic classes used by this group, in terms of the share of claimants dispensed the drugs. It also reports the average number of claims made by this group for each class of drug. This list includes drugs that may be used to treat conditions related to pain: N06A – antidepressants, to treat the depression of living with pain; N03A – anti-epileptics (gabapentin), which are often used to treat neuropathic pain; N05B – anxiolytics (benzodiazepines), to assist patients with sleep or deal with anxiety; A06A – drugs for constipation, to counter one of the most common side-effects of opioid use.
Other drugs on the list may be dispensed to treat general health conditions likely unrelated to the pain. These include C10A – lipid modifying agents; C07A – beta blocking agents; C09A – ace inhibitors; C03C – high-ceiling diuretics used in the treatment of high-blood pressure and heart disease; A02B – drugs for peptic ulcer disease and GORD; and N05A – antipsychotics.
Table 5.4.2 Concomitant drug use top 10 ATC-3 therapeutic classes for high-use claimants of opioids, by select public drug plan, 2012/13
|
ATC level 3 |
Share of high-use claimants |
Average claims per high-use claimant |
| 1 |
N06A |
Antidepressants |
59% |
26 |
| 2 |
A02B |
Drugs for peptic ulcer and gastro-oesophageal reflux disease (GORD) |
50% |
18 |
| 3 |
N03A |
Antiepileptics |
33% |
20 |
| 4 |
C10A |
Lipid modifying agents, plain |
37% |
17 |
| 5 |
N05A |
Antipsychotics |
22% |
25 |
| 6 |
A06A |
Drugs for constipation |
34% |
15 |
| 7 |
N05B |
Anxiolytics |
34% |
14 |
| 8 |
C07A |
Beta blocking agents |
24% |
18 |
| 9 |
C09A |
Ace inhibitors, plain |
25% |
17 |
| 10 |
C03C |
High-ceiling diuretics |
21% |
20 |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
The extent to which these drugs are dispensed within this group is fairly consistent across the public drug plans. Figure 5.4.1 captures the percentage of high-use opioid claimants with claims for the top six ATC-3 classes reported above. Between 83.0% and 93.6% of these claimants were prescribed at least one of these six drug classes.
Figure 5.4.1 Percentage of high-use claimants of opioids with claims for at least one of the top five ATC-3 treatments, by select public drug plan, 2012/13

Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure 5.4.2 offers more detail into the extent to which these top five drug categories are used by the high-use claimants across the selected public drug plans. The bar chart reports on the percentage of claimants utilizing each of these drug classes, while the table reports on the average number of claims each claimant made. The data shows that in most plans more than 50% of high-use opioid claimants made claims for the top two drug categories, with the A02B drugs dominating in Alberta, New Brunswick, Nova Scotia and Prince Edward Island.
Figure 5.4.2 Treatment intensity of top five ATC-3 therapeutic classes of high-use claimants of opioids, by select public drug plan, 2012/13

blank
| Average claims per opioid claimant |
|
AB |
SK |
MB |
ON |
NB |
NS |
PEI |
| N06A - Antidepressants |
 |
14 |
11 |
22 |
28 |
15 |
13 |
13 |
| A02B - Drugs for peptic ulcer/GORD |

|
9 |
9 |
15 |
18 |
10 |
8 |
10 |
| N03A - Antiepileptics |

|
12 |
10 |
19 |
22 |
13 |
10 |
12 |
| C10A - Lipid modifying agents |

|
9 |
10 |
14 |
19 |
9 |
8 |
10 |
| N05A - Antipsychotics |

|
12 |
10 |
27 |
27 |
16 |
11 |
17 |
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
6 Conclusion
An analysis of public drug plan data between 2006/07 and 2012/13 shows that the utilization of opioids both in terms of claims and the potency of the drugs dispensed has increased. While in some cases, most notably in Nova Scotia and Prince Edward Island, much of the increase was related to changes in plan design, the data suggests a gradual increase in the number of opioid claimants, the average number of claims they made and the potency of the drugs that were dispensed.
Although 12% to 20% of the beneficiary population made a claim for an opioid in 2012/13, they accounted for relatively a small percentage of the total drug plan claims, on average 3.4%. The reason for this discrepancy is that most opioid claimants were probably acute patients, with around 60% of claimants making only 1 or 2 claims in a given year. Typically this group was dispensed weaker opioids, ranging from between 47.8% of the total opioid claims in Nova Scotia to 79.9% in Manitoba in 2012/13.
The data shows a variation in utilization patterns among the various public drug plans. Much of this variation could be related to plan design. The senior population, the demographic with the most consistent coverage across the plans, shows some consistency in terms of the number of claims and morphine equivalents dispensed. This is not the case for non-seniors. In Ontario, New Brunswick, Nova Scotia and Prince Edward Island, claimants under 65 made, on average, twice as many claims in 2012/13 as the same group in Saskatchewan and Manitoba. The lowest average use per claimant in terms of claims and morphine equivalents occurred in Saskatchewan and Manitoba, the two provinces offering universal benefits and with the highest percentage of beneficiaries under 65. A noticeable shift downward in both these averages occurred in Nova Scotia, coinciding with the launch of its Family Pharmacare program in 2007.
An important demographic from the standpoint of utilization is the group of claimants making more than 10 claims for an opioid per year. While this group of high-use claimants represented between 10% and 18% of all opioid claimants in 2012/13, they were responsible for between 47% and 69% of all opioid claims and as much as 85% of opioid drug costs and 83% of the share of morphine equivalents dispensed. When compared to other groups, these high-use claimants, on average, made more than twice as many claims for an opioid (24.7 compared to 7.7) and were dispensed more than three times the morphine equivalents (36,916/claimant compared to 9,056/claimant) in a year than the group with the next largest utilization, those making between 6 and 10 claims per year.
One of the major factors driving the increase in morphine equivalents was the increase in the dispensing of more potent opioids, particularly oxycodone and hydromorphone. Combined, the average number of claims per 1000 beneficiaries for these drugs grew 60.6% in the drug plans studied between 2006/07 and 2012/13. Hydromorphone alone grew 125.3%. This increase did not occur in the high-use claimant group alone. Generally all beneficiary groups were dispensed more potent opioids in 2012/13 than in 2006/07, regardless of how many claims they made in a year.
Appendix A – Prescription Drug Monitoring Programs
blank
| Public drug plan |
Monitoring program / agency |
Description |
Other |
|
British Columbia
|
Prescription Review Program / College of Physicians and Surgeons of British Columbia |
Using the PharmaNet database, the College performs periodic reviews of the prescribing practices of controlled substances in the province looking for possible evidence of abuse, misuse or diversion. Prescribers are notified of specific incidents of concern. The purpose of the program is educational rather than disciplinary. |
PharmaNet is a database managed by the BC Ministry of Health that captures prescription drugs dispensed in the province. The Controlled Prescription Program is a duplicate prescription program administered by the College of Pharmacists of BC designed to prevent forgeries and reduce inappropriate prescribing of selected drugs. |
|
Alberta
|
Triplicate Prescription Program / College of Physicians and Surgeons of Alberta |
The purpose of the program is to monitor the use of drugs prone to misuse and abuse for non-medical purposes. A copy of each prescription issued is sent by the dispensing pharmacist to the program for manual entry. Prescribers are notified if high-risk use or prescribing patterns by patients are identified. |
Physician Prescribing Practices Program is an initiative of the College, offering physicians educational materials, peer support and practice tools as a way of contributing to the overall quality of prescribing in Alberta.
Alberta Netcare is the province's public electronic health record service. Authorized prescribers and pharmacists have access to a patient's prescription and dispensing information. Coverage is not yet universal.
|
|
Saskatchewan
|
The Prescription Review Program / College of Physicians and Surgeons of Saskatchewan |
The objective of the Prescription Review Program (PRP) is to reduce the abuse, misuse and diversion of a select panel of prescription drugs. Pharmacies submit prescription data on benefit and non-benefit drugs to the Drug Plan's electronic network for Drug Plan beneficiaries and non-beneficiaries who have a Saskatchewan Health Services card. The Drug Plan electronically provides specific data on monitored drugs to the PRP. The PRP reviews the data and either alerts physicians of possible inappropriate use or requires an explanation of prescribing practices. The PRP provides education to encourage appropriate prescribing, and may pursue regulatory intervention if required. |
The Ministry also administers the Pharmaceutical Information Program, which is a secure, web-based computer application that provides authorized health care users with access to medication histories of Saskatchewan patients and other tools to help the users select the best medication, avoid drug interactions, avoid duplication of therapy and identify inappropriate drug dosages. |
|
Manitoba
|
Manitoba Prescribing Practices Program / Manitoba Pharmaceutical Association |
The Manitoba Prescribing Practices Program is a multi-prescription program aimed at promoting and supporting appropriate drug use management. Prescribers complete prescriptions on a personalized duplicate pad. The original is sent to the pharmacist and a copy is kept by the prescriber. Based on the information supplied on the prescription and by the patient, the pharmacist decides whether or not to fill the prescription. The prescriber is notified about the decision not to fill. All prescriptions dispensed are entered into the provincial Drug Programs Information Network database. |
The Manitoba Drug Programs Information Network maintains a database of all prescriptions for all Manitoba citizens regardless of how they are reimbursed. The program and database are administered by the Manitoba government. |
|
Ontario
|
Narcotics Monitoring System / Ontario Ministry of Health and Long-Term Care |
The Narcotics Monitoring System is a centralized database of all prescribing and dispensing activities of monitored drugs in the province. In addition to using the data to identify possible inappropriate prescribing practices and utilization, the Ministry uses the information for educational and public health purposes. The system also warns the pharmacist at time of dispensing of possible double-doctoring, poly-pharmacy use, early or late refilling or duplicate dispensing. |
|
|
New Brunswick
|
An electronic prescription monitoring system is under development |
The proposed system will allow community pharmacists to view a patient's history of narcotics and controlled drugs in real-time prior to dispensing a prescription for monitored drugs. It will also allow doctors and dentists to see a patient's prescription history. |
|
|
Nova Scotia
|
Nova Scotia Prescription Monitoring Program / Medavie Blue Cross |
Nova Scotia Prescription Monitoring Program was established to promote the appropriate use of monitored drugs and to reduce their abuse or misuse. Prescriptions of all monitored drugs are entered into the program's database by the pharmacist at dispensing. Since 2012, prescribers and pharmacists have had access to patient profiles, and claims are processed in real time. Reviews of the data are made on a monthly basis and incidents of inappropriate prescribing or use are communicated to the appropriate prescribers. |
|
|
Prince Edward Island
|
N/A |
N/A |
The Pharmaceutical Information Program captures information on all prescriptions dispensed to Island residents. This system is not designed to uniquely monitor the prescription of controlled substances. All prescriptions are tracked, and patient profiles are available to prescribers and pharmacists in real time. |
|
Newfoundland and Labrador
|
Tamper resistant prescription drug pad program / The Department of Health and Community Services, in partnership with the province's medical, dental and veterinary colleges and professional associations and the Royal Newfoundland Constabulary |
The purpose of tamper resistant prescription drug pads is to reduce prescription drug abuse and diversion by reducing the likelihood for prescription forgeries and/or alterations. The tamper resistant prescription pads contain a number of security features that make it difficult to duplicate or alter. These features assist prescribers to fulfill their professional responsibilities by allowing appropriate access of these medications to patients who require them, while being vigilant against some of the factors that facilitate drug abuse and drug diversion. |
It is important to note that the tamper resistant drug pad program is NOT a monitoring program. The purpose of tamper resistant prescription drug pads is to reduce prescription drug abuse and diversion by reducing the likelihood for prescription forgeries and/or alterations. The tamper resistant prescription pads contain a number of security features that make it difficult to duplicate or alter. |
|
NIHB
|
Prescription Monitoring Program / Non-Insured Health Benefits, Health Canada |
The NIHB monitors patient claims from in its internal database for evidence of possible double-doctoring, poly-pharmacy use by patients prescribed benzodiazepines, opioids, stimulants and/or gabapentin. If a beneficiary's pattern of drug utilization exceeds a safe established limit, then that patient is asked to choose a single prescriber to write all prescriptions for the drug in question. This allows the prescriber to track the extent of the patient's use of these drugs. |
|
Appendix B – Public Drug Plan Coverage by Jurisdiction
blank
| Public drug plan |
Description of coverage |
| British Columbia |
Fair PharmaCare covers all BC residents with active BC Medical Services Plan Coverage. Other subplans cover residential care facilities; recipients of income assistance; cystic fibrosis patients; children in the At-Home program; psychiatric medication and palliative care. |
| Alberta |
Alberta covers seniors 65 and older and eligible dependants; widows and dependants; palliative residents treated at home; non-group residents younger than 65; and rare diseases. Claims dispensed to residents of long-term facilities, through income support, Alberta Adult Health Benefit, Assured Income for Severely Handicapped and Alberta Child Health Benefit programs are not submitted to NPDUIS. |
| Saskatchewan |
With the exception of those eligible under another agency (primarily federal programs), all residents with a valid Saskatchewan Health Services card are eligible for coverage under the Saskatchewan Drug Plan. The universal program is the income-tested Special Support program which assists those whose benefit drug costs are high in relation to their income. Other beneficiary groups and plans include: Seniors' Drug Plan; seniors receiving the Guaranteed Income Supplement or the Saskatchewan Income Plan supplement; Children's Drug Plan; Supplementary Health and Family Health Benefits (eligibility established through Social Services); Palliative Care; Emergency Assistance; and Saskatchewan Aids to Independent Living (drug coverage related to Cystic Fibrosis, Paraplegic and Renal Programs). Claims for Formulary and Exception Drug Status drugs are submitted to NPDUIS. Drugs that are covered under special programs including the Saskatchewan Cancer Agency are not submitted to NPDUIS. |
| Manitoba |
Manitoba Pharmacare covers all provincial residents who are eligible for benefits under the Prescription Drugs Cost Assistance Act and includes residents as defined by the Health Services Insurance Act. A person must be a member of a family unit whose members have in a benefit year spent more on specified drugs than the deductible amount determined. Other subplans cover those who receive benefits from the Employment and Income Assistance Program; residents of personal care homes; and residents who are terminally ill and wish to remain at home. Products available through Part III of the Manitoba Formulary are not submitted to NPDUIS and are reported as exceptional status products in NPDUIS claims reports. |
| Ontario |
The Ontario Drug Benefit (ODB) program covers Ontarians 65 and older; residents of long-term care homes and homes for special care; recipients of professional home services; and social assistance recipients. The Trillium Drug Program provides drug benefits for Ontario residents who have high drug costs in relation to their household income. A Special Drug Program covers expensive outpatient drugs used to treat specific diseases. The New Drug Funding Program for Cancer Care covers drug benefits for new, intravenous drugs, typically administered in hospitals and cancer care facilities. The Ministry provides 75% of the funding and hospitals provide 25% for these intravenous drugs. |
| New Brunswick |
New Brunswick provides coverage to seniors who receive the Guaranteed Income Supplement (GIS) or who qualify for benefits based on an income test. Other areas covered include: cystic fibrosis patients; adult residential facilities; holders of a health card from the department of social development; special needs children; patients with prescriptions by a neurologist for Avonex, Rebif, Betaseron or Copaxane; organ or bone marrow transplant patients; individuals with growth hormone deficiency; HIV/AIDS patients; and registered nursing homes. |
| Nova Scotia |
Nova Scotia's Family Pharmacare Program and the Seniors Pharmacare Program – for residents who are age 65 or older – covers residents with a Nova Scotia health card who apply for the program and are not enrolled in other provincial drug programs. Subplans include: Drug Assistance for Cancer Patients for families with gross income no greater than $15,720; Nova Scotia Diabetes Assistance Program; and the Department of Community Services Programs for clients in receipt of income assistance and the Seniors Pharmacare Program for residents who are age 65 or older. Claims dispensed through the Department of Community Services Drug Program are not submitted to NPDUIS. Nova Scotia also offers Palliative Home Care Drug Coverage Program; however, the data related to the plan is not reported to NPDUIS. |
| Prince Edward Island |
Prince Edward Island has numerous subplans: They include: Seniors Cost Assistance for persons aged 65 or older; a high-cost drug program; a diabetes control program; a family health benefit program for families with income less than a threshold; residents in government manors or private nursing homes eligible for coverage under the Long- Term Care Subsidization Act; Sexually Transmitted Disease Program; Quit Smoking Program; and a children-in-care financial assistance program that covers children under the care of child welfare. Claims for other subplans, including the Palliative Home Care program, are not submitted to NPDUIS. |
| Newfoundland and Labrador |
The Newfoundland and Labrador Prescription Drug Program is a publicly funded program administered by the Department of Health and Community Services. There are five plans under the Program: the Foundation Plan provides coverage for persons and families in receipt of Income Support benefits; the 65Plus Plan provides coverage to residents in receipt of both Old Age Security benefits and the Guaranteed Income Supplement (GIS); the Access Plan provides drug coverage for residents that fall within established income thresholds; the Assurance Plan offers protection for individuals and families against high drug costs; and the Select Needs Plan provides coverage for disease-specific medications and supplies for residents with cystic fibrosis and growth hormone deficiency. |
| NIHB |
The Non-Insured Health Benefits for First Nations and Inuit plan covers registered Indians according to the Indian Act; Inuk recognized by one of the Inuit Land Claim organizations; Innu members of one of two Innu communities in Labrador; infant age one whose parent is an eligible recipient and is not covered by another agreement. |
Appendix C – OxyNEO Formulary Listing by Public Drug Plan
blank
| Public drug plan |
OxyNEO formulary listing decision at launch |
| British Columbia |
On February 20, 2012, BC PharmaCare announced the discontinuation of coverage for Oxycontin in the treatment of moderate to severe pain and restricted the coverage of OxyNEO to an exceptional case-by-case basis. Patients with an annual Special Authority approval for OxyContin as of March 8, 2012, continued to receive coverage of Oxycontin and OxyNEO until their Special Authority approval ended. Patients with indefinite Special Authority approval were granted transitional coverage until February 28, 2013. Physicians seeking to continue treatment with OxyNEO beyond either point were required to apply for Special Authority. Palliative care patients continue to receive coverage for Oxycontin and OxyNEO through PharmaCare Plan P. |
| Alberta |
On March 1, 2012, Alberta Blue Cross announced that OxyContin and OxyNEO would be made interchangeable on the Alberta Health and Wellness drug benefit list. |
| Saskatchewan |
On February 21, 2012, the Saskatchewan government announced that OxyContin and OxyNEO would not be made interchangeable and that coverage of OxyNEO would be available through the province's Exceptional Drug Status program. Cancer or palliative pain patients continued to receive coverage for OxyNEO, but new patients who didn't meet the criteria were made responsible for the full cost of a prescription. An exception was made for existing patients that were covered for OxyContin in the 3 months before March 1, 2012. These patients were eligible for of OxyNEO coverage. All provincial beneficiaries switching to OxyNEO required a new prescription. |
| Manitoba |
In March 2010, Manitoba restricted coverage of OxyContin to those patients with an Exceptional Drug Status approval. This requirement was extended to OxyNEO coverage once OxyContin was removed from the market. |
| Ontario |
In February, 2012, Ontario announced that OxyContin and OxyNEO would not be considered interchangeable. ODB recipients with a claim for OxyContin in the 6 months before the removal of the drug from the formulary received automatic coverage for OxyContin for an addition month. If a recipient was subsequently prescribed OxyNEO by their physician, they were eligible for coverage for one year. Beyond February 2013, all OxyNEO claims required Exceptional Access Program approval for coverage. |
| New Brunswick |
On February 9, 2012, the New Brunswick Prescription Drug Program announced that OxyNEO would not be listed on the provincial formulary. Only beneficiaries who had received coverage of OxyContin in the 3 months prior to March 1, 2012, were eligible to receive coverage of OxyNEO. Those beneficiaries changing to OxyNEO required a new prescription, if their physician deemed it appropriate. Nova Scotia Effective March 1, 2012, Nova Scotia Pharmacare announced that OxyContin and OxyNEO would not be interchangeable and would not be listed on their formulary. Only beneficiaries who had received coverage of OxyContin in the 3 months prior to March 1, 2012, were eligible to receive coverage for OxyNEO. Beneficiaries changing to OxyNEO required a new prescription, if their physician deemed it appropriate. Coverage for patients suffering from cancer or palliative pain was made possible in circumstances in which alternative medications on the formulary had failed or were not appropriate. |
| Prince Edward Island |
On February 6, 2012, Health PEI announced that OxyNEO would not be listed on its formulary. Beneficiaries with a current, active, approved Special Authorization Request for OxyContin were directed to contact their physician to have their prescription changed to OxyNEO. Those patients were made eligible to receive OxyNEO coverage if their physician deemed it appropriate and a new prescription was issued. |
| Newfoundland and Labrador |
On February 13, 2012, the Newfoundland and Labrador Prescription Drug Program (NLPDP) announced that OxyNEO would not be considered for coverage under the program. NLPDP beneficiaries currently receiving OxyContin (defined as active coverage between December 1, 2011, and March 1, 2012) would be permitted to receive coverage of OxyNEO until a comprehensive review of oxycodone had been completed by the AEAC. NLPDP beneficiaries changing to OxyNEO would require a new prescription if their physician deemed it appropriate, but would not need a new detailed Special Authorization Request, as their approved coverage for OxyContin would apply to OxyNEO. Additionally, no new requests for OxyContin would be considered by the NLPDP as of February 20, 2012. |
| NIHB |
On February 15, 2012, OxyContin was removed from NIHB's drug benefit list. The plan announced that the coverage of OxyContin would be considered in exceptional circumstances when alternatives were shown to have failed or were inappropriate. Clients who had received coverage for OxyContin in the 3 months prior were eligible for coverage of OxyNEO. |
1 OxyContin was replaced by OxyNEO in April 2012, a new form of the drug the manufacturer claims makes it difficult to abuse.
2 NOUGG. Canadian Guideline for Safe and Effective Use of Opioids for Chronic Non-Cancer Pain. Part B – Recommendations for Practice. Version 5.6. National Opioid Use Guideline Group; 2010.
3 In some drug plans, the actual percentage of seniors is somewhat less than reported. For some public drug plans, the data for specific sub-plans is not available in the NPDUIS Database. Detailed information is available in Appendix B.
4 NOUGG. Canadian Guideline for Safe and Effective Use of Opioids for Chronic Non-Cancer Pain. Part B – Recommendations for Practice. Version 5.6. National Opioid Use Guideline Group; 2010.