ISSN 2369-4904
Preamble
The Patented Medicine Prices Review Board (PMPRB) is committed to making the price review process more open and transparent to all stakeholders.
One of the primary objectives of the Compendium of Policies, Guidelines and Procedures (Compendium) is to ensure that patentees are aware of the policies, guidelines and procedures under which Board Staff reviews the prices of patented drug products sold in Canada, and the procedures normally undertaken in the scientific and price review processes and when a price appears to be excessive.
From time to time, the PMPRB finds it necessary to update the Guidelines under which it operates to ensure that they remain relevant and appropriate, as well as uphold the principles of fairness, transparency, openness, and predictability. When considering Guidelines amendments, the PMPRB consults with its stakeholders through its Notice and Comment process.
The Compendium is organized as follows:
Part A – Legal Framework
A.1 Origin of the PMPRB
A.1.1 The PMPRB was established pursuant to amendments to the Patent Act (the Act) that came into force on December 7, 1987. Prior to 1987, Canada sought to moderate the prices of patented medicines by means of compulsory licenses to increase competition. Under the 1987 amendments, Canada strengthened patent protection of medicines to provide patentees with an incentive to invest in more pharmaceutical research and development (R&D) in Canada.
A.1.2 The 1987 amendments to the Patent Act also established the PMPRB as the consumer protection pillar of the pharmaceutical patent law reform to ensure that the prices of patented medicines are not excessiveFootnote 1. The PMPRB was also tasked with a reporting role which contributes to informed decisions and policy making in health care.
A.1.3 Further amendments to the Act, which came into force on February 15, 1993, abolished the granting of compulsory licenses for patented medicines so that patentees have a statutory monopoly for the entire term of their patents. In order to fill the vacuum created by the abolition of compulsory licenses, these amendments also expanded the PMPRB’s remedial powers so that it could now order payment of excess revenues derived by patentees while selling a medicine at an excessive price, in addition to ordering price reductions, thereby further protecting the interests of consumersFootnote 2.
A.2 Mandate of the PMPRB
A.2.1 The Patent Act sets out a dual role for the PMPRB:
- Regulatory – To ensure that the prices charged by patentees for patented medicines sold in Canada are not excessive; and
- Reporting – To report on pharmaceutical trends and on the research and development (R&D) spending by patenteesFootnote 3.
A.3 Structure and Operation of the PMPRB
A.3.1 The PMPRB is an independent and autonomous quasi-judicial body. To ensure this independence and autonomy, the Act provides no power, either expressly or implicitly, to the government to direct the PMPRB or to review its decisions and orders.
A.3.2 Decisions of the PMPRB are subject to judicial review by the Federal Court on substantive or procedural grounds in accordance with administrative law principles.
A.3.3 The PMPRB is composed of Board members, appointed pursuant to subsection 91(1) of the Act, and staff (Board Staff), appointed pursuant to subsection 94(1) of the Act.
A.3.4 The PMPRB has the authority to develop policies and procedures as to how it will carry out its statutory duties in a fair and effective manner. Part of the process by which the PMPRB has determined to carry out its statutory obligations is by the administrative separation of its review and prosecutorial functions, performed by Board Staff, and its adjudicative function performed by Board members.
A.3.5 Board Staff carries out the day-to-day work of the PMPRB including the administration of the Patented Medicines Regulations (the Regulations) to ensure compliance with the prescribed filing requirements. The review of prices of patented medicines is carried out in accordance with the Guidelines, which are approved by the Board.
A.3.6 If the Chairperson of the Board decides that it is in the public interest that a hearing be held, pursuant to subsection 83(6) of the Act, to determine whether a patented medicine is being or has been sold at an excessive price in any market in Canada, the Chairperson will issue a Notice of Hearing and will appoint a panel of Board members to preside at the hearing (Hearing Panel).
A.3.7 To preserve the impartiality of Board members, until a matter is brought before a Hearing Panel at a public hearing, no Board member is informed of the results of Board Staff’s review into an instance of possible excessive pricing, other than the Chairperson in his management capacity as the Chief Executive Officer of the PMPRB, pursuant to subsection 93(2) of the Act, which is done solely for the purpose of determining whether a hearing is in the public interest.
A.4 Jurisdiction of the PMPRB Pertaining to Price Regulation
A.4.1 The Act gives the PMPRB jurisdiction to determine whether a patentee or former patentee of an invention pertaining to a medicine is selling or has sold the medicine at an excessive price in any market in Canada if the following criteria are satisfiedFootnote 4:
A.4.1.1 Patentee or Former Patentee
- Subsection 79(1) of the Act defines a “patentee” as a person for the time being entitled to the benefit of a patent for an invention, including any other person entitled to exercise rights in relation to the patent, with the exception of a person granted a compulsory license by the Commissioner of Patents before December 20, 1991, that was not terminated before the day amendments to the Act came into force on February 15, 1993.
- The PMPRB also has jurisdiction over a former patentee of an invention, while it was a patentee.
A.4.1.2 Patent pertains to the medicine
Medicine
- The term “medicine” is not defined in the Act. Please refer to the Board’s Policy with respect to the Meaning of Medicine (see section B.3).
Patent
- Subsection 79(2) of the Act provides that a patent for an invention pertains to a medicine if the invention is intended or capable of being used for medicine or for the preparation or production of medicine.
- The PMPRB considers a patent to include any Canadian patent of invention that pertains to a medicine. This includes, but is not restricted or limited to:
- Patents for active ingredients;
- Patents for processes of manufacture;
- Patents for a particular delivery system or dosage form that are integral to the delivery of the medicine;
- Patents for indications/uses; and
- Patents for formulations.
- A patent pertains to a medicine if it is capable of being used, whether or not it is being worked.
- On the face of the patent, there must be a rational connection or nexus between the invention described in the patent and the medicine, which can be one of the merest slender threadFootnote 5.
A.4.1.3 Sale in any market in Canada
- The patentee or former patentee must be selling or have sold the patented medicine in any market in Canada.
- With the exception of medicines sold under compulsory licenses granted by the Commissioner of Patents before December 20, 1991, that were not terminated before the day amendments to the Act came into force on February 15, 1993, all patented medicines sold in any market in Canada for human or veterinary use are covered by the PMPRB’s price review jurisdiction, including patented medicines sold pursuant to Notices of Compliance, under the Special Access Programme, through Clinical Trial Applications, or as Investigational New Drugs.
- The PMPRB reviews the prices of the first sale of a patented medicine at arm’s-length by the patentee, directly to a class of customer, namely a wholesaler, hospital, pharmacy or other. The PMPRB has no authority over prices charged by wholesalers or retailers or over pharmacists’ professional fees.
- Prices do not need to be approved by the PMPRB before patented medicines are sold in Canada. At the request of the patentee, Board Staff may provide pre-sale and/or pre-patent advisory assistance on whether a price would appear to be excessive.
- The PMPRB does not set the prices at which patented medicines can be sold but determines the Maximum Average Potential Price and the Non-Excessive Average Prices at which these medicines can be sold in Canada.
A.5 Price Regulation Factors
A.5.1 Subsection 85(1) of the Act stipulates those factors that the Board, during the course of a hearing, must take into consideration when determining whether a patented medicine is being sold or has been sold at an excessive price in any market in Canada by a patentee or former patentee. These factors are:
- The prices at which the medicine has been sold in the relevant market;
- The prices at which other medicines in the same therapeutic class have been sold in the relevant market;
- The prices at which the medicine and other medicines in the same therapeutic class have been sold in countries other than Canada;
- Changes in the Consumer Price Index; and
- Such other factors as may be specified in any regulations made for the purposes of this subsection.
A.5.2 If after considering the above factors, the Board is unable to determine if a price is excessive, subsection 85(2) of the Act stipulates that it may consider the costs of making and marketing the medicine, as well as other factors which can be specified by regulations or that the Board considers relevant in the circumstances.
A.5.3 The Board, following considerable deliberation and consultation with all stakeholders, pursuant to subsection 96(5) of the Act, published the PMPRB’s Guidelines pursuant to subsection 96(4) of the Act. Although the Guidelines are not binding on the Board or the patentee, they establish an approach and methodology in applying the factors set out in subsection 85(1) of the Act.
A.6 Remedies
A.6.1 Where the Board finds that a patentee is selling a patented medicine in any market in Canada at an excessive price, the Board may order the patentee to reduce the maximum price at which the patentee sells the medicine in that market.
A.6.2 In addition, where the Board finds that a patentee or former patentee, while a patentee, has sold a patented medicine in any market in Canada at an excessive price, the Board may order the patentee to offset the amount of excess revenues estimated by it to have been derived by the patentee or former patentee from the sale of the medicine at an excessive price.
A.6.3 Where the Board finds that the patentee or former patentee has engaged in a policy of selling the medicine at an excessive price, the Board may order the patentee to offset up to twice the amount of excess revenues estimated by it to have been derived by the patentee or former patentee from the sale of the medicine at an excessive price.
A.6.4 In order to offset excess revenues, the Board may order a patentee or former patentee to:
- reduce the price at which the patentee sells the medicine in any market in Canada;
- reduce the price at which the patentee sells one other patented medicine in any market in Canada; or
- make a payment to Her Majesty in right of Canada.
A.7 Filing Requirements Pertaining to Price Regulation
A.7.1 The PMPRB must have timely and accurate information to fulfill its regulatory mandate.
A.7.2 The Act and the Regulations set out the filing requirements pertaining to price regulation for a patentee or former patentee of an invention pertaining to a patented medicine that falls under the jurisdiction of the PMPRB. Further details on each element of information to be reported, and how and when the information is to be submitted to the PMPRB, can be found in the Patentee’s Guide to Reporting (available on the PMPRB’s websiteFootnote 6).
Notification of Intention to Sell a Patented Medicine
- Section 82 of the Act requires a patentee to notify the PMPRB of its intention to offer a patented medicine for sale in a market in Canada in which it has not previously been sold, and of the date on which sales are expected to begin, as soon as it is practicable to do so.
- The Board may order a patentee to provide information relating to the price at which it intends to sell the patented medicine. However, information relating to the price need not be provided earlier than 60 days before the date on which the product is intended to be sold.
Form 1 (Medicine Identification Sheet)
- Subsection 3(1) of the Regulations requires a patentee or former patentee of an invention pertaining to a medicine to report to the PMPRB prescribed information identifying the patented medicine (Form 1). Form 1 is required for all patented medicines for human or veterinary use and shall be accompanied by the product monograph for the patented medicine or, if an NOC has not been issued in respect of the patented medicine, by information similar to that contained in a product monograph.
- Subsections 3(2) and 3(3) of the Regulations require that Form 1 information must be reported if an NOC has been issued in respect of the medicine or if the medicine is being offered for sale in Canada, within seven days after the day on which the first NOC is issued in respect of the medicine, or within seven days after the day on which the medicine is first offered for sale in Canada, whichever comes first.
- If a patentee or former patentee begins selling a medicine in Canada during the pre-grant period, once the patent is issued the patentee or former patentee is required to file Form 1 information with the PMPRB.
Form 2 (Information on the Identity and Prices of the Medicine)
- Subsection 4(1) of the Regulations requires a patentee or former patentee of an invention pertaining to a medicine, which is selling or has sold the medicine in any market in Canada, to report to the PMPRB prescribed information identifying the medicine and concerning the price of the medicine (Form 2). This includes the date on which the medicine is first sold in Canada, the quantity of medicine sold in final dosage form, and either the average price per package or net revenues from the sales of each dosage form, strength and package size in which the medicine was sold by the patentee or former patentee to each class of customer in each province and territory.
- Subsection 4(4) of the Regulations provides that, in calculating the average price per package or net revenues, the actual price or actual revenue after any reduction including rebates, discounts, refunds, free goods, free services, gifts or any other benefit of a like nature and after the deduction of federal sales taxes shall be used.
- Subsection 4(2) of the Regulations requires that, if the medicine is for human use and contains a controlled substance as defined in the Controlled Drugs and Substances Act, such as opioids, amphetamines, barbiturates and benzodiazepines, or is a substance listed or described in Schedules C or D of the Food and Drugs Act, such as radiopharmaceuticals, vaccines, blood products and insulins, or is listed or described in Schedule F of the Food and Drug Regulations, such as medicines requiring a prescription, the prescribed information under Form 2 must be reported within 30 days after the day on which the medicine is first sold in Canada (for the first day’s sales), and within 30 days after each six month period commencing on January 1 and July 1 of each year, in respect of each of these periods, including the final partial period.
- Subsection 4(3) of the Regulations requires that, for medicines for human use that do not contain a controlled substance or do not contain a substance listed or described in the schedules listed in subsection 4(2), including non-prescription medicines for human use or all medicines for veterinary use, the prescribed information under Form 2 must be reported for all periods of sale, within 30 days after the date on which the PMPRB sends a request in response to a complaint, and for the two years following the request, within 30 days after each reporting period. A patentee or former patentee shall maintain up-to-date Form 2 information from the date of first sale in the event of a request for this information from the PMPRB in response to a complaint.
- A patentee or former patentee who does not voluntarily file Form 2 information for a medicine being sold during the pre-grant infringement period is required to ensure that this information is kept up-to-date for ultimate submission to the PMPRB, upon the issuance of the patent pertaining to the medicine.
A.7.3 All required information referenced in section A.7.2, must be submitted using the appropriate electronic documents Footnote 7 made available on the PMPRB’s website. The completed electronic document, in its original format and file type, must be sent to the e-mail address specified on the PMPRB’s website Footnote 8.
A.7.4 The electronic documents submitted to the PMPRB must bear the electronic signature of an authorized individual, certifying that the information set out in the document is true and complete.
A.8 Consequences of Failure to File Required Information Pertaining to Price Regulation
A.8.1 Evidence of failure to file a Notification of Intent to Sell a Patented Medicine, pursuant to subsection 82(1) of the Act, may be brought to the attention of the Chairperson who may issue an order requiring production of this information.
A.8.2 If a patentee or former patentee, as the case may be, fails to file some or all of its Form 1 or Form 2 information for one or more periods by the regulatory deadlines, it will be advised in writing by Board Staff that it is in failure to file and be given seven days from the date the letter is sent out to file the missing information. If the patentee or former patentee does not comply, Board Staff will bring a motion before the Chairperson seeking a Board Order, pursuant to section 81 of the Act, requiring the patentee or former patentee to file the information within such time as is specified in the Order.
A.8.3 If it appears to the Chairperson or to the Board that the patentee or former patentee failed to file information pursuant to sections 80, 81 or 82 of the Act or pursuant to an Order of the Board, the Board may refer the matter to the Attorney General of Canada to determine if summary conviction proceedings should be commenced under subsection 76.1(1) of the Act.
A.8.4 Pursuant to section 99 of the Act, any Order of the Board may be made an order of the Federal Court or any superior court of a province, enforceable in the same manner as an order of the court.
A.9 Protection of Confidential Information Pertaining to Price Regulation
A.9.1 Pursuant to subsection 87(1) of the Act, any information or document provided to the PMPRB under sections 80, 81 or 82 of the Act, or in any proceeding under section 83, is privileged, and cannot be disclosed without the authorization of the person who provided it, unless it has been disclosed at a public hearing under section 83.
Part B – Policies
Introduction
From time to time, the Board finds it necessary to adopt policies to indicate to stakeholders the principles it applies when interpreting its mandate. The following is a consolidation of the key policies of the Board. The following policies help to promote consistency and transparency for stakeholders.
B.1 Patent Pending Policy
B.1.1 When a medicine subject to a pending patent is being sold in any market in Canada, the PMPRB will, when the patent is issued, review the price as of the date of first sale or the date on which the patent application was laid open, whichever comes later. Once the patent is granted, the PMPRB’s jurisdiction over the price at which the medicine was sold extends to the pre-grant period, as the party selling the medicine derives the benefit of the patent during this period and so is a “patentee”, pursuant to subsection 79(1) of the ActFootnote 9.
B.2 Patent Dedication Policy
B.2.1 The PMPRB will continue to assert jurisdiction over the price at which a patented medicine is sold in any market in Canada after the patent has been dedicated until the cancellation or surrender of the patent pursuant to the express provisions of the Act or the expiry of the term of the patent. The Act, which is the mechanism by which the state grants patents, and which confers rights and benefits for the duration of the term of the patent, does not expressly recognize patent dedication as a mechanism by which patent rights may be terminated before the normal expiry of the patent term.
B.3 Policy on the Meaning of Medicine
B.3.1 A medicine is defined as any substance or mixture of substances made by any means – whether produced biologically, chemically or otherwise – that is applied or administered in vivo in humans or in animals to aid in the diagnosis, treatment, mitigation or prevention of disease, symptoms, disorders, abnormal physical states, or in modifying organic functions in humans or animals, however administered.
B.3.2 For greater certainty, this definition includes vaccines, topical preparations, anaesthetics and diagnostic products used in vivo, regardless of delivery mechanism (e.g., transdermally, capsule form, injectable, inhaler, etc.). This definition excludes medical devices, in vitro diagnostic products and disinfectants that are not used in vivoFootnote 10.
B.4 Policy on Unit of Price Review
B.4.1 The PMPRB reviews the average price of each strength of an individual, final dosage form of each patented drug product sold in Canada, including:
- Drug products that have been assigned a Drug Identification Number (DIN) by Health Canada;
- Drug products available under the Special Access Programme;
- Drug products available through a Clinical Trial Application; and
- Investigational New Drug Products.
B.4.2 Each strength of an individual, final dosage form of a patented medicine is referred to as a “patented drug product” throughout this Compendium.
B.4.3 The average price of a patented drug product will normally be expressed as the price per unit in which that patented drug product is sold (i.e., tablet, millilitre, inhaler, etc.) rounded to the fourth decimal place.
B.5 Policy for When a Price May be Considered Excessive
B.5.1 The price of a patented drug product will be presumed to be excessive in the following cases:
- If at introduction, the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the Maximum Average Potential Price;
- Thereafter, if the National Average Transaction Price exceeds the National Non-Excessive Average Price, the Market- Specific Average Transaction Prices would be reviewed and would be considered excessive if they exceed their respective Market-Specific Non-Excessive Average Prices.
B.5.2 If the National Average Transaction Price exceeds the Maximum Average Potential Price or National Non-Excessive Average Price, but does not trigger the criteria for commencing an investigation (see Schedule 11), the patentee will be notified and the patented drug product will be reported on the PMPRB Web site as “Does Not Trigger Investigation.” The patentee will be expected to decrease its price and offset any excess revenues (see the PMPRB’s Policy on the Offset of Excess Revenues in section B.7).
B.5.3 If the National Average Transaction Price is found to exceed the Maximum Average Potential Price or the National Non-Excessive Average Price by an amount which triggers the investigation criteria, the patentee will be notified of the commencement of an investigation and the patented drug product will be reported as “Under Investigation” (see section C.13).
B.6 Policy on the Use of Patented and Non-Patented Drug Products in the Price Tests
B.6.1 Board Staff may exclude from the price tests any drug product identified for comparison purposes, both patented and non-patented, if it has reason to believe it is being sold at an excessive price.
B.6.2 Pivotal drug products used for comparison purposes will be assessed against the price tests described in the Guidelines.
B.6.3i The price of a relevant non-patented drug product will be included in the price tests unless Board Staff is of the view that the price of the medicine is excessive as a result of the absence of competition or other market conditions.
B.7 Policy on the Offset of Excess Revenues
B.7.1 The Board may allow a patentee to offset any excess revenues estimated by it to have been derived from the sale of the medicine at an excessive price through either: (i) the reduction of the price of the medicine or the price at which the patentee sells another patented medicine in Canada; or (ii) a payment to Her Majesty in right of Canada.
B.7.2 To offset excess revenues via a price reduction, the average price of a patented drug product will only be considered to have been reduced if it is below the previous year’s Non-Excessive Average Price; not taking an allowable price increase will not be considered for purposes of offsetting excess revenues.
B.7.3 Cumulative excess revenues cannot fall below zero.
B.8 Policy on Generic Medicinesii
B.8.1 In this policy statement, a “Patented Generic Drug” is a drug that falls within a patent for an “invention pertaining to a medicine” as defined in section 79(2) of the Patent Act, and is sold by a “patentee” as defined in section 79(1) of the Patent Act and has been approved for sale in Canada by Health Canada on the basis of: a comparison of that drug to a Canadian reference product as defined in C.08.001.1, or otherwise approved pursuant to C.08.002.1 of the Food and Drug Regulations; has been declared interchangeable to a Canadian reference product by a provincial or territorial drug reimbursement formulary; or is a licensed version of an existing Brand reference product sold in Canada (e.g. a drug product approved by Health Canada pursuant to a cross-reference to an original drug submission, such as a X-REF NDS).
B.8.2 The PMPRB has jurisdiction over the prices of all patented medicines, including Patented Generic Drugs. The PMPRB recognizes that Patented Generic Drugs represent a potentially lower risk category of patented medicines, and therefore a lower priority for PMPRB regulatory investigation.
B.8.3 Board Staff will only review information relating to the identity of the medicine and/or pricing upon the commencement of an investigation by Board Staff.
B.8.4 Board Staff will commence an investigation into the price of a Patented Generic Drug if all of the following three conditions are met:
- A complaint has been received in respect of the Patented Generic Drug;
- The patentee of the Patented Generic Drug is the only company in Canada which is selling a generic version of the drug in Canada; and
- The Patented Generic Drug is not the subject of a pricing agreement with the pan-Canadian Pharmaceutical Alliance (pCPA) to which it is compliant. The onus of proving to Board Staff that a Patented Generic Drug is subject to, and compliant with, a pricing agreement with pCPA will rest with the patentee for that Patented Generic Drug.
B.8.5 Any Patented Generic Drug that is the subject of an investigation will be reviewed by Board Staff in accordance with the applicable Guidelines and Policies. With respect to domestic and international price comparisons for Patented Generic Drugs, Board Staff will consider, but will not be limited to, using the domestic and international prices of the brand reference product as comparators. If the reference brand product is no longer being sold in the Canadian or the international market, Board Staff will consider, but will not be limited to, using the former brand reference product price as a comparator (as adjusted by CPI).
Part C – Guidelines and Procedures
Preface
The following Guidelines and procedures represent direction from the Board, to patentees and Board Staff, in order to provide assistance on how to comply with the Patent Act and the Patented Medicines Regulations. Please note: These Guidelines are not binding on patentees nor the Board in the context of a hearing.
The Guidelines are organized as follows:
The Scientific Review Process: An evidence-based process that assesses the level of therapeutic improvement of a patented drug product and recommends, where appropriate, the drug products to be used for comparison purposes and the comparable dosage regimens.
The Price Review Process: The level of therapeutic improvement of a patented drug product is used to determine the Maximum Average Potential Price at introduction. Following introduction, the price of an existing patented drug product is reviewed according to the relevant price tests to establish the National and Market-Specific Non-Excessive Average Prices.
Investigations: The approach used and procedures undertaken when a price appears to exceed the investigation criteria (see Schedule 11).
Schedules: All Schedules form part of the Guidelines.
The Scientific Review Process
C.1 Introduction
C.1.1 The PMPRB’s scientific review is an evidence-based process that recommends the level of therapeutic improvement of a patented drug product.
C.1.2 The scientific review process for all new patented drug products (including those with an NOC or available through the Special Access Programme, Clinical Trial Applications and as Investigational New Drugs) will be undertaken using the following Guidelines and procedures.
C.2 Sources of Scientific Information
C.2.1 The scientific review of a new patented drug product is based on information from a variety of sources:
- Patentee Submission – Patentees may provide Board Staff with a brief submission (see Schedule 1), which clearly explains the rationale for the patentee’s proposals relative to the level of therapeutic improvement, drug products identified for comparison purposes and comparable dosage regimens.
- Research by a Drug Information Centre (DIC) – Board Staff uses the services of various drug information centres to obtain scientific information, such as clinical trial information, clinical practice guidelines, etc. The basis of the review by the DIC is the product monograph or information similar to that contained in a product monograph if an NOC has not been granted.
- Research by Board Staff – Board Staff may also update research and supplement data and evidence from the patentee and DIC using other sources.
- Research by the Human Drug Advisory Panel (HDAP) Members – Members of HDAP may also undertake their own research and supplement the evidence obtained from the patentee, the DIC and Board Staff for a review.
C.3 Human Drug Advisory Panel
C.3.1 HDAP provides expertise and advice to Board Staff in conducting the scientific review. HDAP performs the following functions:
- Reviews and evaluates scientific information provided as described in section C.2;
- Considers advice from other experts (when deemed necessary);
- Recommends the level of therapeutic improvement of the new patented drug product, and identifies drug products for comparison purposes and dosage regimens where possible; and
- Identifies significant uncertainties in the evidence which may affect the analysis on which its recommendations are based.
C.3.2 In general, new patented drug products are referred to HDAP. However, the following new patented drug products will not be referred to HDAP unless the patentee files a submission claiming therapeutic improvement:
- The new patented drug product represents a new DIN of an existing dosage form of an existing drug product, or a new DIN of another dosage form of the existing drug product that is comparable to the existing dosage form as per Schedule 2 and has the same indication or use as the existing DIN; or
- The new patented drug product is a combination drug product, the individual components of which are sold in Canada and have the same indication or use; or
- The new patented generic drug product is considered by Health Canada to be bioequivalent to the reference brand drug product sold in Canada; or
- The new patented generic drug product is a licensed version of an existing brand drug product sold in Canada.
Procedures
C.3.3 HDAP is composed of members with recognized expertise in drug therapy who have experience in clinical research methodology, statistical analysis and the evaluation of new drug products.
C.3.4 HDAP and its individual members do not meet with patentees.
C.3.5 The names of the members of HDAP are posted on the PMPRB’s website.
C.3.6 The dates of HDAP meetings are posted on the PMPRB’s website.
C.3.7 At the request of a patentee, a new patented drug product will also be referred to HDAP to provide pre-sale and/or pre-patent advisory assistance.
C.3.8 For a new patented drug product referred to HDAP, a patentee must file a submission which contains the elements referred in Schedule 1 at least two months prior to an HDAP meeting.
C.3.9 In the event that a large number of submissions are received for any one HDAP meeting, priority will be determined as follows:
- Drug products that are patented and sold;
- Drug products that are patented and about to be sold;
- Drug products that are patented but not sold;
- Drug products that are not patented but sold;
- Drug products that are not patented and are not sold.
C.3.10 The patentee will be advised of the date of the HDAP meeting at which its submission will be considered.
C.3.11 The HDAP report will include recommendations on the level of therapeutic improvement, the drug products to be used for comparison purposes and comparable dosage regimens, as well as an explanation of how the primary and secondary factors (see section C.6) were applied and a description of the evidence (see section C.7) relied upon.
C.3.12 A copy of the HDAP report will be sent to the patentee.
C.4 Determining the Primary Indication/ Use of a New Patented Drug Product
C.4.1 Determining the primary approved indication (or proposed indication if an NOC is pending), or primary use if not approved for market in Canada, is important for the assessment of the level of therapeutic improvement of a new patented drug product with multiple approved indications/multiple uses.
Procedures
C.4.2 The level of therapeutic improvement for new patented drug products with multiple approved indications or multiple uses will be based on the approved indication or use for which the drug product offers the greatest therapeutic advantage in relation to alternative therapies for the same indication/use in a significant patient population. This would exclude rare medical conditions or diseases (i.e., low incidence and prevalence in Canada).
C.4.3 This approved indication or use will be considered the “primary indication” for the purpose of selecting drug products to be used for comparison purposes.
C.4.4 Where there is no apparent single approved indication or use for which the new patented drug product offers the greatest therapeutic advantage, the approved indication or use representing, potentially, the greatest proportion of sales will be the basis for recommending its level of therapeutic improvement and selection of drug products to be used for comparison purposes.
C.4.5 Estimates of potential sales can be based on several sources including actual prescribing patterns (when available), epidemiological data (Canadian incidence and prevalence) and prescribing patterns in other countries.
C.5 The Level of Therapeutic Improvement
C.5.1 HDAP utilizes the following set of definitions to recommend the level of therapeutic improvement of a drug product:
Breakthrough: A breakthrough drug product is the first one to be sold in Canada that treats effectively a particular illness or addresses effectively a particular indication.
Substantial Improvement: A drug product offering substantial improvement is one that, relative to other drug products sold in Canada, provides substantial improvement in therapeutic effects.
Moderate Improvement: A drug product offering moderate improvement is one that, relative to other drug products sold in Canada, provides moderate improvement in therapeutic effects.
Slight or No Improvement: A drug product offering slight or no improvement is one that, relative to other drug products sold in Canada, provides slight or no improvement in therapeutic effects.
C.6 Factors Considered in Recommending the Level of Therapeutic Improvement
C.6.1 The following factors are to be used in recommending the level of therapeutic improvement of a drug product:
Primary Factors
- Increased efficacy
- Reduction in incidence or grade of important adverse reactions
Secondary Factors
- Route of administration
- Patient convenience
- Compliance improvements leading to improved therapeutic efficacy
- Caregiver convenience
- Time required to achieve the optimal therapeutic effect
- Duration of usual treatment course
- Success rate
- Percentage of affected population treated effectively
- Disability avoidance/savings
C.6.2 The primary factors will be given the greatest weight, followed by an assessment of any additional improvement as a result of the secondary factors.
C.6.3 In recommending the level of therapeutic improvement of new patented drug products, factors such as the following will generally not be taken into consideration, unless the impact of these factors results in either increased efficacy and/or a reduction in the incidence or grade of important adverse reactions:
- The mechanism of action
- A new chemical entity
- A different pharmacokinetic profile
Procedures
C.6.4 Primary factors will be considered in order to assess if the new patented drug product is a breakthrough, or represents substantial, moderate or slight/no improvement relative to other drug products available in Canada.
C.6.5 Secondary factors will then be considered. These factors will be weighed by HDAP based on sound evidence and reasonable clinical judgement. These secondary factors could result in the level of therapeutic improvement being assessed at up to the level of moderate therapeutic improvement.
C.7 Methodology for the Evaluation of the Level of Therapeutic Improvement
C.7.1 An evidence-based approach will be used to assess the new patented drug product under review using the hierarchy of evidence from the Oxford Centre for Evidence-Based Medicine (see Schedule 1).
Procedures
C.7.2 HDAP will critically appraise the evidence with regards to validity, impact and applicability. Level 1 evidence will be given greater weight compared to other levels of evidence in recommending the level of therapeutic improvement and the selection of drug products to be used for comparison purposes.
C.7.3 Since uncertainty in the relative efficacy of a new patented drug product is common, level 1 evidence is preferred for new patented drug products to be assessed as having a breakthrough or substantial level of therapeutic improvement relative to other drug products sold in Canada.
C.7.4 HDAP may consider other levels of evidence, as required, on a case by case basis in order to assess the secondary factors.
C.8 Selection of Drug Products to be Used for Comparison Purposes and Comparable Dosage Regimens
Drug Products to be Used for Comparison Purposes
C.8.1 HDAP uses the World Health Organization (WHO) Collaborating Centre for Drug Statistics Methodology’s Anatomical Therapeutic Chemical (ATC) Classification System in the selection of drug products to be used for comparison purposes.
C.8.2 The chemical substances to be used for comparison purposes will typically be those identified under the ATC classification system at the sub-class level above the single chemical substance. This will normally be the fourth sub-class level. HDAP may also choose from the next higher sub-class or another sub-class. In some instances, it may be appropriate to select from the fifth or single chemical substance level.
C.8.3 HDAP may omit from the comparison a chemical substance of the same ATC therapeutic class as the new patented drug product under review if, in HDAP’s opinion, it is unsuitable for comparison. For example, drug products with a primary indication/use other than the primary indication/use of the new patented drug product under review may be omitted from the comparison.
Procedures
C.8.4 HDAP will identify all drug products to be used for comparison purposes, which have the same approved indication or use as the new patented drug product under review.
Breakthrough
C.8.5 There will be no drug products recommended by HDAP for comparison purposes for a new patented drug product that represents a breakthrough, given that such a drug product is, by definition the first one to be sold in Canada that treats effectively a particular illness or addresses effectively a particular indication.
Substantial Improvement
C.8.6 For new patented drug products that represent a substantial therapeutic improvement, HDAP will identify drug products with the same approved indication or use over which the new patented drug product represents a substantial therapeutic improvement.
Moderate Improvement
C.8.7 For new patented drug products that represent a moderate therapeutic improvement, HDAP will identify drug products with the same approved indication or use over which the new patented drug product represents a moderate therapeutic improvement.
Slight or No Improvement
C.8.8 Any drug product that is not considered a breakthrough and that is not considered to offer substantial or moderate improvement will fall into the category of drug products offering slight or no improvement.
C.8.9 For new patented drug products that represent slight or no therapeutic improvement, HDAP will first attempt to identify comparable drug products, based on the primary and secondary factors set out in section C.6.1, with the same approved indication or use as the new patented drug product under review.
C.8.10 If no comparable drug products are found, HDAP will identify all drug products that are considered superior in treating the approved indication or use, based on primary and secondary factors.
C.8.11 For a new patented drug product that is a new presentation of the same chemical entity as one or more existing drug products, its comparators will be those existing drug products that are available in the same or comparable dosage form and have the same indication or use, unless the patentee makes a submission claiming therapeutic improvement and HDAP identifies the new patented drug product as providing therapeutic improvement. This will apply regardless of whether or not the dosage regimens of the new and existing drug products are the same or differ materially.
C.8.12 The comparable drug products for a new patented combination drug product, where each of the elements of the combination drug product are sold in Canada and have the same indication or use, will be limited to the component parts, unless the patentee makes a submission regarding therapeutic improvement and HDAP identifies the new patented drug product as providing therapeutic improvement.
C.8.13 The comparable drug products for a new patented generic drug product that is bioequivalent to a brand drug product sold in Canada, or that is a licensed version of the same brand drug product sold in Canada, will be limited to that brand drug product.
Comparable Dosage Regimens
C.8.14 The comparable dosage regimen recommended for comparison purposes will normally not be higher than the maximum of the usual recommended dosage in the Product Monograph (or similar information) taking into account relevant clinical variables. The most appropriate strength of the drug product will be chosen for a particular dosage regimen.
C.8.15 Generally, a dosage regimen based on a course of treatment will be applicable to acute indications, while a per-day regimen (based on maintenance dose) will be applicable to chronic situations.
C.9 Provisions for Over-the-Counter (OTC) and Veterinary Drug Products
C.9.1 As per the regulatory and reporting provisions outlined in Part A – Legal Framework, the scientific review for patented OTC and veterinary drug products will only be undertaken following the PMPRB’s receipt of a complaint regarding the price of the patented drug product.
Procedures
C.9.2 Upon receipt of a complaint, the PMPRB will undertake the scientific review of the patented OTC or veterinary drug product in the same manner as is undertaken for all other patented drug products.
C.9.3 If a complaint is received for a patented OTC drug product, the required scientific information will be sent to HDAP to recommend the level of therapeutic improvement, the drug products to be used for comparison purposes and comparable dosage regimens.
C.9.4 If a complaint is received for a patented veterinary drug product, a Veterinary Drug Advisory Panel (VDAP) will be formed to recommend the level of therapeutic improvement, the drug products to be used for comparison purposes and comparable dosage regimens.
The Price Review Process
C.10 Introduction
C.10.1 The Price Review Process is conducted for the purposes of:
- Establishing the Maximum Average Potential Price at introduction for the new patented drug product; and
- Assessing whether or not the price of an existing patented drug product appears to be excessive.
C.11 Review of Prices of New Patented Drug Products at Introduction
Introduction
C.11.1 The test applicable to the introductory price of a new patented drug product is dependent on the level of therapeutic improvement recommended for the new patented drug product during the scientific review process. A detailed description of how the price tests will be applied to the levels of therapeutic improvement can be found in Schedule 8.
C.11.2 For purposes of the following sections Market-Specific Average Transaction Prices refer to those of the pharmacy, hospital and wholesaler customer classes, and for each province and territory.
Breakthrough
C.11.3 The introductory price(s) of a breakthrough new drug product will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the Maximum Average Potential Price at introduction as determined by the Median International Price Comparison test (see Schedule 5).
Substantial Improvement
C.11.4 The introductory price(s) of a new drug product providing substantial improvement will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the Maximum Average Potential Price at introduction as determined by the higher of:
- The highest non-excessive price of the drug products identified pursuant to section C.8.6, based on a Therapeutic Class Comparison (TCC) test (see Schedule 3), and
- The median international price as determined by the Median International Price Comparison test (see Schedule 5).
Moderate Improvement
C.11.5 The introductory price(s) of a new drug product providing moderate improvement will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the Maximum Average Potential Price at introduction as determined by the higher of:
- The mid-point between the price obtained in paragraph (b) below and the median international price determined by the Median International Price Comparison test (see Schedule 5), and
- The highest non-excessive price of the drug products identified pursuant to section C.8.7 based on a TCC test (see Schedule 3).
C.11.6 If it is not possible to conduct a TCC test, the introductory price(s) of a new drug product providing moderate improvement will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the median international price determined by the Median International Price Comparison test (see Schedule 5). This could occur where HDAP is unable to derive comparable dosage regimens for all of the drug products identified pursuant to section C.8.7 or where the prices of these drug products appear to be excessive.
Slight or No Improvement
C.11.7 The introductory price(s) of a new drug product providing slight or no improvement will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the Maximum Average Potential Price as determined by the highest non-excessive price of the comparable drug products identified pursuant to section C.8.9, based on a TCC test (see Schedule 3).
C.11.8 It is possible that HDAP may determine that a new patented drug providing slight or no improvement has no comparable drug products. In such exceptional cases, the introductory price(s) of the new drug product will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the lower of:
- The lowest non-excessive price of the superior drug products identified pursuant to section C.8.10 based on a TCC test (see Schedule 3), and
- The median international price determined by the Median International Price Comparison test (see Schedule 5).
C.11.9 If it is not possible to conduct a TCC test, the introductory price(s) of a new drug product providing slight or no improvement will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the median international price determined by the Median International Price Comparison test (see Schedule 5). This could occur where the HDAP is unable to derive comparable dosage regimens for all of the drug products identified pursuant to section C.8.10 or where the prices of these drug products appear to be excessive.
C.11.10 Unless the patentee makes a submission claiming therapeutic improvement and HDAP identifies the new patented drug product as providing moderate or substantial improvement:
- The introductory price(s) of a new patented generic drug product that is bioequivalent to a brand drug product sold in Canada, or that is a licensed version of the same brand drug product sold in Canada, will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the price of the patented brand drug product.
- Subject to paragraph below, the introductory price(s) of a new patented drug product that is a new presentation of the same chemical entity, with the same or comparable dosage form (as per Schedule 2), the same comparable dosage regimen and the same indication or use, will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the result of the Reasonable Relationship (“RR”) test (see Schedule 4).
- Where the comparable dosage regimen differs materially, the introductory price(s) of the new patented drug product will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the highest non-excessive price of the drug products identified pursuant to section C.8.11 based on a TCC test (see Schedule 3).
- When a new patented drug product is a combination product as defined in section C.8.13, its introductory price(s) will be presumed to be excessive if the National Average Transaction Price or any Market-Specific Average Transaction Price exceeds the sum of the prices of the individual components.
Highest International Price Comparison (“IPC”) Test
C.11.11 Notwithstanding sections C.11.3 to C.11.10, the Maximum Average Potential Price for a new patented drug product at the national level, for the pharmacy and hospital customer classes, and for each province and territory shall not exceed the Highest International Price Comparison test (see Schedule 6).
C.11.12 The PMPRB may review the price of any new patented drug product in any market in Canada (e.g., by class of customer in a province/territory).
Procedures
C.11.13 Board Staff will use public sources for the prices of the drug products used for comparison purposes. The sources of prices which Board Staff will consult are (in alphabetical order): the Association québécoise des pharmaciens propriétaires (AQPP); IMS Health; McKesson Canada; Ontario Drug Benefit (ODB) Programs; PPS Pharma; and the Régie de l’assurance maladie du Québec (RAMQ). Board Staff will select the lowest public price from these sources for each of the drug products identified for comparison purposes. If no price is available for a comparator from these sources, Board Staff will explore additional sources until a public price is found.
C.11.14iii Notwithstanding other elements of section C.11, the prices from sources identified in subsection C.11.13 for a new patented drug product shall not exceed the Maximum Average Potential Price (MAPP) established by appropriate introductory price test(s). If a price from these sources is found to exceed the MAPP, it would trigger an investigation by Board Staff. The first step of this investigation would be to confirm with patentees whether any sales have taken place at this price. If it can be positively ascertained by Board Staff that such sales have taken place, an investigation will proceed as described elsewhere in the Guidelines.
C.11.15 Based on the application of the appropriate introductory price test(s) to the first day of sale price and sales data, Board Staff will provide interim advice to the patentee as to whether or not the price would appear to be excessive.
C.11.16 The introductory price(s) of a new patented drug product will be determined by calculating the National Average Transaction Price and the Market-Specific Average Transaction Prices for each of three classes of customers (hospital, pharmacy and wholesaler) and for each province/territory.
C.11.17 The National Average Transaction Price will be calculated based on total net revenues across all markets divided by total number of units across all markets.
C.11.18 Market-Specific Average Transaction Prices will be calculated based on total net revenues for the market divided by total number of units for the market. (e.g., The Market-Specific Average Transaction Price in the “pharmacy” class of customer will be based on total net revenues for pharmacies across all provinces and territories divided by total number of units for pharmacies across all provinces and territories. The Market-Specific Average Transaction Price in any specific province or territory will be based on total net revenues in a province or territory across all classes of customer divided by total number of units for that province or territory across all classes of customer.)
C.11.19 The introductory period is the period from the date of first sale to the end of the six-month regulatory reporting period (June 30 or December 31), as long as the period covered is greater than one month. If the period is less than one month, the following six-month reporting period will be used.
C.11.20 All pivotal drug products used for comparison purposes will be assessed against the price tests described in the Guidelines. Board Staff may exclude from the price tests any drug product, both patented and non-patented, if it has reason to believe it is being sold at an excessive price.
C.11.21iv The price of a relevant non-patented drug product will be included in the price tests unless Board Staff is of the view that the price of the medicine is excessive as a result of the absence of competition or other market conditions.
C.12 Review of Prices of Existing Patented Drug Products
C.12.1 The price of an existing patented drug product will be presumed to be excessive if the National Average Transaction Price exceeds the National Non-Excessive Average Price as determined by the lower of:
- The change in the CPI as per the CPI-Adjustment Methodology (see Schedule 9); or
- The result of the Highest International Price Comparison test (see Schedule 6).
C.12.2 If the National Average Transaction Price exceeds the National Non-Excessive Average Price by an amount which triggers the investigation criteria (see Schedule 11), Board Staff shall review the Market-Specific Average Transaction prices. Board Staff shall also review the prices in these markets, if a complaint is the trigger for the commencement of an investigation.
- The price in each of three classes of customer (hospital, wholesaler, pharmacy) and in each province/territory will be presumed to be excessive if the Market-Specific Average Transaction Price exceeds the Market-Specific Non-Excessive Average Price as determined by the change in the CPI as per the CPI-Adjustment Methodology (see Schedule 9).
- In addition, the price in each of two classes of customer (hospital and pharmacy) and in each province/territory will be presumed to be excessive if the Market-Specific Average Transaction Price exceeds the Market-Specific Non-Excessive Average Price as determined by the Highest International Price Comparison test (see Schedule 6).
C.12.3v Rescinded.
C.12.4 In addition, when a patentee can demonstrate that an increase in the National Average Transaction Price is due solely to a sales-mix shift and none of the Market-Specific Average Transaction Prices for each class of customer and in each province/territory exceed their respective Market-Specific Non-Excessive Average Prices as determined by the CPI-Adjustment Methodology, the National Average Transaction Price will not be presumed to be excessive.
C.12.5 When the National Average Transaction Price or a Market-Specific Average Transaction Price of a drug product increases from a previous year due to the reduction or end of a benefit(s) and the patentee provides evidence to demonstrate that the price increase was due solely to the reduction or termination of the benefit(s), it may be appropriate to adjust the Non- Excessive Average Prices (national and market-specific) through the DIP Methodology, as described in Schedule 10.
C.12.6 The Board recognizes that there may be cost of making and marketing arguments, whereby it may be appropriate to adjust the Non-Excessive Average Price(s) of a patented drug product (e.g., once a Notice of Compliance has been obtained and the drug product was first sold on a compassionate basis as an Investigational New Drug, through a Clinical Trial Application or under the Special Access Programme).
C.12.7 The PMPRB may review the price of any existing patented drug product in any market in Canada (e.g., by class of customer in a province/territory).
C.12.8vi/vii Where an existing drug product is sold in Canada by persons other than the initial patentee as a result of a merger or acquisition agreement, the PMPRB’s Guidelines will apply to the DINs sold by these persons as if they were the DINs of the initial patentee (also known as the DIN continuation policy). For example, if as part of a merger or acquisition, a patentee ceases to sell a patented drug product and the marketing rights to the product are transferred to another patentee, the DIN sold by the new patentee will be considered as a continuation of the original DIN for purposes of the application of the Guidelines—including the DIP and CPI-Adjustment Methodology.
Board Staff will interpret “acquisition” as including individual DIN transfers outside of the specific case of mergers between companies, or acquisitions of entire companies, as long as this interpretation is in compliance with Section 87 of the Patent Act and Section A.9.1 of the Guidelines.
Investigations
C.13 Introduction
C.13.1 When the price of a patented drug product appears to exceed the Guidelines but not by an amount that triggers the investigation criteria (Schedule 11), the patentee will be notified and the patented drug product will be reported on the PMPRB’s Web site as “Does Not Trigger Investigation.” The patentee will be expected to reduce its National Average Transaction Price and Market-Specific Average Transaction Prices and to offset any excess revenues that may have accrued (see Schedule 13), but no immediate action will be taken by Board Staff.
C.13.2 When the National Average Transaction Price of a patented drug product appears to exceed the National Non-Excessive Average Price and the circumstances are within the criteria established by the Board (Schedule 11), the patentee will be notified of the commencement of an investigation and the patented drug product will be reported on the PMPRB’s Web site as “Under Investigation.”
C.13.3 The examination will include an analysis of the pricing history of the patented drug product from introduction for both the National Average Transaction Price and Market-Specific Average Transaction Prices (i.e., for each class of customer (hospital, pharmacy, wholesaler) and each province/territory).
C.13.4 The International Therapeutic Class Comparison (ITCC) test compares the price of the patented drug product with the publicly available ex-factory prices in the comparator countries listed in the Regulations of comparable drug products identified in the domestic price test (i.e., the RR or TCC test). The ITCC test will only be conducted on a case-by-case basis if it appears it might provide information in the context of an investigation into apparent excessive prices. It will not be used as a primary price test. This test is described in Schedule 7.
C.13.5 The period of time available to the patentee to respond to Board Staff following a notification that an investigation has been commenced is ordinarily brief. For example, if the patentee should have known that a price would appear excessive based on its own filings (e.g., where the price increased by more than would be permitted under the CPI-Adjustment Methodology), the period of time may be as short as seven calendar days. A longer period of time, 30 calendar days, may be available if it is reasonable to believe that the patentee might have been unaware that the National Average Transaction Price or Market-Specific Average Transaction Prices may appear to be excessive (e.g., if HDAP has recommended the use of different drug products for comparison purposes or dosage regimens from those which were proposed by, and may have been reasonably anticipated by, the patentee).
C.13.6 There are three possible outcomes to an investigation:
- The National Average Transaction Price and/or Market-Specific Average Transaction Prices do not appear to be excessive; or
- The National Average Transaction Price and/or Market-Specific Average Transaction Prices appear to be excessive and the patentee submits an acceptable Voluntary Compliance Undertaking (VCU); or
- The National Average Transaction Price and/or Market-Specific Average Transaction Prices appear to be excessive and the patentee does not submit an acceptable VCU in which case Board Staff will refer the matter to the Chairperson and recommend the issuance of a Notice of Hearing.
C.14 Where the Price Appears Non-Excessive
C.14.1 If the investigation concludes that the National Average Transaction Price and/or Market-Specific Average Transaction Prices of the patented drug product do not appear to be excessive, the investigation will be terminated and the patentee will be advised accordingly.
C.15 Voluntary Compliance Undertaking
C.15.1 If the investigation confirms that the National Average Transaction Price and/or Market-Specific Average Transaction Prices appear to be excessive, the patentee will be given an opportunity to submit a written proposal in the form of a VCU to reduce its price and offset any excess revenue accrued as a result of sales at a price presumed to be excessive (see Schedule 13).
C.15.2 The proposal of a VCU does not constitute an admission by the patentee that the National Average Transaction Price and/or Market-Specific Average Transaction Prices of the drug product are or were excessive.
C.15.3 Board Staff will assist a patentee with the preparation of a VCU, and may provide sample documents or other advice as may be appropriate to the situation.
C.15.4 If a patentee submits a VCU consistent with the Guidelines, it is the policy of the Board that only the Chairperson (or, if the VCU is submitted after the issuance of a Notice of Hearing, the Board Hearing Panel) may approve the VCU.
C.15.5 The Chairperson is not authorized to enter into negotiations on the terms of a VCU with a patentee.
C.15.6 The proposed VCU should include a statement as to the Maximum Average Potential Price at introduction and subsequent National and Market-Specific Non-Excessive Average Prices with which the patentee agrees to comply and the means by which the patentee proposes to offset any excess revenues.
C.15.7 In most cases, the VCU should specify a payment to Her Majesty in right of Canada as the means to offset excess revenues.
C.15.8 In deciding whether to accept a VCU, the Chairperson (or Board Hearing Panel) will be guided by section 83 of the Act.
C.15.9 The PMPRB will report publicly on all VCUs accepted by the Chairperson or a Board Hearing Panel. The information reported will ordinarily include the name of the patented drug product and/or the patentee and such other information as is considered appropriate. This information will be included in the PMPRB’s Annual Report and be published on the PMPRB Web site. It may also be published in the NEWSletter or other publications.
Schedule 1 – Submissions by Patentees on Level of Therapeutic Improvement
Each submission should clearly explain the rationale behind the patentee’s proposals for level of therapeutic improvement, drug products for comparison purposes and comparable dosage regimens.
The patentee should provide its submission and all supporting references according to the PMPRB filing requirementsFootnote 11, viii/ix. Board Staff will verify that the supporting references mentioned or listed in the submission have been included and advise the patentee if any information is missing.
1. Supporting Clinical Evidence
1.1 Drug Product: Drug Product: name, class, brief description of mechanism, approved indication(s) or use(s), and approved or proposed dosing.
1.2 Product Monograph (or similar information if no NOC): Submitted with Form 1, Identity of the Medicine.
1.3 Individual Trials/Studies:
- Level 1 Evidence: Published randomized clinical trials (RCTs) of the new patented drug product under review versus active comparators, if any; Published RCTs of the new patented drug product under review versus placebo; High quality unpublished RCTs, if available.
- Published clinical trials with lower levels of evidence (e.g., outcome studies, systematic reviews of cohort and case-controlled trials) if Level 1 evidence is unavailable. Note: In relation to both Level 1 and other levels of evidence, the patentee is encouraged to focus the submission on key trials that lead to an NOC or to a change in clinical practice, or would be of the highest quality/best evidence the patentee has available.
- Editorials and errata of published clinical trials.
- Other clinical evidence, such as ecological studies, case series and community surveys of the new patented drug product under review if the patentee is proposing therapeutic improvements due to secondary factors.
1.4 Summary of trials included in submission in tabular format:
- Study reference(s) (abstracts and publications if published), and study identification assigned by the patentee.
- Brief description of the study and outcomes measures.
- Trial Phase (i.e., Phase II, III or IV); Phase I trials will not be reviewed.
1.5 Brief overview of standards of therapy or accepted clinical practice for which the new patented drug product under review is indicated or used:
- For example, class reviews, systematic reviews/meta-analyses.
1.6 Published Clinical Practice Guidelines regarding the indication or use of the new patented drug product under review if available:
- Peer reviewed Canadian guidelines are preferred; American, UK, Australian and European guidelines will be considered.
2. Proposal of the Patentee
2.1 Executive Summary:
- Brief description of the new patented drug product and its place in therapy, as well as a summary of the clinical evidence.
2.2 Proposed level of therapeutic improvement.
2.3 Proposed Comparators:
- Evidence of the same approved indication or use as the new patented drug product under review.
2.4 Proposed comparable dosage regimens for the comparator and the new patented drug product under review:
- Approved or proposed doses.
- Doses used in clinical trials.
- Doses recommended in clinical practice guidelines.
3. Hierarchy of Evidence for Recommending Level of Therapeutic Improvement
3.1 Table 1, below, outlines the hierarchy of evidence that will be considered by HDAP in recommending the level of therapeutic improvement of a new patented drug product.
Table 1Footnote 12 – Hierarchy of evidence for recommending level of therapeutic improvement
| Level |
Therapy/Prevention |
Economic and decision analyses |
| 1a |
SR (with homogeneity*) of RCTs |
SR (with homogeneity*) of Level 1 economic studies |
| 1b |
Individual RCT (with narrow Confidence Interval) |
Analysis based on clinically sensible costs or alternatives; systematic review(s) of the evidence; and including multi-way sensitivity analyses |
| 1c |
All or none§ |
Absolute better-value or worse-value analyses † |
| 2a |
SR (with homogeneity*) of cohort studies |
SR (with homogeneity*) of Level >2 economic studies |
| 2b |
Individual cohort study (including low quality RCT; e.g., <80% follow-up) |
Analysis based on clinically sensible costs or alternatives; systematic review(s) of the evidence, or single studies; and including multi-way sensitivity analyses |
| 2c |
“Outcomes” Research; Ecological studies |
Audit or outcomes research |
| 3a |
SR (with homogeneity*) of case-control studies |
SR (with homogeneity*) of 3b and better studies |
| 3b |
Individual Case-Control Study |
Analysis based on limited alternatives or costs, poor quality estimates of data, but including sensitivity analyses incorporating clinically sensible variations. |
| 4 |
Case series (and poor quality cohort and case-control studies§§) |
Analysis with no sensitivity analysis |
| 5 |
Expert opinion without explicit critical appraisal, or based on or based on physiology, bench research or “first principles” |
Expert opinion without explicit critical appraisal, economic theory or “first principles” |
* Homogeneity means a systematic review that is free of worrisome variations (heterogeneity) in the directions and degrees of results between individual studies. Not all systematic reviews with statistically significant heterogeneity need be worrisome, and not all worrisome heterogeneity need be statistically significant. Studies displaying worrisome heterogeneity should be tagged with a “-” at the end of their designated level.
§ Met when all patients died before the Rx became available, but some now survive on it; or when some patients died before the Rx became available, but none now die on it.
§§ Poor quality cohort study is defined as a study that failed to clearly define comparison groups and/or failed to measure exposures and outcomes in the same (preferably blinded), objective way in both exposed and non-exposed individuals and/or failed to identify or appropriately control known confounders and/or failed to carry out a sufficiently long and complete follow-up of patients. A poor quality case-control study is defined as a study that failed to clearly define comparison groups and/or failed to measure exposures and outcomes in the same (preferably blinded), objective way in both cases and controls and/or failed to identify or appropriately control known confounders.
†Better-value treatments are clearly as good but cheaper, or better at the same or reduced cost. Worse-value treatments are as good and more expensive, or worse and equally or more expensive.
SR: Systematic Review RCT: Randomized Clinical Trials Rx: Therapy
Schedule 2 – Comparable Dosage Forms
This Schedule identifies comparable dosage forms for the purpose of the Reasonable Relationship (RR) test for new patented drug products. Formulations within each group are considered comparable, but dosage forms in a different group are not.
The PMPRB reviews the list of comparable dosage forms periodically to ensure that it includes those currently used.
Comparable Dosage Formsx
| Topical (T) |
Nasal (N)/Pulmonary (P) |
Oral Solid (S) |
|
Aerosol
Aerosol (foam)
Cream
Disc (extended release)
Disc
Dressings
Gel
Gel (controlled release)
Liposomes
Liquid
Lotion
Ointment
Pad
Paint
Paste
Patch
Patch (Extended Release)
Pencil
Plaster
Powder
Shampoo
Soap Bar
Solution
Sponge
Spray
Spray (bag-on-valve)
Spray (metered dose)
Stick
Strip
Swab
Tincture
|
Aerosol
Aerosol-metered dose
Drops
Gas
Metered dose preparation
Powder
Powder (metered dose)
Solution
Solution (extended release)
Spray
Spray (metered dose)
Stick
|
Bar (chewable)
Caplet
Capsule
Effervescent granules
Effervescent powder
Effervescent tablet
Film (soluble)
Globules
Granules
Gum
Lozenge
Modified release caplet
Modified release capsule
Modified release tablet
Pellet
Piece (chewable)
Powder (extended release)
Strip
Tablet
Tablet (chewable)
Tablet (oral disintegrating)
Tablet for suspension
Wafer
|
blank
| Oral Liquid (L) |
Vaginal (V) |
Parenteral (J) |
|
Drops
Elixir
Emulsion
Gel
Granules for solution
Granules for suspension
Granules for suspension (delayed release)
Granules for suspension (extended release)
Liquid
Modified release liquid
Powder (extended release)
Powder for solution
Powder for suspension
Solution
Solution (extended release)
Spray
Suspension
Suspension (extended release)
Syrup
Syrup (extended release)
Tea (herbal)
Tincture
|
Cone
Cream
Douche
Foam
Gel
Gel (controlled release)
Implant
Insert
Insert (extended release)
Ovule
Pellet
Ring (slow release)
Sponge
Suppository
Suppository (sustained release)
Tampon
Vaginal tablet
Vaginal tablet (effervescent)
|
Bolus
Implant
Kit
Liposomes
Modified release injection
Pellet (implantable)
Powder for solution
Powder for suspension (sustained-release)
Solution
Solution (extended release)
Suspension for emulsion
Suspension (extended release)
|
blank
| Otic (E)/Ophthalmic (Y) |
Rectal (R) |
Dental/Sublingual Buccal (M) |
|
Drops
Gel
Gel (controlled release)
Implant
Insert
Insert (extended release)
Liquid
Modified release ocular device
Ointment
Powder for solution
Solution
Solution (extended release)
Suspension
|
Cream
Enema
Foam
Insert
Ointment
Ovule
Stick
Suppository
Suppository (sustained release)
Suspension
Suspension (extended release)
|
Emulsion
Film (soluble)
Floss
Gel
Gel (controlled release)
Gum
Lozenge
Metered-dose pump
Modified release buccal tablet
Mouthwash (gargle)
Paste
Powder (effervescent)
Powder for suspension
Solution
Solution (extended release)
Spray – buccal
Spray – sublingual
Stick
Strip
Sublingual tablet
Suspension
Suspension (extended release)
Swab
Tablet (orally disintegrating)
Tablet
Tooth paste
Tooth powder
Wafer
|
Schedule 3 – Therapeutic Class Comparison Test
1. Approach
The Therapeutic Class Comparison (TCC) test compares a new patented drug product’s National Average Transaction Price and the Market-Specific Average Transaction Prices in each class of customer – hospital, pharmacy, wholesaler and province/territory with the price of drug products identified for comparison purposes that are sold at prices that the PMPRB considers not to be excessive. Drug products are first identified for comparison purposes pursuant to sections C.8.1 to C.8.13 and then their prices are compared against those of the new patented drug product under review.
2. Measuring the Price
The PMPRB considers it appropriate to compare the prices of drug products used for comparison purposes taking into consideration the comparable dosage regimens determined pursuant to sections C.8.14 and C.8.15. The PMPRB will make these price comparisons in terms of the price per course of treatment or price per day, whichever is more applicable. Generally, the price per course of treatment will be applicable to acute indications, whereas price per day (based on maintenance dose) will be applicable to chronic situations.
Board Staff will use public sources for prices for the drug products used for comparison purposes. The sources of prices which Board Staff will consult are (in alphabetical order): the Association québécoise des pharmaciens propriétaires (AQPP); IMS Health; McKesson Canada; Ontario Drug Benefit (ODB) Programs; PPS Pharma; and the Régie de l’assurance maladie du Québec (RAMQ). Board Staff will select the lowest public price from these sources for each of the drug products identified for comparison purposes. If no price is available for a comparator from these sources, Board Staff will explore additional sources until a public price is found.
Board Staff may exclude from the TCC test any drug product it has reason to believe is being sold at an excessive price (see the PMPRB’s Policy on the Use of Patented and Non-Patented Drug Products in the Price Tests in section B.6).
Schedule 4 – Reasonable Relationship Test
In order to conduct the Reasonable Relationship (RR) test, the new patented drug product under review must meet four requirements:
- It must be the same chemical entity as the comparable drug product(s);
- It must have the same indication or use as the comparable drug product(s);
- It must be in the same or comparable dosage form as the comparable drug product(s) (see Schedule 2); and
- It must have the same dosage regimen as the comparable drug product(s).
Unless a patentee makes a submission claiming therapeutic improvement and HDAP identifies the new drug product as providing moderate or substantial therapeutic improvement, the RR test will be conducted if the four requirements are met.
Reasonable relationship refers to the association between strength per unit (see the Policy on Unit of Price Review in section B.4) and price. The RR test defines a Maximum Average Potential Price for the new strength of the patented drug product.
This schedule describes in general terms the process by which the reasonable relationship may be determined.
Board Staff will use public sources for prices for the drug products used for comparison purposes. The sources of prices which Board Staff will consult are (in alphabetical order): the Association québécoise des pharmaciens propriétaires (AQPP); IMS Health; McKesson Canada; Ontario Drug Benefit (ODB) Programs; PPS Pharma; and the Régie de l’assurance maladie du Québec (RAMQ). Board Staff will select the lowest public price from these sources for each of the drug products identified for comparison purposes. If no price is available for a comparator from these sources, Board Staff will explore additional sources until a public price is found.
Board Staff may exclude from the RR test any drug product it has reason to believe is being sold at an excessive price (see the PMPRB’s Policy on the Use of Patented and Non-Patented Drug Products in the Price Tests in section B.6).
The determination of reasonable relationship will be based on one of three possible tests to be considered in the following descending order:
Test 1: Same Strength Test
If there are one or more comparable drug products of the same strength as the new patented drug product, then the highest priced comparable drug product of the same strength determines the Maximum Average Potential Price for the new patented drug product. Prices above this threshold are considered to be excessive. The result of this test takes precedence over the other two tests.
In Figure 1 below, given three comparable drug products of equal strength but different prices (P1, P2, and P3) a new patented drug product will have a Maximum Average Potential Price (MAPP) equal to that of the highest priced comparable drug product, in this case P1.
Figure 1 – Same Strength Test
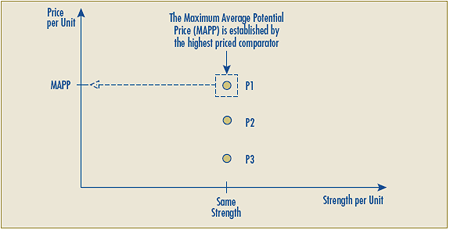
Test 2: Linear Relationship Test
If there are two or more comparable drug products, and none are the same strength as the new patented drug product, this test will be conducted.
The test is conducted in a series of steps:
- As shown in Figure 2A below, lines are drawn for all possible pairs of comparable drug products (e.g., A to B, A to C, B to C).
- The pair with a slope that is greater than or equal to zero and with the highest Y-axis intercept determines the Y-intercept for the line which will set the Maximum Average Potential Price. In the example in Figure 2A, the highest Y-intercept results from the line running from A to B.
Figure 2A – Linear Relationship Test – Representing Steps 1-2
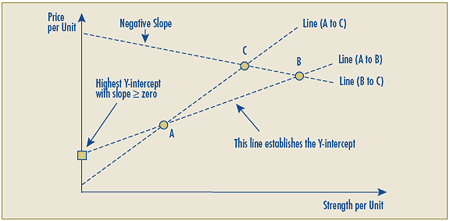
- A new line joining this Y-intercept with the point representing the per unit price of the highest priced comparable drug product is drawn. In the example in Figure 2B, the comparable drug product C has the highest price per unit.
- The National and Market-Specific Average Transaction Prices of the new patented drug product will not be presumed to be excessive if they do not exceed the Maximum Average Potential Price (MAPP) Line illustrated in Figure 2B below.
Figure 2B – Linear Relationship Test – Representing Steps 3-4
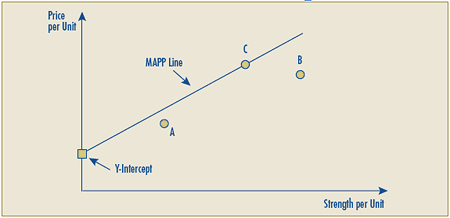
- It could happen that none of the lines with slopes greater than or equal to zero created by drawing lines through the prices of pairs of comparable drug products produces a Y-intercept greater than or equal to zero (i.e., only negative Y-intercepts, implying the Maximum Average Potential Price for some strengths would also be negative). Should this occur, the Maximum Average Potential Price line will then be established by drawing a line between the origin (0) and the per unit price of the highest priced comparable drug product. In the example in Figure 2C, the original Y-intercept would have been negative (determined using the same methodology as above in step 2). The Y-intercept used to establish the Maximum Average Potential Price (MAPP) Line cannot be less than zero, so the new Y-intercept is established at the origin (0). In this example, the Maximum Average Potential Price Line is drawn from the origin (0) through the point established by drug product C. This line would be used to establish the relationship between the strength of the new product and its Maximum Average Potential Price.
Figure 2C – Linear Relationship Test – Representing Step 5
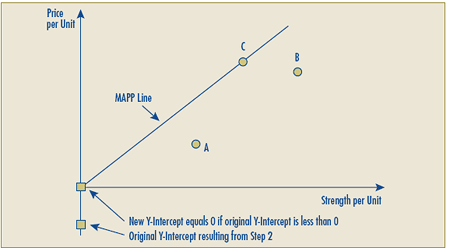
Test 3: Different Strength Test
This test is used when there is only one other (higher or lower) strength of a comparable drug product sold in Canada. Although there is only one other strength sold, there could be several products of this strength sold at different prices. The highest priced comparable drug product of the different strength is used for this test.
When the strength of the new patented drug product is higher than the strength of the comparable drug product, the Maximum Average Potential Price for the new patented drug product will be determined based on the proportional relationship of the strength of the new patented drug product compared to the comparable drug product multiplied by the price of the comparable drug product.
Example 1 (higher strength is introduced):
A 5 mg strength drug product is being sold and the highest price at which it is sold is $10.
A 7.5 mg strength new patented drug product is introduced.
The price of the new 7.5 mg patented drug product will be presumed to be excessive if it exceeds $15.00.
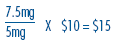
When the strength of the new patented drug product is lower than the strength of the comparable drug product, the Maximum Average Potential Price for the patented drug product will be equal to the price of the higher strength comparable drug product.
Example 2 (lower strength is introduced):
A 5 mg strength drug product is being sold and the highest price at which it is sold is $10.
A 2.5 mg strength new patented drug product is introduced.
The price of the new 2.5 mg patented drug product will be presumed to be excessive if it exceeds $10.00.
Schedule 5 – Median International Price Comparison Test
1. Median International Price Comparison (MIPC) Test
1.1 The median of the ex-factory prices of the same strength and dosage form of the same patented drug product for each country listed in the Regulations (France, Germany, Italy, Sweden, Switzerland, the United Kingdom, and the United States) will set the Maximum Average Potential Price for a new patented drug product when the Median International Price Comparison test is the pivotal introductory price test.
1.2 When the Median International Price Comparison test is being conducted and the new patented drug product is sold in an even number of countries, the median will be the simple average of the middle two prices.
1.3 When the new patented drug product is sold in fewer than five countries at the time it is first sold in Canada, the median international price will be calculated on an interim basis. At the end of three years or when the same patented drug product with the same strength and dosage form is sold in at least five countries, whichever occurs first, Board Staff will re-determine the median international price. Whenever this occurs, the drug product’s Non-Excessive Average Price will be the lower of:
- The re-determined median international price, and
- The Non-Excessive Average Price derived from the ordinary application of the CPI-Adjustment Methodology (see Schedule 9).
1.4 Where the re-determined median international price establishes a drug product’s Non-Excessive Average Price pursuant to section 1.3 above, the patentee is expected to reduce its National Average Transaction Price and Market-Specific Average Transaction Prices to the level of the Non-Excessive Average Price calculated in accordance with section 1.3 within the next two six-month reporting periods. If the patentee complies with this timeframe, its price will not be presumed to have been excessive.
2. Indirect International Price Comparison
2.1 When a direct international price comparison of the drug product under review is not possible because the drug product is only sold in Canada, the most similar strengths of comparable dosage forms (as per Schedule 2) of the same patented drug product may be considered.
3. Exchange Rates
3.1 To calculate the Median International Price Comparison test for a new patented drug product, the exchange rates used are the simple average of the thirty-six monthly average noon spot exchange rates for each country (taken to eight decimal places) as published by the Bank of Canada for the thirty-six months ending four months before the date of the first sale of the drug product.
e.g., if the new patented drug product under review was first sold in October 2009, the exchange rates used are for the months of June 2006 through May 2009.
3.2 Exchange rates Footnote 13 are published on the PMPRB website on a monthly basis.
Schedule 6 – Highest International Price Comparison Test
1. Highest International Price Comparison (HIPC) Test
1.1 Subject to Schedule 6, section 1.2, both at introduction and in future years, the Average Transaction Price of a patented drug product at the national level, for the pharmacy and hospital customer classes, and for each province and territory will be presumed to be excessive if it exceeds the highest price of the same strength and dosage form of the same patented drug product for each country listed in the Regulations (France, Germany, Italy, Sweden, Switzerland, the United Kingdom, and the United States).
1.2 Where the price of a patented drug product at the national level, for the pharmacy and hospital customer classes, and for each province and territory is above the highest international price for the same drug product, but there are other drug products of the same or comparable dosage forms of the same medicine and patentee (multiple DINs) also sold in Canada, the Maximum Average Potential Price and/or the Non-Excessive Average Price established for the patented drug product under review will not be lower than the price of a same or lower strength drug product which is within the Guidelines.
2. Exchange Rates
2.1 To calculate the HIPC test for a new patented drug product, the exchange rates used are the simple average of the thirty-six monthly average noon spot exchange rates for each country (taken to eight decimal places) as published by the Bank of Canada for the thirty-six months ending four months before the date of the first sale of the drug product. For example, if the new patented drug product under review was first sold in October 2009, the exchange rates used are for the months of June 2006 through May 2009.
2.2 To calculate the HIPC test for an existing patented drug product, the exchange rates used are the simple average of the thirty-six monthly average noon spot exchange rates for each country (taken to eight decimal places), as published by the Bank of Canada for the thirty-six months ending with the last month of the pricing period under review. For example, if the pricing period under review is July to December 2009, the exchange rates used are for the months of January 2006 through December 2009.
2.3 Exchange rates Footnote 14 are published on the PMPRB website on a monthly basis.
3. Existing Drug Products with Unusual Circumstances
3.1 The Guidelines require that patentees take appropriate action when an investigation concludes that the price of its patented drug product appears excessive. There are, however, circumstances where a patented drug product whose price does not appear to be excessive in one review period then appears excessive in a subsequent period, due to the application of the HIPC test. This could be as a result of events beyond the control of the patentee. The following are examples of three such circumstances:
- Exchange rate variations;
- A foreign regulator forcing price reductions; or
- The highest priced drug product is removed from the market.
Under the circumstances identified above, patentees will be notified that the patented drug product’s price appears excessive and will be expected to adjust the National Average Transaction Price and Market-Specific Average Transaction Prices for the pharmacy and hospital customer classes, and for each province and territory by the end of the next two reporting periods, in which case the price will not be presumed to have been excessive. Failing this, the patentee would be requested to submit a Voluntary Compliance Undertaking (VCU) and repay any excess revenues dating back to the first period in which the price exceeded the HIPC test. If the patentee declines to submit a VCU, then the matter would be reported to the Chairperson with the recommendation that a Notice of Hearing be issued.
Schedule 7 – International Therapeutic Class Comparison Test
1. Concept and Application
1.1 The International Therapeutic Class Comparison (ITCC) test compares the National Average Transaction Price of the patented drug product under review with the prices of comparable drug products that are sold in the seven comparator countries listed in the Regulations (France, Germany, Italy, Sweden, Switzerland, the United Kingdom, and the United States).
1.2 The ITCC test is not considered a primary price test. However, it may be appropriate to conduct an ITCC test in order to provide information in the context of an investigation into apparent excessive prices.
2. Selection of Comparable Drug Products
2.1 For the purpose of the ITCC test, the comparable drug products identified in the TCC test will be used. For more details on the selection of comparable drug products for the TCC test, please refer to sections C.8.1 to C.8.13.
2.2 In terms of comparable generic drug products, only those sold by the same company in the comparator country that sells the generic drug product in Canada will be included. For greater clarity, if a comparable generic drug product is sold by company “X” domestically, but internationally it is sold by companies “X” and “Y”, then only the generic drug product sold by company “X” will be considered in the ITCC test.
3. Derivation of the ITCC Test
3.1xi The following two methods may be used to calculate the ITCC test:
- The Straight Class Approach: The prices of all comparable drug products in the seven comparator countries listed in the Regulations are identified. The median international price is determined and compared against the National Average Transaction Prices of the patented drug product in Canada.
- The Ratio Approach: The prices of the drug product under review in the seven countries listed in the Regulations are identified. The prices of all comparable drug products in the seven comparator countries are also identified. The ratios between the price of the drug product under review and the price of comparable drug products are determined for each combination within each comparator country. The median of all the resulting ratios is then applied to the price of the pivotal comparator (i.e., the comparator used to establish the Maximum Average Potential Price in the domestic price test), which is then compared to the National Average Transaction Price of the patented drug product under review in Canada.
3.2 Where the price of a comparable drug product was excluded from the TCC test because it appeared to be excessive, it will also be excluded from the ITCC test.
4. Exchange Rates
4.1 To calculate the ITCC test for a new patented drug product, the exchange rates used are the simple average of the thirty-six monthly average noon spot exchange rates for each country (taken to eight decimal places), as published by the Bank of Canada for the thirty-six months ending four months before the date of the first sale of the drug product. For example, if the new patented drug product under review was first sold in October 2009, the exchange rates used are for the months of June 2006 through May 2009.
4.2 To calculate the ITCC test for an existing patented drug product, the exchange rates used are the simple average of the thirty-six monthly average noon spot exchange rates for each country (taken to eight decimal places), as published by the Bank of Canada for the thirty-six months ending with the last month of the pricing period under review. For example, if the pricing period under review is July to December 2009, the exchange rates used are for the months of January 2006 through December 2009.
4.3 Exchange rates Footnote 15 are published on the PMPRB website on a monthly basis.
Schedule 8 – Application of Price Tests for New Drug Products
| Level of Therapeutic Improvement |
Price Test Used for New Drug Product |
| Breakthrough |
MIPC test |
| Substantial Improvement |
Higher of:
- Top of the TCC test comprised of all drug products identified by HDAP pursuant to section C.8.6; and
- MIPC test.
|
| Moderate Improvement |
Higher of:
- Midpoint of:
- Top of the TCC test comprised of all drug products identified by HDAP pursuant to section C.8.7 and
- MIPC test; and
- Top of the TCC test comprised of all drug products identified by HDAP pursuant to section C.8.7. If it is impossible to conduct a TCC test (i.e., unable to derive comparable dosage regimens or the prices of the drug products used for comparison purposes appear to be excessive), then use the MIPC test.
|
| Slight or No Improvement
|
- Top of the TCC test comprised of all comparable drug products identified by HDAP pursuant to section C.8.9.
- In the exceptional cases where HDAP does not identify any comparable drug products, use the lower of
- the bottom of the TCC test comprised of all superior drug products identified by HDAP pursuant to section C.8.10 and
- the MIPC test.
- If it is impossible to conduct a TCC test (i.e., unable to derive comparable dosage regimens or the prices of the drug products used for comparison purposes appear to be excessive), then use the MIPC test.
Please note that additional Guidelines are applicable to the following drug products, unless the patentee makes a submission claiming therapeutic improvement and HDAP identifies the new patented drug product as providing moderate or substantial improvement:
- A patented generic drug product;
- A drug product that is a new presentation of the same chemical entity, with the same or comparable dosage form (as per Schedule 2), and the same indication or use;
- A combination drug product.
The Guidelines specific to these particular drug products are outlined in section C.11.10.
|
| All Levels of Therapeutic Improvement |
Notwithstanding the price tests above, the Average Transaction Price of a patented drug product at the national level, for the pharmacy and hospital customer classes, and for each province and territory will be presumed to be excessive if it exceeds the highest price of the same strength and dosage form of the same patented drug product for each country listed in the Regulations (France, Germany, Italy, Sweden, Switzerland, the United Kingdom, and the United States). |
Schedule 9 – CPI-Adjustment MethodologyFootnote 16 / xii
1. Consumer Price Index (CPI)-Adjustment Methodology
1.1 Subject to Schedule 9, subsection 1.5 below and Schedule 10, the National Average Transaction Price and the Market-Specific Average Transaction Prices of an existing patented drug product will be presumed to be excessive if they increase by more than that allowed under the Board’s CPI-Adjustment Methodology, as long as this price does not exceed the HIPC test.
1.2xiii The CPI-Adjustment Methodology involves the following calculations:
- Adjusting the benchmark prices of the drug product for the cumulative change in the CPI from the benchmark year to the year under review (CPI-Adjusted Price); and
- Applying a cap on the maximum price increase in any one year, equal to 1.5 times the change in the latest actual lagged CPI. In periods of high inflation (over 10%), the limit will be five percentage points more than the latest actual lagged change in the CPI.
1.3 The lower of the results of both calculations will set the Non-Excessive Average Price for a particular year.
1.4 The calculation of the Non-Excessive Average Price will be performed independently for each market (national, class of customer (pharmacy, hospital, wholesaler), province/territory) based on the price history in that market.
1.5 When a price reduction below the Non-Excessive Average Price is taken in one or more markets specifically to offset excess revenues (see Schedule 13), following said offset of excess revenues, the Average Transaction Prices in those markets may increase in the next reporting period up to the level of the Market-Specific Non-Excessive Average Transaction Prices prior to the price reduction.
2. Terminology
2.1 Forecast year:
The forecast year is the year for which Non-Excessive Average Prices are being determined.
2.2 Introductory Period:
The introductory period for new patented drug products is the period from the date of first sale to the end of the six-month regulatory reporting period (i.e., June 30 or December 31) when that period is greater than one month. For example, a patented drug product first sold in March 2009 would have an introductory period of March to June 2009, whereas a patented drug product first sold in December 2008 would have an introductory period of January to June 2009.
2.3 Benchmark Year:
- For patented drug products first sold in Canada more than three years prior to the forecast year, the benchmark year is the calendar year three years preceding the forecast year. For example, for 2009 the corresponding benchmark year is 2006.
- For patented drug products first sold three years or less prior to the forecast year, the benchmark year is the year in which the patented drug product was first sold in Canada.
2.4 Benchmark Prices:
- For patented drug products first sold three years or less prior to the forecast year, the national and market-specific benchmark prices of the patented drug product are, respectively, its National Average Transaction Price and Market- Specific Average Transaction Prices in the introductory period based on the patentee’s Form 2, Block 4 submission, or if those prices appear to be excessive, the Maximum Average Potential Price.
- For patented drug products first sold in Canada more than three years prior to the forecast year, the national and market-specific benchmark prices of the patented drug product are, respectively, its National Average Transaction Price and Market-Specific Average Transaction Prices in the benchmark year based on the patentee’s Form 2, Block 4 submission, or if those prices appear to be excessive, the Non-Excessive Average Price.
2.5xiv Base CPI:
Calculated as the annual average of the monthly increases in the CPI, as published by Statistics Canada, two years previous to the benchmark year. The base CPI figures are calculated annually by the PMPRB. For example, a 2010 base CPI will be used for the benchmark year of 2012.
2.6xv Actual Lagged CPI:
The actual lagged CPI used for the forecast year is based on the latest actual CPI published by Statistics Canada adjusted for the latest annual actual inflation also published by Statistics Canada. The actual lagged CPI is also published annually in the PMPRB’s January NEWSletter. There is a lag between the year of the actual CPI used and the year it is applied to. For example, the actual lagged CPI used for the forecast year of 2015 is the actual 2013 CPI published by Statistics Canada in January 2014.
2.7xvi CPI-Adjustment Factor:
The actual lagged CPI divided by the base CPI, rounded to three decimal places.
2.8 CPI-Adjusted Price:
This is the benchmark price multiplied by the CPI-adjustment factor for the benchmark year.
2.9xvii Cap:
In any year, the price increase of a patented drug product may not exceed 1.5 times the latest change in the actual lagged CPI. In times of high inflation (greater than 10%), the limit will be five percentage points more than the actual change in the CPI.
2.10xviii Example of the application of the CPI-Adjustment Methodology at the national level:
- Forecast Year: Jan – Dec 2015
- First sale: 1998
- Benchmark Year: 2012
- National Average Transaction Price in Benchmark Year: $10.0000
- National Average Transaction Price in 2013: $10.3900
- CPI-adjusted price: 1.054 (assuming this is the CPI-adjustment factor for 2012) × $10.0000 (National ATP in Benchmark Yr.) = $10.5400
- Cap: 1.020 (1.5 x actual lagged CPI for 2013 assumed to be 1.3%) × $ 10.3900 = $10.5978
- The 2015 National Non-Excessive Average Price for the patented drug product is the lower of the CPI-adjusted price and Cap: $10.5400.
Schedule 10 – DIP Methodology
1. DIP Methodology
1.1 Defining the DIP Methodology
If a price increase in excess of that allowable under the CPI-Adjustment Methodology is claimed by the patentee as due to the reduction or termination of benefit(s) – and the patentee provides the necessary evidence of the benefit(s) – the drug product’s National Non-Excessive Average Price and/or Market-Specific Non-Excessive Average Price(s) may increase beyond the level allowable under the CPI-Adjustment Methodology.
1.2 Eligible Benefits
Benefits are defined as “any reduction given as a promotion or in the form of rebates, discounts, refunds, free goods, free services, gifts or any other benefits of a like nature.”
1.3 Evidence of Benefits Required
Patentees wishing to invoke the DIP Methodology are expected to:
- Demonstrate that the recipient of the benefit was aware in advance that it was receiving a benefit not offered to all customers;
- Identify the type and value of benefit(s) and when/how it was offered;
- Provide evidence of the termination or reduction of a benefit(s);
- Whether the same customer is still receiving other benefits.
The exact form of this required evidence (e.g., a contract), and type of data (e.g., quantity of free goods, price discount, rebate value) will depend on the specifics of each case.
The DIP Methodology will not apply to cases where the apparent excessive increase in the National Average Transaction Price was due solely to a sales mix shift or when the price was reduced to offset excess revenues (see Schedule 13).
1.4 Application of the DIP Methodology to the Calculation of Non-Excessive Average Prices
When a Market-Specific Average Transaction Price of a drug product increases by more than that allowable under the CPI-Adjustment Methodology and the patentee provides the required evidence, the price of the drug product may increase up to the highest Non-Excessive Average Price of another class of customer (province/territory) without being presumed to be excessive, as long as this price does not exceed the HIPC test.
The ability to rebound to the highest Non-Excessive Average Price of another market is contingent on evidence demonstrating that benefits are no longer offered in the market employing the DIP Methodology. In markets where some benefits are ongoing, the DIP Methodology limits the rebound in price to a price commensurate with the remaining benefit. Markets are defined as hospital, wholesaler and pharmacy markets within the class of customer markets and each province and territory within the provincial/territorial markets.
1.5 Example of the application of the DIP Methodology
|
Hospital
MS-ATP* |
Wholesaler
MS-ATP* |
Pharmacy
MS-ATP* |
National
ATP** |
| Year 1 (Introduction) |
$8.00 |
$9.00 |
$10.00 |
$9.00 |
| Year 2 |
$6.00 |
$8.00 |
$10.00 |
$8.00 |
| Year 3 |
$10.00 |
$9.00 |
$10.00 |
$9.67 |
* MS-ATP refers to Market-Specific Average Transaction Price
** National Average Transaction Price
In year 3, the National Average Transaction Price increases beyond what would generally be permitted by the CPI-Adjustment Methodology. The patentee claims and provides evidence as follows:
- Hospital:
- Contracts offering a 20% discount off list price were negotiated through a Group Purchasing Organizations from date of first sale.
- Certain hospitals were offered even deeper discounts in year 2 due to the high volume of sales.
- In year 3, a new competitor entered the hospital market and the contracts were not renewed. Those hospitals that did purchase the patentee’s drug product paid full list price of $10.00, which is not presumed to be excessive due to the evidence of benefits provided.
- Wholesaler:
- Wholesalers were charged $9.00 in the first year and then the price was temporarily discounted in year 2 to preferred clients. By year 3, the one-year discount ended.
- The patentee provided evidence of the preferred pricing in year 2. No evidence of an introductory benefit was provided. The price of $9.00 is not presumed to be excessive due to the evidence of a benefit.
Schedule 11 – Criteria for Commencing an Investigation
The following criteria balance the need for pricing flexibility on the part of patentees with the PMPRB’s mandate of ensuring that the prices of patented drug products are not excessive. The Board publishes its criteria for commencing an investigation to improve transparency and to provide patentees with greater certainty as to their responsibilities.
A price is generally considered to be non-excessive if the National and Market-Specific Average Transaction Prices are equal to or below the Maximum Average Potential Price for the introductory period and their respective Non-Excessive Average Prices for all subsequent periods.
In order to allocate its resources to investigations as efficiently as possible the Board has developed criteria for when a price that exceeds the Guidelines will become the subject of an investigation.
| Criteria for Commencing an Investigationxix |
|
Board Staff will commence an investigation into the price of a patented drug product when any of the following criteria are met:
- The National Average Transaction Price or any Market-Specific Average Transaction Price of a new drug product exceeds the Maximum Average Potential Price during the introductory period by more than 5%.
- Excess revenues for a new or existing drug product are $50,000 or more.
- PMPRB receives a complaint.
|
Where the percentage by which the National Average Transaction Price exceeds the Maximum Average Potential Price or the National Non-Excessive Average Price, or the amount of excess revenues, is too small to trigger an investigation in one year, the patentee is expected to reduce the price of the patented drug product and to offset any excess revenues, as per the PMPRB’s Policy on the Offset of Excess Revenues (see section B.7). Evidence of persistent excessive pricing, even by amounts which do not trigger the investigation criteria, may result in an investigation.
Should the National Average Transaction Price of a patented drug product or its cumulative excess revenues meet the criteria, an investigation will be initiated. Patentees will be advised of the compliance status and cumulative excess revenues for each of their patented drug products.
Schedule 12 – “Any Market” Price Reviewsxix
The application of “Any Market” Price Reviews, for new and existing patented drug products, is generally limited to patented drug products first sold in Canada on or after January 1, 2010.
For New Patented Drug Products
For new patented drug products, the Maximum Average Potential Price will apply to the national level, for the pharmacy, hospital and wholesaler customer classes, and for each province and territory. In situations where the Maximum Average Potential Price is established by the results of the HIPC test, the wholesaler class of customer will not be constrained by the results of this test, and this market will instead be limited to the results of the appropriate domestic introductory price test.
Example 1
| Maximum Average Potential Price = |
$10 |
blank |
| National ATP* = |
$9 |
✔ |
| Market-Specific ATP* (Hospital) = |
$8 |
✔ |
| Market-Specific ATP* (Wholesaler) = |
$9 |
✔ |
| Market-Specific ATP* (Pharmacy) = |
$10 |
✔ |
No price would be presumed excessive, since neither the National Average Transaction Price nor the Market-Specific Average Transaction Prices exceed the Maximum Average Potential Price
* ATP means Average Transaction Price
Example 2
| Maximum Average Potential Price = |
$10 |
blank |
| National ATP* = |
$9 |
✔ |
| Market-Specific ATP* (Hospital) = |
$6 |
✔ |
| Market-Specific ATP* (Wholesaler) = |
$9 |
✔ |
| Market-Specific ATP* (Pharmacy) = |
$12 |
X |
Although the National Average Transaction Price is not presumed to be excessive, one Market-Specific Average Transaction Price (pharmacy) exceeds the Maximum Average Potential Price and would therefore be presumed to be excessive.
* ATP means Average Transaction Price
For Existing Patented Drug Products
Following the introductory period, Board Staff will only actively monitor the National Average Transaction Price of a patented drug product and compare it to the National Non-Excessive Average Price based on the application of the CPI-Adjustment Methodology and the Highest International Price Comparison test.
Board Staff will further investigate the changes in prices at the level of specific markets (class of customer; and/or province/territory) when an investigation is triggered, or as required as part of the monitoring of compliance with a VCU or Board Order.
To do this, each specific market’s Average Transaction Price will be compared against the Non-Excessive Average Price for that market. The Average Transaction Price at the national level, for the pharmacy and hospital customer classes, and for each province and territory will be compared against the results of the Highest International Price Comparison test.
Three possibilities could arise:
- No Market-Specific Average Transaction Price is presumed to be excessive. This could occur if the apparent increase in the National Average Transaction Price was solely due to a shift in the sales mix – i.e., the quantity sold in each market changed such that proportionately more was sold in a market with a higher Market-Specific Average Transaction Price than in the previous reporting period.
- One or more markets are found to have taken price increases that appear to be excessive. The patentee will be expected to reduce the Market-Specific Average Transaction Price(s) to the level of the Non-Excessive Average Price for the respective market(s). The National Average Transaction Price following the price reduction would then be considered to be non-excessive. Rather than calculate excess revenue based solely on the market(s) where the price was excessive, the excess revenues will be calculated based on the amount generated at the level of the National Average Transaction Price.
- The patentee may provide evidence that the increase in the particular market’s price was due solely to the reduction or termination of a “benefit” in that market. In this case, it may be appropriate to deviate from the CPI-Adjustment Methodology, as per the DIP Methodology outlined in Schedule 10.
Schedule 13 – Offset of Excess Revenues
Approaches to offset excess revenues
1.1 Subject to section 1.3.1 below, if the investigation criteria have not been triggered, patentees will be given the opportunity to take a voluntary price reduction to offset excess revenues.
1.2 Once the investigation criteria have been triggered, patentees will only be permitted to offset cumulative excess revenues pursuant to the specific terms of an approved VCU or a Board Order.
Offset excess revenues
1.3xxi Patentees are expected to offset excess revenues in a timely mannerxxii
1.3.1 In cases where the investigation criteria are not triggered (in accordance with Part B – Policies, section B.5.2), the Board will ensure transparency with respect to the potential accumulation of excess revenues by reporting these drugs as “does not trigger.”
1.3.2 As new investigations are commenced based on regulatory filings covering a full calendar year, offset of excess revenues shall also be calculated on the full calendar year.
1.3.3 In the context of a VCU, and subject to the specific terms of the VCU, patentees will generally be allowed:
- 30 days following the Board’s acceptance of the VCU to make payment; or
- Until the end of the following reporting period to offset excess revenues through a price reduction. Any excess revenues remaining at the end of the specified period would be due in payment.
Resumption of price level following excess revenue offset through a price reduction
1.4 When a price reduction below the Non-Excessive Average Price is taken in one or more markets specifically to offset excess revenues. Following the offset of excess revenues, the Average Transaction Prices in those markets may return in the next reporting period up to the Market-Specific Non-Excessive Average Prices prior to the price reduction.
Updates to the Compendium of Policies, Guidelines and Procedures
Since the new Guidelines came into effect on January 1, 2010, the PMPRB has been closely monitoring the effect of various changes. Any necessary additions, amendments and/or clarifications are promptly communicated to the patentees and are published in the quarterly NEWSletter on an ongoing basis. An updated version of the Compendium of Policies, Guidelines and Procedures is released annually in June.
The table below summarizes the revisions to date:
Blank
| Updated |
Subject |
Reference in Compendium |
NEWSletter Citation |
| February 2017 |
Policy on Generic Medicines |
Part B, Section B.8 |
Vol. 21, No. 1 |
| August 2016 |
Review of Prices of New Patented Drug Products at Introduction |
Section C.11.14 |
Vol. 20, No. 3 |
| June 2015 |
Comparable Dosage Forms |
Schedule 2, Table |
Vol. 19, No. 3 |
| June 2015 |
Adoption of lagged CPI-adjustment methodology |
Section C.12.3; Schedule 9, Section 1.2; Schedule 9, Section 2.5; Schedule 9, Section 2.6; Schedule 9, Section 2.7; Schedule 9, Section 2.9; Schedule 9, Section 2.10 |
Vol. 19, No.1
Blank
Blank
*October 18, 2013
|
| June 2015 |
Clarification: DIN transfer between patentees |
Part C, Section C.12.8 |
Vol. 19, No. 1 |
| April 2015 |
PMPRB electronic filing requirements |
Schedule 1 |
Vol. 19, No. 2 |
| April 2014 |
Requirements for HDAP Submissions |
Schedule 1 |
Vol. 18, No. 2 |
| January 2014 |
New Lagged CPI-Adjustment Methodology (to be applied beginning in 2015) |
Schedule 9, Footnote |
Vol. 18, No. 1* |
| May 2012 |
Criteria for Commencing an Investigation |
Schedule 11, Table |
Vol. 16, No. 2* |
| May 2012 |
Offset Excess Revenues |
Schedule 13, Section 1.3 |
Vol. 16, No. 2* |
| April 2012 |
“Any Market” Price Reviews |
Schedule 12 |
Vol. 16, No. 2 |
| January 2011 |
Existing Drug Products Subsequently Sold by Another Patentee |
Part C, Section C.12.8 |
Vol. 15, No. 1 |
| October 2010 |
Clarification that excess revenue is calculated on an annual basis |
Schedule 13, Section 1.3 (formerly indicated as Section 1.3.1) |
Vol. 14, No. 4 |
| October 2010 |
Policy on the Use of Patented and Non-Patented Drug Products in the Price Tests |
Part B, Section B.6.3, Part C, Section C.11.20 |
Vol. 14, No. 4 |
| April 2010 |
Derivation of the International Therapeutic Class Comparison Test |
Schedule 7, Section 3.1 |
Vol. 14, No. 2 |
*Consultation through Notice and Comment.
Note that revisions to the text are made on an as-required basis. Thus, in some years, there may not be any changes. Small editorial or grammatical changes that do not impact the substance of the text are not included in this table.
Endnotes:
- i Updated October 2010
- ii Updated February 2017
- iii Updated August 2016
- iv Updated October 2010
- v Updated June 2015
- vi Updated January 2011
- vii Updated June 2015
- viii Updated April 2014
- ix Updated April 2015
- x Updated June 2015
- xi Updated April 2010
- xii Updated January 2014
- xiii Updated June 2015
- xiv Updated June 2015
- xv Updated June 2015
- xvi Updated June 2015
- xvii Updated June 2015
- xviii Updated June 2015
- xix Updated May 2012
- xx Updated April 2012
- xxi Updated October 2010
- xxii Updated May 2012