
ISSN: 1495-0561
Catalogue no.: H78E-PDF
PDF version (2.60 MB)
Statistical Highlights 2016
Regulatory Mandate
- 1,435 patented drug products for human use were reported to the PMPRB, including 128 new drug products.
- 12 Voluntary Compliance Undertakings were accepted as at December 31, 2016.
- $5.0 million in excess revenues were offset by way of payment to the Government of Canada, in addition to price reductions.
Reporting Mandate
Sales Trends:
- There were $15.5 billion in sales of patented drug products in Canada in 2016, an increase of 2.6% from 2015.
- Patented drug products accounted for 60.8% of the total drug sales in Canada, a decrease from 61.6% in 2015.
Price Trends:
- Prices of existing patented drug products were stable, while the Consumer Price Index rose by 1.4%.
- Canadian prices were fourth highest among the seven PMPRB comparator countries, lower than prices in Switzerland, Germany and the US.
Research and Development R&D-to-sales ratios unchanged in 2016:
- 4.4% for all patentees, unchanged from 2015.
- 4.9% for Innovative Medicines Canada members, unchanged from 2015.
Research and Development (R&D):
- $918.2 million in total R&D expenditures were reported by patentees, an increase of 5.7% over 2015.
- $769.9 million in R&D expenditures were reported by Innovative Medicines Canada members, an increase of 0.3% over 2015.
Letter to the Minister
September 29, 2017
The Honourable Ginette Petitpas Taylor, P.C., M.P.
Minister of Health
House of Commons
Ottawa, Ontario
K1A 0A6
Dear Minister:
I have the pleasure to present to you, in accordance with sections 89 and 100 of the Patent Act, the Annual Report of the Patented Medicine Prices Review Board for the year ended December 31, 2016.
Yours very truly,
Dr. Mitchell Levine
Acting Chairperson
Acting Chairperson’s Message
As Acting Chairperson, I am pleased to present the Patented Medicine Prices Review Board’s (PMPRB) 2016 Annual Report. The PMPRB is a consumer protection agency with a dual regulatory
and reporting mandate. Its regulatory mandate is to ensure that the prices of patented medicines sold in Canada are not excessive. Its reporting mandate is to provide stakeholders with information on the latest trends in pharmaceutical sales and pricing and on pharmaceutical research and development (R&D) spending in Canada.
In December 2015, the PMPRB published its much anticipated 2015-2018 Strategic Plan, an important turning point in the organization’s history as it looks to reform how it carries out its consumer protection mandate in light of recent significant changes in its operating environment. 2016 marked the first full year that the strategic objectives in that document were in effect at the PMPRB and their impact was made apparent on a number of fronts. Chief among these was the Rethinking the Guidelines consultation initiative, which is a key initiative in the PMPRB’s short term efforts to modernize its regulatory framework. Phase 1 of this consultation, which ran from June to October 2016, sought stakeholder and public feedback on the Guidelines Modernization Discussion Paper. The written submissions received from interested parties in response to the discussion paper have been made available online and the PMPRB-led consultation process will resume following publication of the Minister of Health’s recently proposed amendments to the Patented Medicines Regulations in Part I of the Canada Gazette, which is anticipated to take place in the fall.
The PMPRB’s renewed emphasis on consumer-focused regulation made for another busy year of compliance and enforcement activity in 2016 with the acceptance of 12 Voluntary Compliance Undertakings (VCUs) and the paying back of excess revenues totalling $5,041,226.52 in addition to reductions in price for the affected drug products. As at May 31, 2017, an additional VCU was accepted in the amount of $31,000,000.00 from GlaxoSmithKline, a record one-time excess revenue repayment to the Government of Canada. In addition, the PMPRB’s first excessive price hearing in several years, and the first such case in which both public and private insurers sought to participate, continued to wind its way to disposition on the merits, with closing argument taking place in the spring of 2017.
In terms of its reporting mandate, the PMPRB continued to build strategic partnerships and raise public awareness of its mandate by being more responsive to the specific information needs of payers while at the same time expanding on the scope of its reporting to appeal to a broader stakeholder audience. An example of the latter is the release of the first edition of the PMPRB’s new Meds Entry Watch publication under the NPDUIS banner. This is an annual publication which explores the market entry dynamics of new drugs launched in Canada and other international markets and is designed to inform decision makers, researchers and patients of the evolving market dynamics associated with emerging drug therapies.
An example of the former is the PMPRB’s Market Intelligence Report on Biologic Response Modifying Agents, the first in a series of such reports which are designed to provide greater insight on specific therapeutic market segments of particular importance to public and private payers in Canada.
2016 saw many changes in our regulatory and reporting functions begin to take shape at the PMPRB which may have a profound effect on the pharmaceutical environment, but the departure of some cherished colleagues this past year was a change that was felt even more keenly and immediately by those of us within the organization. In June, my friend and fellow Board member, Mary Catherine Lindberg, completed her second and final term on the Board. Over the course of her tenure as a Board member, Ms. Lindberg made an invaluable contribution to the leadership of the PMPRB, particularly in her last five years as Chairperson, where she set the PMPRB on its current trajectory towards renewal, reform, and framework modernization. In December, Ginette Tognet, our Director of Regulatory Affairs and Outreach, announced her retirement after a long and illustrious career in the public service. Ms. Tognet played a formative role in shaping the professional lives of many staff who worked with her these last 18 years at the PMPRB. Ms. Lindberg and Ms. Tognet will be sorely missed and we wish them the very best in their future endeavours.
In closing, as many of our readers will know, Budget 2017 proposed a significant increase in funding for the PMPRB, as part of the Government’s commitment to making prescription drugs more accessible and affordable for Canadians. We take this as a vote of confidence in our potential to play a more meaningful and relevant role in the sustainability of Canada’s health systems. December of 2017 will mark the 30 year anniversary of the PMPRB and the timing could not be more opportune to celebrate our past success and embrace what looks to be a very promising future.
Dr. Mitchell Levine
Acting Chairperson
About the Patented Medicine Prices Review Board: Acting in the Interest of Canadians
The Patented Medicine Prices Review Board (PMPRB) was created in 1987 as the consumer protection “pillar” of Bill C-22, legislation which also strengthened the patent rights of pharmaceutical manufacturers in order to spur investment in research and development (R&D) in Canada.
The PMPRB protects consumers by regulating the price of patented drugs at the factory gate level and by keeping a vigilant eye on pricing trends and industry R&D. Through our reporting function, we serve as an objective, centralized source of information on pharmaceutical trends for policy makers, drug companies, private insurers and other stakeholders.
We are an Independent Authority
that administers provisions of Canada’s Patent Act related to patented medicines. As a member of the Health Portfolio, we contribute to sustainable health care systems for all Canadians.
1,435 Patented Drug Products
1,435 patented drug products were reported to the PMPRB in 2016.
The PMPRB is an independent, quasi-judicial body established by Parliament in 1987 under the Patent Act (Act).
The PMPRB is a consumer protection agency with a dual regulatory and reporting mandate. Through its regulatory mandate, it ensures that the prices of patented medicines sold in Canada are not excessive. The PMPRB also reports on trends in pharmaceutical sales and pricing for all medicines and on research and development (R&D) spending by patentees. Its reporting mandate provides pharmaceutical payers and policy makers with information to make rational, evidence-based reimbursement and pricing decisions.
Protecting Consumers in a Complex Marketplace
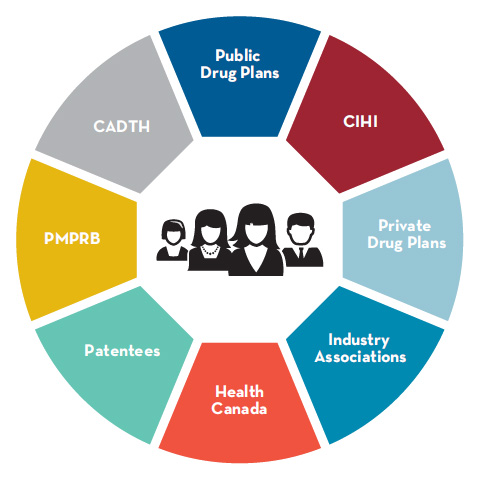
Figure description
Canadian consumers are symbolically represented by a graphic of a generic male and female in the centre of a segmented circle. Each segment of the circle represents an organization or category of organizations that are part of the Canadian pharmaceutical environment: Public Drug Plans; CIHI; Private Drug Plans; Industry Associations; Health Canada; Patentees; PMPRB; CADTH.
The PMPRB is part of the Health Portfolio, which includes Health Canada, the Public Health Agency of Canada, the Canadian Institutes of Health Research and the Canadian Food Inspection Agency. The Health Portfolio supports the Minister of Health in maintaining and improving the health of Canadians.
Although part of the Health Portfolio, because of its quasi-judicial responsibilities, the PMPRB carries out its mandate at arm’s length from the Minister of Health, who is responsible for the sections of the Act pertaining to the PMPRB. It also operates independently of other bodies such as Health Canada, which approves drugs for marketing in Canada based on their safety, efficacy and quality; federal, provincial and territorial public drug plans, which approve the listing of drugs on their respective formularies for reimbursement purposes; and the Common Drug Review, administered by the Canadian Agency for Drugs and Technologies in Health (CADTH), which recommends drugs that should qualify for reimbursement by participating
public drug plans.
The PMPRB is composed of Board Staff, who are public servants responsible for carrying out the organization’s day to day work, and Board members, Governor-in-Council appointees who serve as hearing panel members in the event of a dispute between Board Staff and a patentee over the price of a patented medicine.
Jurisdiction
Regulatory
The PMPRB regulates the “factory gate” ceiling prices for all patented drug products sold in Canadian markets; that is, the prices at which patentees (companies) sell their products to wholesalers, hospitals, pharmacies and other large distributers. The PMPRB does not regulate the prices of non-patented drugs.
The PMPRB’s jurisdiction is not limited to drug products for which the patent is for the active ingredient. Rather, its jurisdiction also covers drugs for which the patents relate to, but are not limited to, the processes of manufacture, the delivery system or dosage form, the indication/use and any formulations.
Under the Act, patentees (which include any parties who benefit from patents regardless of whether they are owners or licencees under those patents and regardless of whether
they operate in the “brand” or “generic” sector of the market) are required to inform the PMPRB of their intention to sell a new patented drug product. Upon the sale of a patented drug product, patentees are required to file price and sales information at introduction and, thereafter, until all patents pertaining have expired. Although patentees are not required to obtain approval of the price before a drug is sold, they are required to comply with the Act to ensure that the prices of patented drug products sold in Canada are not excessive.
Board Staff reviews the prices that patentees charge for each individual strength and form of a patented drug product. If the price of a patented medicine appears to be excessive, Board Staff will first try to reach a consensual resolution with the patentee. Failing this, the Chairperson can decide that the matter should proceed to a hearing. At the hearing, a panel composed of Board members acts as a neutral arbiter between Board Staff and the patentee. If a panel finds that the price of a patented medicine is excessive, it can order a reduction of the price to a non-excessive level. It can also order a patentee to make a monetary payment to the Government of Canada in the amount of the excess revenues earned and, in cases where the panel determines there has been a policy of excessive pricing, it can double the amount of the monetary payment.
Reporting
The PMPRB is a reliable, objective source of information on drug prices, pharmaceutical trends and R&D investment. The PMPRB reports annually to Parliament through the Minister of Health on its price review activities, the prices of patented medicines and price trends of all prescription drugs, and on the R&D expenditures reported by pharmaceutical patentees, as required by the Act.
Pursuant to an agreement by the federal, provincial and territorial (F/P/T) Ministers of Health in 2001, and at the request of the Minister of Health pursuant to section 90 of the Act, the PMPRB conducts critical analyses of price, utilization and cost trends for patented and non-patented prescription drugs under the National Prescription Drug Utilization Information System (NPDUIS). The PMPRB publishes the results of NPDUIS analyses in the form of research papers, posters, presentations and briefs. This program provides F/P/T governments and other interested stakeholders with a centralized, credible source of information on pharmaceutical trends.
Among other initiatives, the PMPRB also hosts various forums, such as webinars, research forums and information sessions, with academics and policy experts to discuss current research into pharmaceutical use in Canada and emerging areas for study.
Communications and Outreach
The PMPRB is committed to ensuring that stakeholders are consulted and informed of changes in the operating environment and are promptly advised of any updates to the regulatory process. Over the past year, the Regulatory Affairs and Outreach Branch continued to provide regular outreach sessions for patentees.
The PMPRB has also sustained its communication activities, continuing to take a proactive and plain-language approach to its traditional and social media presence. This included press release distribution, an emphasis on targeted social media campaigns, direct engagement with the public via social media as well as more traditional means (e.g., e-mail and telephone) and engagement with domestic, international and specialized media including the CBC, CTV, Radio-Canada, La Presse, The Globe and Mail, Toronto Star, the Canadian Medical Association Journal, Benefits Canada, CBS, Bloomberg News, Boston Globe, and VICE News.
The PMPRB has recently made additional improvements to its website to enhance accessibility of content, and continues to respond to public enquiries and inform the public by publishing updates of Board proceedings and decisions, and research results.
Governance
The Board consists of up to five members who serve on a part-time basis. Board Members, including a Chairperson and a Vice-Chairperson, are appointed by the Governor in Council. The Chairperson is designated under the Act as the Chief Executive Officer of the PMPRB, with the authority and responsibility to supervise and direct its work.
The Members of the Board are collectively responsible for the implementation of the applicable provisions of the Act. Together, they approve the issuance of the guidelines,
rules and other policies of the Board as provided by the Act and consult, as necessary, with stakeholders including the provincial and territorial Ministers of Health and representatives
of consumer groups and the pharmaceutical industry.
Members of the Board
Chairperson
The position of Chairperson is currently vacant.
Vice-Chairperson/Acting Chairperson
Mitchell Levine,
BSc, MSc, MD, FRCPC, FISPE
Dr. Mitchell Levine was appointed Member and Vice-Chairperson of the Board on March 3, 2011. He was reappointed as Vice-Chairperson for a second five year term on November 10, 2016.
Dr. Levine is a professor in the departments of Medicine and Clinical Epidemiology and Biostatistics in the Faculty of Health Sciences at McMaster University in Hamilton, Ontario. He is also Director of the Centre for Evaluation of Medicines at St. Joseph’s Healthcare in Hamilton.
Dr. Levine received his medical degree from the University of Calgary and did postgraduate medical training in Internal Medicine (FRCPC) and in Clinical Pharmacology at the University of Toronto. He received an MSc degree in Clinical Epidemiology from McMaster University.
Prior to his appointment to the Board, Dr. Levine was a member of the PMPRB’s Human Drug Advisory Panel. He currently acts on an ad hoc basis as a clinical pharmacology consultant to the Ontario Ministry of Health and Long-Term Care. In addition, he is Editor-in-Chief of the Journal of Population Therapeutics and Clinical Pharmacology and Associate Editor of the ACP Journal Club: Evidence-Based Medicine.
Members
Normand Tremblay,
ASC, MSc, Adm.A., CMC
Normand Tremblay was appointed Member of the Board on May 31, 2012. His term expires on May 31, 2017.
Mr. Tremblay is President and Chief Executive Officer of an innovative company (diaMentis inc.) which is currently developing a mental health diagnostic tool, and teaches at the Université du Québec in the area of management, project management and innovation. He brings to the Board a vast experience and expertise in strategic and operational planning and organizational development. For over 20 years, Mr. Tremblay has been active in various areas of the business field, nationally and internationally. He has also sat on investment committees and a number of administrative boards, including the National Research Council of Canada (NRC) from 2007 to 2010. Mr. Tremblay holds a master’s degree in project management (MSc) with a specialization in best practices in product development from the Université du Québec à Trois Rivières, as well as a certificate in business governance from Laval University
and is a Certified Management Consultant. He is also a member of the Order of Certified Administrators of Québec.
Carolyn Kobernick,
B.C.L., LL.B.
Carolyn Kobernick was appointed Member of the Board on June 13, 2014.
Ms. Kobernick is a lawyer and former public servant. Prior to her retirement in 2013, Ms. Kobernick was Assistant Deputy Minister of Public Law for the Department of Justice. As principal counsel to the Minister of Justice and Attorney General of Canada, Ms. Kobernick was instrumental in the development and delivery of policy for the Public Law sector. In
addition to identifying key strategic, legal and operational matters, she tackled cross-cutting national issues as the liaison between the Department of Justice and other government organizations.
Ms. Kobernick joined the Department of Justice in 1980, where she practiced litigation and tax law at the Toronto Regional office. In 1991, she was appointed Senior General
Counsel, Deputy Head, Business and Regulatory Law Portfolio, after working for over a decade in the legal services unit of the Correctional Service of Canada. In her role as Senior General Counsel, Ms. Kobernick was involved in complex federal policy and operational issues, including the Alaska Pipeline and Mackenzie Valley Pipeline files and the Sponsorship file.
During her career with the public service, Ms. Kobernick actively participated in many high-profile initiatives. She was Chair of the National Legal Advisory Committee and Departmental Champion for Aboriginal People and Gender Equity. She also served as the Senior Department of Justice official at the Domestic Affairs Cabinet Committee, and was appointed Senior Legal Advisor to the Government of Canada for the 2004 Gomery Inquiry.
Ms. Kobernick holds a B.C.L. and L.L.B. from McGill University and is a member of the bar of Ontario. In 2012 she obtained a Certificate in Adjudication for Administrative Agencies, Boards and Tribunals from the Osgoode Hall Law School and the Society of Ontario Adjudicators and Regulators.
As at May 31, 2017 two Member positions are vacant.
Organizational Structure and Staff
PMPRB Organizational Chart
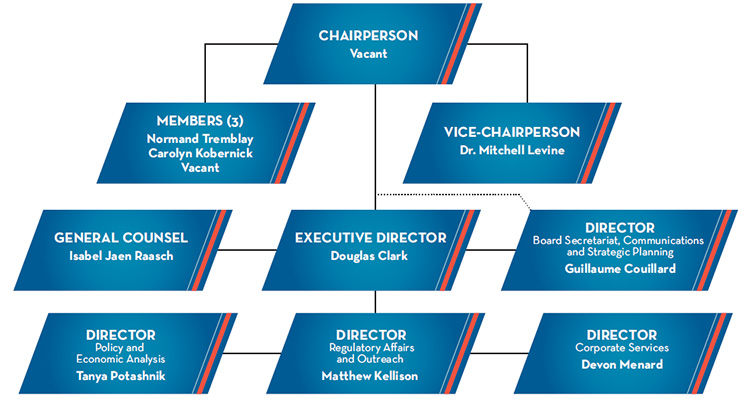
Figure description
This organizational chart illustrates the high-level reporting structure within the PMPRB, and lists the current Board and Senior Staff members. Board: Chairperson—Vacant; Vice-Chairperson—Dr. Mitchell Levine; Members—Normand Tremblay, Vacant, and Carolyn Kobernick. Senior Staff: Executive Director—Douglas Clark; General Counsel—Isabel Jaen Raasch; Director Board Secretariat, Communications and Strategic Planning—Guillaume Couillard; Director Policy and Economic Analysis—Tanya Potashnik; Director Regulatory Affairs and Outreach—Matthew Kellison; Director Corporate Services—Devon Menard.
Executive Director
The Executive Director is responsible for advising the Board and for the leadership and management of the Staff.
Regulatory Affairs and Outreach
The Regulatory Affairs and Outreach Branch reviews the prices of patented drug products sold in Canada to ensure that they are not excessive; encourages patentees to comply voluntarily with the Board’s Guidelines; implements related compliance policies; and investigates complaints into the prices of patented medicines. This branch also informs and educates patentees on the Board’s Guidelines and filing requirements.
Policy and Economic Analysis
The Policy and Economic Analysis Branch develops policy and strategic advice; makes recommendations on possible amendments to the Board’s Guidelines; conducts research and analysis on the prices of drugs, pharmaceutical market developments and R&D trends; and publishes studies aimed at providing F/P/T governments and other interested stakeholders with centralized, credible information in support of evidence based policy.
Corporate Services
The Corporate Services Branch provides advice and services in relation to human resources management; facilities; procurement; health, safety and security; information technology; and information management. It is also responsible for financial planning and reporting, accounting operations, audit and evaluation, and liaising with federal central agencies on these topics.
Board Secretariat, Communications and Strategic Planning
The Board Secretariat, Communications and Strategic Planning Branch develops and manages the PMPRB’s communications, media relations, and public enquiries; manages the Board’s meeting and hearing processes, including the official record of proceedings; and coordinates activities pursuant to the Access to Information Act and the Privacy Act. It is also responsible for strategic planning and reporting.
General Counsel
The General Counsel advises the PMPRB on legal matters and leads the legal team representing Board Staff in proceedings before the Board.
Budget
In 2016-17, the PMPRB had a budget of $10.965 million and an approved staff level of 71 full-time equivalent employees.
Table 1. Budget and Staffing
|
2015-16 |
2016-17 |
2017-18 |
| Budget* |
$10.945 M |
$10.965 M |
$10.866 M |
| Salaries |
$6.937 M |
$6.963 M |
$6.896 M |
| Operating |
$1.538 M |
$1.532 M |
$1.532 M |
| Special Purpose Allotment** |
$2.470 M |
$2.470 M |
$2.438 M |
| Full Time Employees (FTEs) |
71 |
71 |
66 |
* The amounts are based on the Main Estimates.
** The Special Purpose Allotment is reserved strictly for external costs of public hearings (legal counsel, expert witnesses, etc.). Any unspent funds are returned to the Consolidated Revenue Fund.
Regulating Prices of Patented Medicines: Continued Vigilance Necessary
With the population aging and using more prescription drugs—and sometimes more expensive types of drugs—Canada’s spending on pharmaceuticals is expected to increase significantly in the years to come. Medical advancements have introduced many innovative new drugs to the Canadian marketplace to improve existing treatments and to treat conditions that previously had no pharmaceutical therapy. These include high-cost orphan drugs, biologics and cancer drugs. The PMPRB plays an important role in regulating the prices of new and existing patented drug products to ensure the sustainability of Canadian health care systems.
The PMPRB protects the interests of Canadian consumers by ensuring that the prices of patented medicines sold in Canada are not excessive. It does this by reviewing the prices that patentees charge for each individual patented drug product to wholesalers, hospitals and pharmacies and by taking action so that patentees reduce their prices and pay back excess revenues where appropriate.
$195 M Excess Revenues
$195 Million in Excess Revenues have been Recovered by the PMPRB through Voluntary Compliance Undertakings and Board Orders since 1993. As at May 31, 2017, as a result of PMPRB investigations, 13 Voluntary Compliance Undertakings were accepted with $36.0 million in excess revenues offset by way of payment to the Government of Canada.
Reporting Requirements
Patentees are required by law to file information pertaining to the sale of their drug products in Canada. The Patent Act (Act) along with the Patented Medicines Regulations (Regulations) set out the filing requirements and Board Staff reviews pricing information on an ongoing basis to ensure that prices are not excessive until all patents pertaining have expired.
There are several factors used for determining whether a drug product is priced excessively, as outlined in section 85 of the Act. The Compendium of Policies, Guidelines and Procedures (Guidelines) details the price tests used by Board Staff to determine whether the price charged by a patentee falls within the maximum allowable price. The Guidelines were developed in consultation with stakeholders, including the provincial and territorial Ministers of Health, consumer groups, and the pharmaceutical industry. When an investigation determines that the price of a patented drug product may be excessive, the patentee is offered the opportunity to voluntarily lower its price and/or refund its excess revenues through a Voluntary Compliance Undertaking (VCU). If the patentee disagrees with the findings of the investigation and chooses not to submit a VCU, the Chairperson of the Board may issue a Notice of Hearing (NOH). After hearing the evidence, if the Board finds that a price is excessive, it can issue an Order requiring a patentee to reduce that price and/or refund excess revenues. A patentee also has the option of submitting a VCU to resolve the matter after the NOH has been issued. Copies of the Act, the Regulations, the Guidelines, and the Patentee’s Guide to Reporting are posted on the PMPRB’s website.
Failure to Report
The PMPRB relies on patentees’ full and timely disclosure of any and all patented drug products being sold in Canada to which a patent pertains. In 2016, 6 drug products were reported to the PMPRB for the first time despite being patented and sold prior to 2016. In addition, 2 drug products previously reported to the PMPRB, and for which the patents had expired, were reported again as having another patent pertaining. Table 2 lists the drug products that were patented and sold in Canada prior to being reported to the PMPRB.
Table 2. Failure to Report the Sale of Patented Drugs
| Currently sold by |
Brand name |
Generic name |
Year medicine came under
PMPRB's jurisdiction |
Year medicine came under
PMPRB's jurisdiction
with subsequent patent |
| BGP Pharma ULC |
Creon Minimicrospheres
(2 DINs) |
pancreatin |
2015 |
blank |
| Jazz Pharmaceuticals, PLC |
Defitelio
(1 DIN) |
defibrotide |
2014 |
blank |
| Ipsen Biopharmaceuticals Canada Inc. |
Dysport Aesthetic
(1 DIN) |
abobotulinumtoxinA |
2013 |
blank |
| CSL Behring Canada Inc. |
Corifact
(2 DINs) |
factor XIII concentrate, human |
2014 |
blank |
| Allergan Inc. |
Trelstar
(2 DINs) |
triptorelin pamoate |
2006 |
2011 |
Failure to File Price and Sales Data (Form 2)
Failure to file refers to the complete or partial failure of a patentee to comply with the regulatory filing requirements outlined in the Act and the Regulations. There were two
Board Orders issued for failure to file in 2016.
Scientific Review
Human Drug Advisory Panel
All new patented drug products reported to the PMPRB are subject to a scientific evaluation as part of the price review process. The Human Drug Advisory Panel (HDAP) was established by the Board to provide independent expertise and advice to Board Staff. HDAP conducts a review when a patentee makes a claim regarding therapeutic improvement. Panel members review and evaluate the appropriate scientific information available, including any submission by a patentee with respect to the proposed level of therapeutic improvement, the selection of drug products to be used for comparison purposes, and comparable dosage regimens.
HDAP evaluates the therapeutic benefit of new patented drug products according to the following definitions:
- Breakthrough: A drug product that is the first one to be sold in Canada to effectively treat a particular illness or effectively address a particular indication.
- Substantial Improvement: A drug product that, relative to other drug products sold in Canada, provides substantial improvement in therapeutic effects.
- Moderate Improvement: A drug product that, relative to other drug products sold in Canada, provides moderate improvement in therapeutic effects.
- Slight or No Improvement: A drug product that, relative to other drug products sold in Canada, provides slight or no improvement in therapeutic effects.
Figure 1 illustrates the breakdown of new patented drug products in the year of introduction by therapeutic benefit for 2010 to 2016. The largest percentage of patented drug products (82%) introduced since 2010 offer Slight or No Improvement in therapeutic benefit over existing therapiesFootnote 1. The bar “Overall 2016” represents the therapeutic benefit breakdown for all new patented drug products introduced from 2010 to 2016. The bar “Overall 2016 Revenue Share” illustrates the revenue share by therapeutic benefit for all drug new patented products introduced from 2010 to 2016.
Figure 1. Breakdown of New Patented Drug Products by Therapeutic Benefit
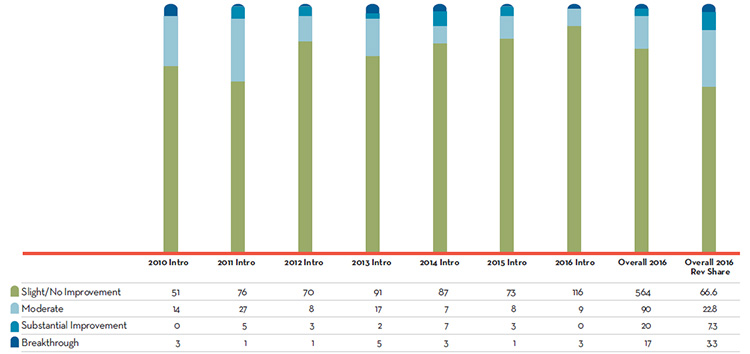
Figure description
This is a bar graph depicting the breakdown of new patented drug products by therapeutic benefit over existing drugs in the year of introduction for the period 2010 to 2016. In 2010: 51 new patented drug products were slight or no improvement, 14 were moderate improvement, 0 were substantial improvement, and 3 were breakthrough; 2011: 76 new patented drug products were slight or no improvement, 27 were moderate improvement, 5 were substantial improvement, and 1 was breakthrough; 2012: 70 new patented drug products were slight or no improvement, 8 were moderate improvement, 3 were substantial improvement, and 1 was breakthrough; 2013: 91 new patented drug products were slight or no improvement, 17 were moderate improvement, 2 were substantial improvement, and 5 were breakthrough; 2014: 87 new patented drug products were slight or no improvement, 7 were moderate improvement, 7 were substantial improvement, and 3 were breakthrough; 2015: 73 new patented drug products were slight or no improvement, 8 were moderate improvement, 3 were substantial improvement, and 3 breakthrough; 2016: 116 new patented drug products were slight or no improvement, 9 were moderate improvement, 0 were substantial improvement, and 3 were breakthrough.
Price Review
The PMPRB reviews the average price of each strength of an individual dosage form of each patented medicine. In most cases, this unit is consistent with the Drug Identification Number (DIN) assigned by Health Canada at the time the drug is approved for sale in Canada.
New Patented Drug Products Reported to the PMPRB in 2016
For the purpose of this report, a new patented drug product in 2016 is defined as any patented drug product first sold in Canada, or previously sold but first patented, between
December 1, 2015, and November 30, 2016.
There were 128 new patented drug products for human use reported as sold in 2016. Some are one or more strengths of a new active substance and others are new presentations of existing medicines. Of these 128 new patented drug products, 3 (2.3%) were being sold in Canada prior to the issuance of the Canadian patent that brought them under the PMPRB’s jurisdiction. Table 3 shows the year of first sale for these drug products.
Table 3. Number of New Patented Drug Products for Human Use in 2016 by Year First Sold
| Year first sold |
Number of drug products |
| 2016 |
125 |
| 2015 |
2 |
| 2013 |
1 |
| Total |
128 |
The list of New Patented Medicines Reported to PMPRB is available on the PMPRB’s website under “Regulating Prices”. This list includes information on the status of the review (i.e., whether the medicine is under review, within the Guidelines, under investigation, or subject to a VCU or Notice of Hearing).
Figure 2 illustrates the number of new patented drug products for human use reported to the PMPRB from 1989 to 2016.
Figure 2. New Patented Drug Products for Human Use
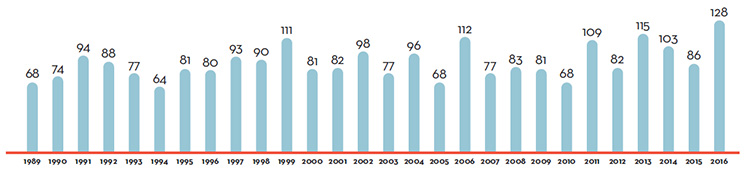
Figure description
This is a bar graph depicting the number of new patented drug products for human use reported to the Patented Medicine Prices Review Board by year. In 1989, 68 patented drug products for human use were reported to the PMPRB.
In 1990: 74; 1991: 94; 1992: 88; 1993: 77; 1994: 64; 1995: 81; 1996: 80; 1997: 93; 1998: 90; 1999: 111; 2000: 81; 2001: 82; 2002: 98; 2003: 77; 2004: 96; 2005: 68; 2006: 112; 2007: 77; 2008: 83; 2009: 81; 2010: 68; 2011: 109; 2012: 82; 2013: 115; 2014: 103; 2015: 86; 2016: 128.
Of these 128 new patented drug products, the prices of 70 had been reviewed as of March 31, 2017:
- 45 were found to be within the thresholds set out in the Guidelines;
- 13 were at a level that appeared to exceed the thresholds set out in the Guidelines by an amount that did not trigger the investigation criteria; and
- 12 were at levels that appeared to exceed the thresholds set out in the Guidelines and resulted in investigations being commenced.
For a complete list of the 128 new patented drug products and their price review status, see Appendix 2.
Price Review of Existing Patented Drug Products for Human Use in 2016
For the purpose of this report, existing patented drug products include all patented drug products that were first sold and reported to the PMPRB prior to December 1, 2015.
At the time of this report, there were 1307 existing patented drug products:
- 856 were priced within the thresholds set out in the Guidelines;
- 254 had prices that exceeded the thresholds set out in the Guidelines by an amount that did not trigger the investigation criteria;
- 89 were the subject of investigations:
- 3 were opened as the result of introductory pricing in 2013;
- 2 were opened as the result of introductory pricing in 2014;
- 8 were opened as the result of introductory pricing in 2015; and
- 76 were opened on the basis of year-over-year prices;
- 8 were under review;
- 39 were identified as patented generic drugs;
- 60 were the subject of Voluntary Compliance Undertakings; and
- 1 is the subject of a hearing.
In addition, 1 drug product remains the subject of a hearing although no longer patented in 2016.
A summary of the status of the price review of the new and existing patented drug products for human use in 2016 is provided in Table 4.
Table 4. Patented Drug Products for Human Use Sold in 2016—Status of Price Review as of March 31, 2017
|
New drug products introduced in 2016 |
Existing drug products |
Total |
| Total |
128 |
1307 |
1435 |
| Within Guidelines Thresholds |
45 |
856 |
901 |
| Under Review |
58 |
8 |
66 |
| Does Not Trigger Investigation |
13 |
254 |
267 |
| Under Investigation |
12 |
89 |
101 |
| Subject to Voluntary Compliance Undertakings1 |
0 |
60 |
60 |
| Price Hearings |
0 |
1 |
1 |
| Compliance status not reported as of 20162 |
0 |
39 |
39 |
1 The GlaxoSmithKline patent audit, which was described in the 2015 Annual Report, resulted in a Voluntary Compliance Undertaking (VCU) that included 45 drug products for a total of 60 drug products that were subject to a VCU for 2016.
2 As indicated in the February 2017 NEWSletter, an investigation of the price of a patented generic drug will only be commenced in accordance with the Policy on Generic Medicines.
Update From the 2015 Annual Report
- Reviews of all drug products for human use that were reported as Under Review in the 2015 Annual Report have been completed.
- 59 of the 93 investigations reported in the 2015 Annual Report resulted in one of the following:
- the closure of the investigation where it was concluded that the price was within the thresholds set out in the Guidelines;
- a Voluntary Compliance Undertaking (VCU) by the patentee to reduce the price and offset excess revenues through a payment and/or a reduction in the price of another patented drug product (see Voluntary Compliance Undertakings); or
- a public hearing to determine whether the price was excessive, including any remedial Order determined by the Board (see Hearings).
Patented Over-the-Counter Drug Products and Patented Drug Products For Veterinary Use
Board Staff reviews the prices of patented over-the-counter drug products or patented veterinary products only when a complaint has been received. No such complaints were received in 2016.
Voluntary Compliance Undertakings and Hearings
Voluntary Compliance Undertakings
A VCU is a written undertaking by a patentee to adjust its price to conform to the Board’s Guidelines. Under the Guidelines, patentees are given an opportunity to submit a VCU when Board Staff concludes, following an investigation, that the price of a patented drug product sold in Canada appears to have exceeded the thresholds set out in the Guidelines. A VCU represents a compromise between the PMPRB and the patentee as a result of negotiations between the parties geared towards a satisfactory resolution of an investigation initiated by Board Staff as per the Guidelines. A VCU takes into account the specific facts and underlying context of a particular case. As such, VCUs are not intended to have precedential value. A VCU can also be submitted by a patentee following the issuance of a Notice of Hearing.Footnote 2
In 2016, twelve VCUs were accepted. In addition to price reductions for certain drug products, excess revenues totaling $5,041,226.52 were offset by way of payments to the Government of Canada.
In 2017, as at May 31, 2017, one VCU has been approved by the Chairperson, in the matters of various GlaxoSmithKline Inc. drug products, in which $31,000,000.00 in excess revenues were offset by way of a payment to the Government of Canada.
Patentees are to ensure that the prices of their patented drug products are within the Board’s Guidelines during all periods in which the drug products are under the PMPRB’s jurisdiction.
Table 5. Voluntary Compliance Undertakings in 2016 up to May 31, 2017
| Patented drug product |
Therapeutic use |
Patentee |
Date of approval |
Offset of excessive revenues |
| Price reduction |
Payment to the government |
| VCUs in 2016 |
Actimmune1
(1 drug product) |
Chronic granulomatous disease and severe, malignant osteoporosis |
Horizon Pharma Ireland Limited |
May |
blank |
$590,519.57 |
Angiomax
(1 drug product) |
An anticoagulant in patients undergoing percutaneous coronary intervention and in the treatments of patients with moderate to high risk acute coronary syndromes due to unstable angina or non-ST-segment elevation in whom early percutaneous coronary intervention is planned |
Sunovion Pharmaceuticals Canada Inc. |
March |
blank |
$88,412.60 |
Apprilon
(1 drug product) |
Treatment of only inflammatory lesions (papules and pustules) or rosacea in adult patients |
Galderma Canada LP |
March |
✔ |
blank |
Cialis
(4 drug products) |
Treatment of erectile dysfunction and/or benign prostatic hyperplasia |
Eli Lilly Canada Inc. |
May |
✔ |
$3,256,005.27 |
Fibristal
(1 drug product) |
Treatment of moderate to severe signs and symptoms of uterine fibroids in adult women of reproductive age, who are eligible for surgery |
Allergan Inc. |
June |
blank |
$809,568.89 |
Mitosol
(1 drug product) |
Adjunct to ab externo glaucoma surgery |
Labtician Ophthalmics Inc. |
January |
blank |
$190.58 |
Neoral2
(1 drug product) |
Prevention of graft rejection following solid organ transplantation and treatment of transplant rejection |
Novartis Pharmaceuticals Canada Inc. |
February |
✔ |
$96,466.51 |
Oncaspar3,4
(1 drug product) |
A component of a multi-agent chemotherapeutic regimens to treat acute lymphoblastic leukemia |
Baxalta Canada Corporation |
December |
✔ |
blank |
Pergoveris
(1 drug product) |
Simulation of follicular development in women with severe LH and FSH deficiency |
EMD Inc. |
December |
blank |
$63.10 |
Samsca
(2 drug products) |
Treatment of clinically important, nonhypovolemic-hyponatremia |
Otsuka Canada Pharmaceutical Inc. |
April |
blank |
$200,000.00 |
Spiriva Respimat
(1 drug product) |
An add-on maintenance bronchodilator treatment in adult patients with asthma who are currently treated with the maintenance combination of inhaled corticosteroids (ICS ≥ 800 µg budesonide/day or equivalent) and long-acting ß agonists (LABA) and who experienced one or more severe exacerbations in the previous year |
Boehringer Ingelheim (Canada) Ltd |
May |
✔ |
blank |
Xalkori
(2 drug products) |
Monotherapy for use in patients with anaplastic lymphoma kinase (ALK)-positive locally advanced (not amenable to curative therapy) or metastic non-small cell lung cancer. |
Pfizer Canada Inc. |
June |
✔ |
blank |
| Total |
|
|
|
|
$5,041,226.52 |
| VCUs in 2017, up to May 31 |
| Various brand names |
Various drug products for various indications. |
GlaxoSmithKline Inc. |
March |
blank |
$31,000,000.00 |
| Overall total |
|
|
|
|
$36,041,226.52 |
1 Actimmune is not approved in Canada. It is made available by Horizon Pharma Inc., through one or more subsidiaries and affiliates, (collectively, “Horizon”) to Canadian patients under the Health Canada Special Access Programme. In the US, Actimmune is approved for Chornic Granulomatous Disease (“CGD”) and severe, malignant osteoporosis (“SMO”). It is currently being studied in Friederich’s Ataxia, a rare disease with no treatment.
2 These drug products were no longer patented in 2015 therefore, they are not included in the number of VCUs reported in Table 4 – Patented Drug Products for Human Use Sold in 2015.
3 Oncaspar is not approved in Canada. It is made available by Baxalta Canada Corporation to Canadian patients under the Health Canada Special Access Programme. In the US, Oncaspar is approved as a component of multi-agent chemotherapeutic regimens to treat acute lymphoblastic leukemia.
4 Excess revenues were offset by payments to customers that purchased Oncaspar between July 1, 2015 and June 30, 2016.
Hearings
The PMPRB holds hearings into two types of matters:
- excessive pricing; and
- failure to file–jurisdiction.
Excessive Pricing
In the event that the price of a patented medicine appears to be excessive, the Board can hold a public hearing. If it finds that the price is excessive, it may issue an order to reduce the price of the patented medicine in question (or of another patented medicine of the patentee) and/or to offset revenues received as a result of the excessive price. Judicial review of Board decisions can be sought in the Federal Court of Canada.
In January 2015, the PMPRB announced it would hold a public hearing in the matter of the price of the patented medicine Soliris, and Alexion Pharmaceuticals Inc. (Alexion), the pharmaceutical company that holds the patent for Soliris and sells the medicine in Canada. The purpose of this hearing is to determine whether the medicine has been or is being sold in any market in Canada at a price that, in the Board’s opinion, is or was excessive; and, if so, what order, if any, should be made to remedy the excessive pricing. The hearing was held in January, February and April 2017 and the matter is currently under advisement.
Remaining before the Board is the matter of whether Apo-Salvent CFC Free was excessively priced.
Failure to File–Jurisdiction
When Board Staff finds a patentee has failed or refused to provide the PMPRB with the pricing and sales information required by law, Board Staff will recommend that the Chairperson call a public hearing to determine whether the patentee is, in fact, bound by the reporting requirements of the Act and Regulations (i.e., under the PMPRB’s jurisdiction). If the Board Panel finds, as the result of a public hearing, that the patentee is in breach of its reporting requirements, the Board Panel may order the patentee to provide the PMPRB with the required pricing and sales information.
Following public complaints addressed to Board Staff concerning the price of two drug products, the PMPRB announced in February and March 2016 that it would hold separate public hearings with respect to whether Galderma Canada Inc. (Galderma) and Baxalta Canada Corporation (Baxalta) are required to provide the PMPRB with the pricing and sales information stipulated in the Patent Act and the Patented Medicines Regulations.
The Hearing Panel in the Galderma proceedings issued an Order on December 19, 2016 relating to the medicines Differin and Differin XP. The Hearing Panel found that one of the three patents at issue in the proceedings pertains to Differin and ordered Galderma to provide PMPRB staff with pricing and sales information required by section 80 of the Patented Medicines Regulations with respect to that medicine for the period between January 1, 2010 and March 14, 2016. The Hearing Panel dismissed PMPRB staff’s application with respect to the other two patents at issue. Following the Board's decision, Galderma brought an application for judicial review which is currently pending Federal Court.
The Hearing Panel in the Baxalta proceedings issued an Order on October 28, 2016, on consent of the parties, discontinuing the application, following Baxalta’s agreement to provide the information sought by Board Staff for the period commencing July 1, 2015, when Baxalta began to sell Oncaspar in Canada.
One Failure to File matter remains before the Board involving Apotex Inc.
Summary
Excess revenues totaling $36,041,226.52 were offset by way of payments to the Government of Canada through VCUs and Board Orders in 2016 and up to May 31, 2017.
Since 1993, a total of 121 VCUs have been approved and 30 public hearings initiated. These measures resulted in price reductions and the offset of excess revenues by way of additional price reductions and/or payments to the Government of Canada. Over $195 million has been collected through VCUs and Board Orders by way of payments to the Government of Canada and/or to customers such as hospitals and clinics.
Matters Before the Federal Court, Federal Court of Appeal and Supreme Court of Canada
On September 8, 2016, ratiopharm inc. (now Teva Canada Limited) and Sandoz Canada Inc. discontinued their application to the Supreme Court of Canada for leave to appeal the Federal Court of Appeal’s November 6, 2015 decision. This decision held that a person who sells a patented medicine pursuant to an express or an implied licence under a patent (or patents) is a “patentee” and subject to the PMPRB’s jurisdiction.
On January 18, 2017, Galderma Canada Inc. filed an application for judicial review of the Board’s decision dated December 19, 2016 in respect of its finding that Canadian Patent No. 2,478,237 pertains to Differin and ordering Galderma to file the required information for the period between January 1, 2010 and March 14, 2016. The matter is ongoing before the Federal Court.
There were also applications for judicial review before the Federal Court and Federal Court of Appeal in respect of Board decisions made in the context of the Soliris hearing, as detailed in Table 6 below. In particular, on September 11, 2015, Alexion filed an application for judicial review regarding the constitutionality of the Board. The Federal Court granted the Attorney General’s motion to strike this application on June 23, 2016. This was further upheld by a Federal Court Order dated December 28, 2016. On February 15, 2017, Alexion appealed this decision to the Federal Court of Appeal. The matter is ongoing.
Table 6. Status of Board Proceedings in 2016 up to May 31, 2017
Allegations of Excessive Pricing
| Patented drug product |
Indication/
use |
Patentee |
Issuance of notice of hearing |
Status |
| Apo-Salvent CFC-Free |
Asthma |
Apotex Inc. |
July 8, 2008 |
Ongoing |
| Soliris |
Paroxysmal nocturnal hemoglobinuria
Atypical hemolytic uremic syndrome |
Alexion Pharmaceuticals Inc. |
January 20, 2015 |
Board decision pending |
Allegations of Failure to File
| Patented drug product |
Indication/
use |
Patentee |
Issuance of notice of hearing |
Status |
| All medicines for which Apotex is a "patentee" |
blank |
Apotex Inc. |
March 3, 2008 |
Ongoing |
Differin
Differin XP
TactuPump
TactuPump Forte |
Acne |
Galderma Canada Inc. |
February 23, 2016 |
Hearing Panel issued an Order requiring Galderma to file information with respect to the medicine Differin for the period between January 1, 2010 and March 14, 2016. Galderma filed application for judicial review. |
| Oncaspar |
Acute Lymphoblastic
Leukemia |
Baxalta Canada
Corporation |
March 22, 2016 |
Hearing Panel issued an Order on October 28, 2016, on consent of the parties, discontinuing the application |
Judicial Review of Board Decisions and Appeals
| Patented drug product |
Indication/
use |
Patentee |
Issue |
Date of notice of hearing/status |
| ratio-Salbutamol HFA |
Asthma |
ratiopharm Inc.
(now Teva Canada
Limited) |
Allegations of
excessive pricing |
July 18, 2008
Application for leave to appeal at the Supreme Court of Canada discontinued: September 8, 2016 |
ratiopharm Inc.
(now Teva Canada
Limited) |
Failure to file
( jurisdiction) |
August 28, 2008
Application for leave to appeal at the Supreme Court of Canada discontinued: September 8, 2016 |
| Sandoz Canada Inc. |
Failure to file
( jurisdiction) |
March 8, 2010
Application for leave to appeal at the Supreme Court of Canada discontinued: September 8, 2016 |
| Soliris |
Paroxysmal Nocturnal Hemoglobinuria
Atypical Hemolytic Uremic Syndrome |
Alexion Pharmaceuticals Inc. |
Allegations of excessive pricing |
January 20, 2015
Court File T-1537-15
Notice of Appeal filed at Federal Court of Appeal: February 15, 2017 (A-51-17)
AG’s motion to strike granted: June 23, 2016 (2016 FC 716, aff’d 2017 FC 22)
Alexion filed application for judicial review: September 11, 2015
Court File T-1855-15
Order staying application: February 10, 2016
Alexion filed application for judicial review of October 5, 2015 Board Decision: November 3, 2015
Court File T-1160-16
Federal Court Order dismissing application: September 2, 2016 (aff’d 2017 FC 21)
Alexion filed application for judicial review of June 10, 2016 Board Decision: July 13, 2016
Court File T-110-17
Application for judicial review filed at the Federal Court: January 23, 2017, application abandoned: January 31, 2017 |
Differin
Differin XP |
Acne |
Galderma Canada Inc. |
Failure to file (jurisdiction) |
February 23, 2016
Court File T-83-17
Application for judicial review filed at the Federal Court: January 18, 2017 |
Key Pharmaceutical Trends: Drug Sales are on the Rise
Overall spending on pharmaceuticals is influenced by many factors, including price, utilization, the market entry of newer, more expensive drugs, and older drugs “going generic”. In 2016, sales of patented drugs increased by 2.6% and Canadian prices were fourth highest among the PMPRB’s comparator countries (PMPRB7).
$15.5 Billion Sales in Patented Drug Products
In 2016, sales of patented drug products increased to $15.5 billion from $15.1 billion in 2015.
Between 2006 and 2016 the number of medicines in Canada …
with an annual per beneficiary cost of at least $10,000 increased by over 200% and now account for 40% of patented drug sales as compared to 7.6% in 2006.
In 2016, half of the 10 top-selling drugs …
had annual treatment costs exceeding $10K.
Patented Drug Prices Increased Less than CPI
In 2016, the increase in patented drug prices was, on average, less than the rate of inflation, as measured by the Consumer Price Index (CPI), and therefore, did not contribute to sales growth.
15.4% Growth
Blood and blood forming organs had the greatest impact on sales growth in 2016. This class of drugs accounted for 5.7% of sales in 2016, an increase of 15.4% from the previous year.
Canada is a Top 10 Global Market
Canada is an important market for pharmaceuticals representing 1.9% of worldwide sales. Canada is consistently in the top 10 global markets for pharmaceuticals. Despite this, R&D-to-sales ratios are on average 5 times higher in PMPRB comparator countries than Canada, whereas prices are lower in the majority of these countries.
In 2014, Canadians spent 1.7% of gross domestic product on drugs.
This is the 2nd highest share in the PMPRB7, behind only the United States.
The PMPRB is responsible for reporting on trends in pharmaceutical sales and pricing for all medicines and for reporting research and development spending by patentees. In addition, the PMPRB undertakes studies and conducts analyses on a variety of topics related to pharmaceutical pricing and costs.
Trends in Sales of Patented Drug Products
Patentees are required under the Patented Medicines Regulations to submit detailed information on their sales of patented drug products, including quantities sold and net revenues received for each product by class of customer in each province/territory. The PMPRB uses this information to analyze trends in sales, prices and utilization of patented drug products.Footnote 3 This section provides key statistical results from this analysis.
Canadians spend much more today on patented drug products than they did a decade ago, but it is important to understand that an increase in drug spending does not in itself imply rising drug prices. For example, the PMPRB’s Annual Reports from 1995 through 2003 noted that sales of patented drug products grew at annual rates consistently exceeding 10%, while average annual rates of change for prices were less than 1%. In these instances, sales growth was driven by changes in the volume and composition of drug utilization.
A variety of factors can produce such changes. These include:
- increases in total population
- changes in the demographic composition of the population (for example, shifts in the age distribution toward older persons with more health problems)
- increases in the incidence of health problems requiring drug therapy
- changes in the prescribing practices of physicians (for example, a shift away from older, less expensive drug products to newer, more expensive medications, or a shift toward higher, more frequent dosages)
- increases in the use of drug therapy instead of other forms of treatment
- the use of new drug products to treat conditions for which no effective treatment existed previously
- the use of new drug products that enter the market at a higher price than previous treatments for a given condition
Sales Trends
Table 7 reports patentees’ total sales of patented drug products in Canada for 1990 through 2016. In 2016, sales of patented drug products increased to $15.5 billion from $15.1 billion in 2015, an increase of 2.6%. This is the lowest growth rate since 2012.
The third column of Table 7 gives sales of patented drug products as a share of overall drug sales. This share rose from 43.2% in 1990 to a peak of 72.7% in 2003. It declined over the 2004 to 2010 period, but has been quite stable since. That is, sales of non-patented brand and generic drug products have generally grown at similar rates as the sales of patented drug products in recent years.
The fourth column of Table 7 gives sales of patented drug products per Capita. Patented drug sales per capita rose from $61.6 in 1990 to $428.2 in 2016. The last column gives sales of patented drug products per GDP. Patented drug sales per GDP rose from 0.25 in 1990 to 0.77 in 2016.
Drivers of Sales Growth
Table 8 decomposes the sales growth that occurred between 2015 and 2016 into distinct elements reflecting the impacts of:
- previously patented drug products that have gone off-patent or left the Canadian market (“exiting drug effect”)
- patented drug products introduced to the Canadian market in 2016 (“new drug effect”)
- changes in prices among patented drug products with sales in Canada in both 2015 and 2016 (“price effect”)
- differences in the quantities of such drug products sold in the two years (“volume effect”)
- interactions of price and quantity changes (“cross effect”)
The first row of Table 8 gives these impacts as dollar amounts. The second row expresses the impacts as proportions of the overall change in sales between 2015 and 2016. For the sake of comparison, the third row provides average year-over-year proportionate impacts for 2011 through 2015.Footnote 5
The results in this table show that the increase in total sales that occurred between 2016 and 2015 was the result of two factors: increases in the quantity of existing drug products sold, and strong sales for new drugs, which offset a relatively large exiting drug effect. Proportionally, the 2016/2015 decomposition values are nearly identical to those in 2015/2014.
Table 7. Sales of Patented Drug Products, 1990–2016
| Year |
Patented drug products
Sales ($billions) |
Patented drug products
Change (%) |
Sales of patented drug products
as a share of all drug sales (%)* |
Patented drug sales per Capita |
Change (%) |
Patented drug sales per GDP (%) |
| 2016 |
15.5 |
2.6 |
60.8 |
$428.2 |
1.5 |
0.765 |
| 2015 |
15.1 |
9.4 |
61.6 |
$421.8 |
8.5 |
0.760 |
| 2014 |
13.8 |
3.1 |
59.9 |
$388.7 |
1.8 |
0.696 |
| 2013 |
13.4 |
4.2 |
60.7 |
$381.8 |
2.7 |
0.706 |
| 2012 |
12.9 |
0.1 |
59.2 |
$371.8 |
-1.2 |
0.708 |
| 2011 |
12.9 |
3.5 |
58.3 |
$376.1 |
3.1 |
0.729 |
| 2010 |
12.4 |
-4.3 |
55.8 |
$364.7 |
-5.7 |
0.746 |
| 2009 |
13.0 |
2.9 |
59.6 |
$386.9 |
1.9 |
0.829 |
| 2008 |
12.6 |
4.6 |
61.7 |
$379.5 |
2.9 |
0.762 |
| 2007 |
12.1 |
3.2 |
63.2 |
$368.9 |
2.5 |
0.769 |
| 2006 |
11.7 |
7.4 |
67.8 |
$360.0 |
6.3 |
0.784 |
| 2005 |
10.9 |
4.2 |
70.6 |
$338.5 |
2.8 |
0.769 |
| 2004 |
10.5 |
7.8 |
72.2 |
$329.2 |
7.2 |
0.789 |
| 2003 |
9.7 |
9.0 |
72.7 |
$307.0 |
8.0 |
0.776 |
| 2002 |
8.9 |
17.5 |
67.4 |
$284.3 |
16.0 |
0.748 |
| 2001 |
7.6 |
18.9 |
65.0 |
$245.2 |
19.1 |
0.666 |
| 2000 |
6.3 |
16.7 |
63.0 |
$205.9 |
15.9 |
0.571 |
| 1999 |
5.4 |
27.0 |
61.0 |
$177.6 |
24.3 |
0.538 |
| 1998 |
4.3 |
18.9 |
55.1 |
$142.9 |
15.4 |
0.459 |
| 1997 |
3.7 |
22.6 |
52.3 |
$123.7 |
22.1 |
0.409 |
| 1996 |
3.0 |
12.8 |
45.0 |
$101.4 |
14.2 |
0.350 |
| 1995 |
2.6 |
10.8 |
43.9 |
$88.7 |
7.2 |
0.314 |
| 1994 |
2.4 |
-2.1 |
40.7 |
$82.8 |
-1.4 |
0.304 |
| 1993 |
2.4 |
9.4 |
44.4 |
$83.9 |
7.9 |
0.322 |
| 1992 |
2.2 |
14.0 |
43.8 |
$77.7 |
8.8 |
0.307 |
| 1991 |
2.0 |
13.1 |
43.2 |
$71.4 |
16.0 |
0.286 |
| 1990 |
1.7 |
– |
43.2 |
$61.6 |
- |
0.245 |
* The denominator in this ratio comprises sales of patented, non-patented brand and generic drug products. Starting with the estimate for 2005, this value is derived from data contained in IMS Health’s MIDAS™ database. In previous years, IMS data were used to calculate sales of generic drug products only, while sales of non-patented brand products were estimated from data submitted by patentees. This approach was abandoned because of anomalies related to year-to-year changes in the set of companies reporting to the PMPRB. Ratios reported for years before 2005 likely overstate the patented share, but by only a small amount. This small bias in no way invalidates the strong upward trend evinced by the results for the years 1990 through 2003. Ratios since 2009 have also been revised slightly as a result of data updates from IMS Health - none of these adjustments resulted in a change greater than 0.4%.
Sources: PMPRB; MIDAS™ database, 2005–2016, IMS AG. All rights reserved.Footnote 6
Table 8. Decomposition of Changes in Sales of Patented Drug Products
|
Total change |
Exiting drug effect |
New drug effect |
Price effect |
Volume effect |
Cross effect |
Sales impact, 2016/2015
($millions) |
423.90 |
-385.43 |
296.17 |
-113.94 |
751.82 |
-124.71 |
Proportion of total change,
2016/2015 (%) |
100.00 |
-90.93 |
69.87 |
-26.88 |
177.36 |
-29.42 |
Average proportion of total change,
2011–2015 (%) |
100.00† |
-213.00 |
247.25 |
32.90 |
56.21 |
-23.35 |
† Value may not add due to rounding.
Source: PMPRB
Over the last decade there has been a significant shift in pharmaceutical development toward more specialized drugs, with an increasing number of higher-cost drugs compounded by a notable uptake in their use. As illustrated in Figure 3, for many years, the majority of the top 20 selling patented drugs had annual treatment costs in hundreds of dollars; however, the last two years marked a turning point, as most of the top 20 selling patented drugs now cost thousands or tens of thousands of dollars. This shifting trend is reflected in the exceptional tenfold growth in the median annual treatment cost of these drugs, which reached $8,584 in 2016. In addition to their higher cost, these drugs have had a remarkable uptake in utilization, elevating the weighted average annual treatment cost for the top 20 selling patented drugs to $17,770. While a decade ago this level marked the maximum average annual treatment cost, in 2016, the new maximum was $60,249.
Figure 3. Treatment Cost for Top 20 Selling Patented Drugs, 2006 to 2016
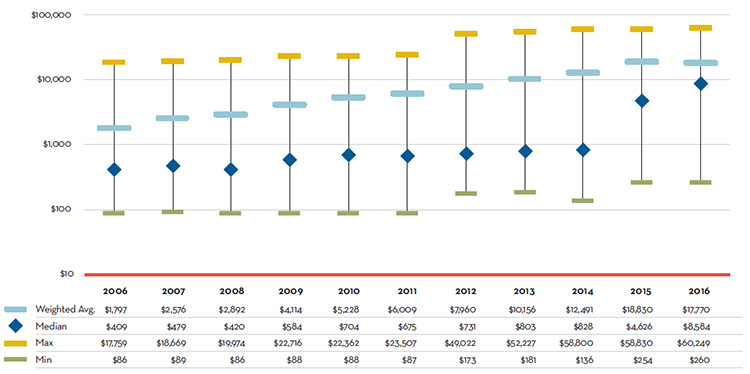
Source: PMPRB & QuintilesIMS, Private Drug Plan Direct Drug Plan Database, 2006-2016
Figure description
This is a bar graph depicting the minimum, maximum, median and weighted average annual treatment costs for the top 20 selling patented drug products in Canada from 2006 to 2016.
blank
| Treatment cost |
2006 |
2007 |
2008 |
2009 |
2010 |
2011 |
2012 |
2013 |
2014 |
2015 |
2016 |
| Maximum |
$17,759 |
$18,669 |
$19,974 |
$22,716 |
$22,362 |
$23,507 |
$49,022 |
$52,227 |
$58,800 |
$58,830 |
$60,249 |
| Weighted average |
$1,797 |
$2,576 |
$2,892 |
$4,114 |
$5,228 |
$6,009 |
$7,960 |
$10,156 |
$12,491 |
$18,830 |
$17,770 |
| Median |
$409 |
$479 |
$420 |
$584 |
$704 |
$675 |
$731 |
$803 |
$828 |
$4,626 |
$8,584 |
| Minimum |
$86 |
$89 |
$86 |
$88 |
$88 |
$87 |
$173 |
$181 |
$136 |
$254 |
$260 |
Figure 4 shows that high-cost drugs represent an increasingly significant share of the total cost of the patented drug market, rising steeply from 7.6% in 2006 to a remarkable 40.3% in 2016. This sustained growth was evident in all cost bands (10K to 20K; 20K to 50K and 50K+), with the steepest increase in the highest-cost drugs. While the new direct-acting antiviral drugs for hepatitis C were a major contributor to the growth in high-cost drugs, other high-cost drugs played an even more pronounced role. Despite the sharp increase in the share of costs, the number of patients benefiting from these drugs remained at less than 1% of the population.
Figure 4. Revenue Share of High-Cost Patented Drugs, 2006 to 2016
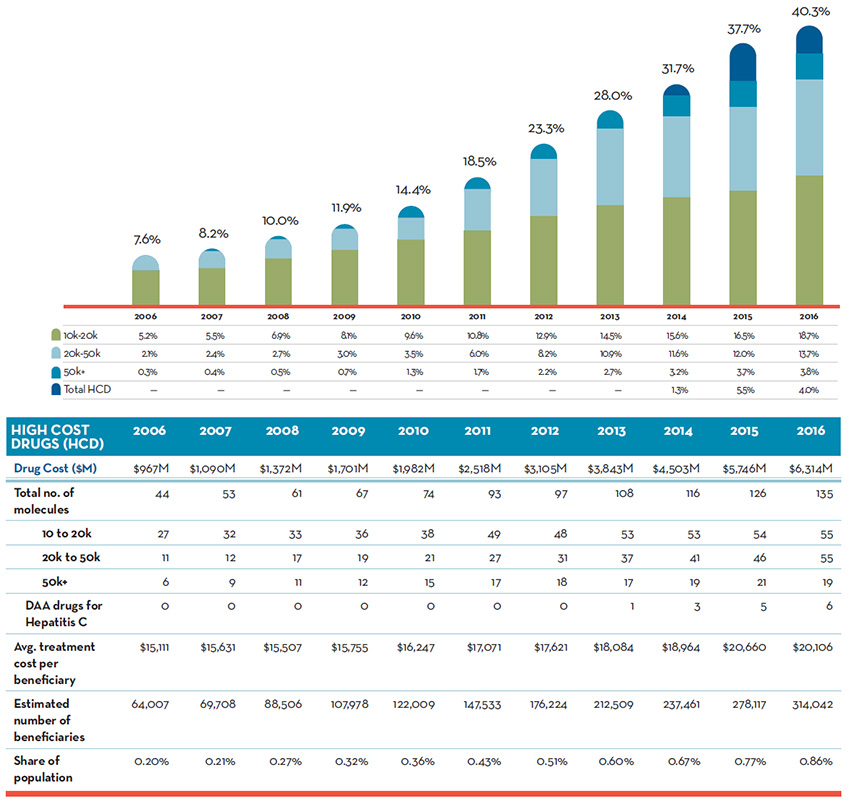
Numbers may not add due to rounding.
Source: PMPRB & QuintilesIMS, Private Drug Plan Direct Drug Plan Database, 2006-2016
Figure description
This is a bar graph depicting the high-cost drugs share of the total patented drug costs per year as a percentage for the period from 2006 to 2016. The bars are further subdivided to illustrate the share of total patented drug costs for high-cost drugs with an average annual cost per beneficiary of: $10 to $20 thousand; $20 to $50 thousand; greater than $50 thousand; and, direct-acting antiviral (DAA) drugs for hepatitis C
blank
| Year |
Share for drugs costing $10 to $20 thousand |
Share for drugs costing $20 to $50 thousand |
Share for drugs costing greater than $50 thousand |
DAA drugs for hepatitis C |
Total share of high-cost drugs |
| 2006 |
5.2% |
2.1% |
0.3% |
|
7.6% |
| 2007 |
5.5% |
2.4% |
0.4% |
|
8.2% |
| 2008 |
6.9% |
2.7% |
0.5% |
|
10.0% |
| 2009 |
8.1% |
3.0% |
0.7% |
|
11.9% |
| 2010 |
9.6% |
3.5% |
1.3% |
|
14.4% |
| 2011 |
10.8% |
6.0% |
1.7% |
|
18.5% |
| 2012 |
12.9% |
8.2% |
2.2% |
|
23.3% |
| 2013 |
14.5% |
10.9% |
2.7% |
|
28.0% |
| 2014 |
15.6% |
11.6% |
3.2% |
1.3% |
31.7% |
| 2015 |
16.5% |
12.0% |
3.7% |
5.5% |
37.7% |
| 2016 |
18.7% |
13.7% |
3.8% |
4.0% |
40.3% |
The table below the graph gives additional information including the total drug cost, number of molecules, average annual treatment cost per patient and number of users.
blank
|
2006 |
2007 |
2008 |
2009 |
2010 |
2011 |
2012 |
2013 |
2014 |
2015 |
2016 |
| Total drug cost |
$967 million |
$1,090 million |
$1,372 million |
$1,701 million |
$1,982 million |
$2,518 million |
$3,105 million |
$3,843 million |
$4,503 million |
$5,746 million |
$6,314 million |
| Total number of high-cost molecules |
44 |
53 |
61 |
67 |
74 |
93 |
97 |
108 |
116 |
126 |
135 |
| Number of molecules costing $10 to $20 thousand |
27 |
32 |
33 |
36 |
38 |
49 |
48 |
53 |
53 |
54 |
55 |
| Number of molecules costing $20 to $50 thousand |
11 |
12 |
17 |
19 |
21 |
27 |
31 |
37 |
41 |
46 |
55 |
| Number of molecules greater than $50 thousand |
6 |
9 |
11 |
12 |
15 |
17 |
18 |
17 |
19 |
21 |
19 |
| Number of direct-acting antiviral drugs for hepatitis C |
|
|
|
|
|
|
|
1 |
3 |
5 |
6 |
| Average annual treatment cost per beneficiary |
$15,111 |
$15,631 |
$15,507 |
$15,755 |
$16,247 |
$17,071 |
$17,621 |
$18,084 |
$18,964 |
$20,660 |
$20,106 |
| Estimated number of high-cost drug beneficiaries |
64,007 |
69,708 |
88,506 |
107,978 |
122,009 |
147,533 |
176,224 |
212,509 |
237,461 |
278,117 |
314,042 |
| Share of users of high-cost drugs in general beneficiary population |
0.20% |
0.21% |
0.27% |
0.32% |
0.36% |
0.43% |
0.51% |
0.60% |
0.67% |
0.77% |
0.86% |
Figure 5 breaks down 2016 sales of patented drug products according to the year in which the product was first sold in Canada. Throughout the latter part of the 1990s and early 2000s, sales growth was largely driven by a succession of new “blockbuster” products that ultimately achieved very high sales volumes. Despite the recent patent expiries (“patent cliff”), these products still accounted for a considerable share of patented drug sales in 2016. Most significant, however, was the introduction of several highly effective treatments for Hepatitis C in 2014, which has significantly increased the share of sales attributable to drugs released in that year.
Figure 5. Share of 2016 (%) Sales of Patented Drug Products by Year of Introduction
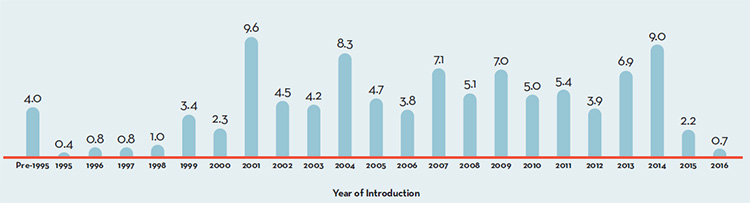
Figure description
This is a bar graph depicting the share of sales, in percent, of patented drug products by the year in which the product was first sold in Canada. Drugs introduced before 1995 comprised 4.0% of the share of sales. In 1995: 0.4; 1996: 0.8; 1997: 0.8; 1998: 1.0; 1999: 3.4; 2000: 2.3; 2001: 9.6; 2002: 4.5; 2003: 4.2; 2004: 8.3; 2005: 4.7; 2006: 3.8; 2007: 7.1; 2008: 5.1; 2009: 7.0; 2010: 5.0; 2011: 5.4; 2012: 3.9; 2013: 6.9; 2014: 9.0; 2015: 2.2; 2016: 0.7.
Sales by Therapeutic Class
The PMPRB classifies drug products according to the World Health Organization’s (WHO) Anatomical Therapeutic Chemical (ATC) system when it conducts analyses. This is a scientific, hierarchical system that classifies drug products according to their principal therapeutic use and chemical composition. At its first level of aggregation (Level 1), the ATC system classifies drug products according to the element of human anatomy with which they are primarily associated.
Table 9 breaks out sales of patented drug products in Canada in 2016 by ATC Level 1. The table gives the 2016 sales for each class, the share of the total sales this represents and the rate at which sales grew relative to 2015. Values in the last column represent the component of overall sales growth attributable to drug products in the corresponding therapeutic class.Footnote 7 By this measure, antineoplastics and immunomodulating agents and alimentary tract and metabolism made the largest contribution to sales growth. Lower sales of both general antiinfectives for systemic use and antiparasitic products, and genito-urinary system and sex hormones drugs also had a significant impact on overall expenditure.
Table 9. Sales of Patented Drug Products by Major Therapeutic Class, 2016
| Therapeutic class |
2016 sales
($millions) |
Share: 2016 sales (%) |
Growth: 2016/2015 ($millions) |
Growth: 2016/2015 (%) |
Impact on change in expenditure (%) |
| A: Alimentary tract and metabolism |
1,717.5 |
11.1 |
118.2 |
7.4 |
28.9 |
| B: Blood and blood forming organs |
883.1 |
5.7 |
117.7 |
15.4 |
28.8 |
| C: Cardiovascular system |
830.4 |
5.4 |
20.7 |
2.6 |
5.1 |
| D: Dermatologicals |
125.2 |
0.8 |
7.4 |
6.2 |
1.8 |
| G: Genito-urinary system and sex hormones |
403.9 |
2.6 |
-132.7 |
-24.7 |
-32.5 |
| H: Systemic hormonal preparations |
70.9 |
0.5 |
8.4 |
13.4 |
2.0 |
J: General antiinfectives for systemic use and
P: Antiparasitic products* |
2,232.7 |
14.4 |
-202.1 |
-8.3 |
-49.5 |
| L: Antineoplastics and immunomodulating agents |
5,143.2 |
33.2 |
484.9 |
10.4 |
118.6 |
| M: Musculo-skeletal system |
396.5 |
2.6 |
34.8 |
9.6 |
8.5 |
| N: Nervous system |
1,552.8 |
10.0 |
-78.6 |
-4.8 |
-19.2 |
| R: Respiratory system |
1,245.2 |
8.0 |
8.9 |
0.7 |
2.2 |
| S: Sensory organs |
798.7 |
5.2 |
7.1 |
0.9 |
1.7 |
| V: Various |
95.9 |
0.6 |
14.1 |
17.2 |
3.4 |
| All therapeutic classes † |
15,496.0 |
100.0 |
408.8 |
2.6 |
100.0 |
† Values in this row may not add due to rounding.
* These groups have been combined for reasons of confidentiality.
Source: PMPRB
As shown in Table 10, high-cost drugs represent an increasingly significant share of the total cost of the patented drug market, rising steeply from 7.6% in 2006 to a remarkable 40.3% in 2016. This sustained growth was evident in all cost bands (10K to 20K; 20K to 50K and 50K+), with the steepest increase in the highest-cost drugs. While the new direct-acting antiviral drugs for hepatitis C were a major contributor to the growth in high-cost drugs, other high-cost drugs played an even more pronounced role. Despite the sharp increase in the share of costs, the number of patients benefiting from these drugs remained at less than 1% of the population.
Table 10. Treatment Cost for Top
10 Selling Patented Drugs,
2006 to 2016
| 2006 |
2011 |
2016 |
| Chemical/Brand |
Annual cost/
beneficiary |
Chemical/Brand |
Annual cost/
beneficiary |
Chemical/Brand |
Annual cost/
beneficiary |
| 1. Atorvastatin Calcium (Lipitor) |
$511 |
1. Rosuvastatin Calcium (Crestor) |
$408 |
1. Infliximab (Remicade)b |
$28,446 |
| 2. Amlodipine Besylate (Norvasc) |
$417 |
2. Infliximab (Remicade)b |
$23,507 |
2. Adalimumab (Humira)b |
$15,843 |
| 3. Ramipril (Altace) |
$271 |
3. Salmeterol Xinafoate/ Fluticasone Propionate (Advair) |
$414 |
3. Ledipasvir/Sofosbuvir (Harvoni)b |
$44,333 |
| 4. Venlafaxine Hydrochloride (Effexor) |
$446 |
4. Adalimumab (Humira)b |
$14,025 |
4. Ranibizumab (Lucentis)a |
$8,643 |
| 5. Pantoprazole Sodium (Pantoloc) |
$330 |
5. Clopidogrel Bisulfate (Plavix) |
$683 |
5. Etanercept (Enbrel)b |
$13,633 |
| 6. Clopidogrel Bisulfate (Plavix) |
$607 |
6. Etanercept (Enbrel)b |
$13,226 |
6. Salmeterol Xinafoate/ Fluticasone Propionate (Advair) |
$451 |
| 7. Rosuvastatin Calcium (Crestor) |
$341 |
7. Esomeprazole (Nexium) |
$441 |
7. Lenalidomide (Revlimid)b |
$60,249 |
| 8. Olanzapine (Zyprexa) |
$977 |
8. Ranibizumab (Lucentis)a |
$6,740 |
8. Immune Globulin Intravenous (Human) (Gammagard)a |
$5,546 |
| 9. Salmeterol Xinafoate/ Fluticasone Propionate (Advair) |
$343 |
9. Oxycodone Hydrochloride (Oxycontin) |
$767 |
9. Insulin Glargine (Lantus) |
$767 |
| 10. Infliximab (Remicade)b |
$17,759 |
10. Immune Globulin Intravenous (Human) (Gammagard)a |
$4,651 |
10. Aflibercept (Eylea)a |
$8,525 |
a Biologics with an annual treatment cost < $10K
b Biologics with an annual treatment cost > $10K
Source: PMPRB & QuintilesIMS, Private Drug Plan Direct Drug Plan Database, 2006-2016
Price Trends
The PMPRB uses the Patented Medicines Price Index (PMPI) to monitor trends in prices of patented drug products. The PMPI measures the average year-over-year change in the ex-factory prices of patented drug products sold in Canada. The index is constructed using a formula that takes a sales-weighted average of price changes observed at the level of individual drug products.Footnote 8 This is similar to the approach Statistics Canada uses to construct the Consumer Price Index (CPI). The PMPI is based on an average transaction price and sales information for a six-month period submitted by patentees.
It is important to understand the conceptual relationship between the PMPI and drug costs. The PMPI does not measure changes in the utilization of patented drug products; a quantity index, the PMQI, is calculated for this purpose (see Utilization of Patented Drug Products). The PMPI does not measure the cost impact of changes in prescribing patterns or the introduction of new medicines. By design, the PMPI isolates the component of sales growth attributable to changes in prices.
Figure 6 provides year-over-year changes in the PMPI for the years 1988 through 2016. As measured by the PMPI, prices of patented drug products decreased from 0.9 in 2015 to -0.5 in 2016.
Figure 6. Annual Rates of Change (%), Patented Medicines Price Index (PMPI), 1988–2016
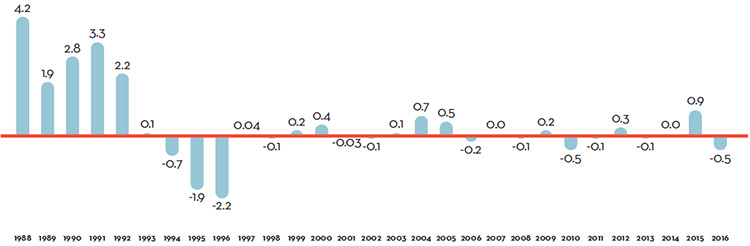
Figure description
This is a bar graph depicting the year-over-year changes, in percent, in the Patented Medicines Price Index (PMPI) for the years 1988 through 2016. In 1988, the rate of change in the PMPI was 4.2. 1989: 1.9; 1990: 2.8; 1991: 3.3; 1992: 2.2; 1993: 0.1; 1994: -0.7; 1995: -1.9; 1996: -2.2; 1997: 0.04; 1998: -0.1; 1999: 0.2; 2000: 0.4; 2001: -0.03; 2002: -0.1; 2003: 0.1; 2004: 0.7; 2005: 0.5; 2006: -0.2; 2007: 0.0; 2008: -0.1; 2009: 0.2; 2010: -0.5; 2011: -0.1; 2012: 0.3; 2013: -0.1; 2014: 0.0; 2015: 0.9; 2016: -0.5.
The Patent Act requires the PMPRB to consider changes in the CPI, among other factors, in determining whether the price of a patented drug product is excessive. Figure 7 plots year-over-year rates of change in the PMPI against corresponding changes in the CPI. General price inflation, as measured by the CPI, has exceeded the average increase in patented drug prices almost every year since 1988. In 2016, the CPI rose by 1.4%, while the PMPI decreased by -0.5% between 2015 and 2016.
Figure 7. Annual Rate of Change, Patented Medicines Price Index (PMPI) and Consumer Price Index (CPI), 1988-2016
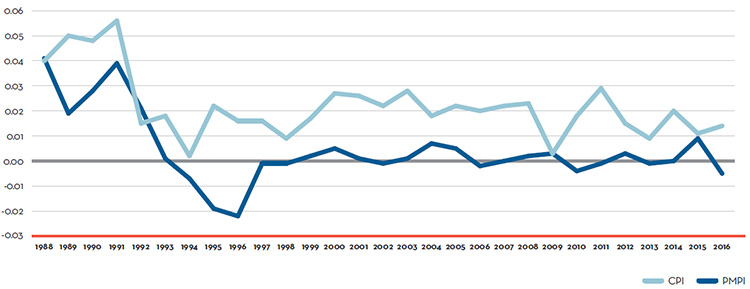
Source: PMPRB, Statistics Canada
Figure description
This is a line graph depicting the year-over-year rates of change in the Patented Medicines Price Index (PMPI) against corresponding changes in the Consumer Price Index (CPI), in percent. In 1988, the rate of change in the PMPI was 0.041. The rate of change in the CPI was 0.04. 1989: PMPI 0.019, CPI 0.05; 1990: PMPI 0.028, CPI 0.048; 1991: PMPI 0.039, CPI 0.056; 1992: PMPI 0.021, CPI 0.015; 1993: PMPI 0.001, CPI 0.018; 1994: PMPI -0.007, CPI 0.002; 1995: PMPI -0.019, CPI 0.022; 1996: PMPI -0.022, CPI 0.016; 1997: PMPI -0.001, CPI 0.016; 1998: PMPI -0.001, CPI 0.009; 1999: PMPI 0.002, CPI 0.017; 2000: PMPI 0.005, CPI 0.027; 2001: PMPI 0.001, CPI, 0.026; 2002: PMPI -0.001, CPI 0.022; 2003: PMPI 0.001, CPI 0.028; 2004: PMPI 0.007, CPI 0.018; 2005: PMPI 0.005, CPI 0.022; 2006: PMPI -0.002, CPI 0.020; 2007: PMPI 0.000, CPI 0.022; 2008: PMPI 0.002, CPI 0.023; 2009: PMPI 0.003, CPI 0.003; 2010: PMPI -0.004, CPI 0.018; 2011: PMPI -0.001, CPI 0.029; 2012: PMPI 0.003, CPI 0.015; 2013: PMPI -0.001, CPI 0.009; 2014: PMPI 0.000, CPI 0.020; 2015: PMPI 0.009, CPI 0.011; 2016: PMPI -0.005, CPI 0.014.
It is not surprising that the PMPI has seldom kept pace with the CPI. The PMPRB’s Guidelines allow the price of a patented drug product to rise by no more than the CPI over any three-year period. (The Guidelines also impose a cap on year-over-year price increases equal to one and one-half times the current year rate of CPI inflation.) This effectively establishes CPI inflation as an upper bound on the amount by which individual prices may rise over any three-year period.Footnote 9 Increases in the PMPI normally do not reach this upper bound because many patentees do not raise their prices by the full amount permitted under the Guidelines.
Price Change by Therapeutic Class
Table 11 provides average rates of price change among patented drug products at the level of major therapeutic classes. Results in this table were obtained by applying the PMPI methodology to data segregated by their ATC Level 1 class. The last column provides a decomposition of overall PMPI change, with each entry representing the component of the overall change attributable to drug products in the corresponding therapeutic class. By this measure, the PMPI (-0.5%) reflects a general state of price stability across therapeutic classes. Note that all of the therapeutic classes saw an average rate of price change below the rate of CPI inflation.Footnote 10
Table 11. Change in the Patented Medicines Price Index (PMPI), by Major Therapeutic Class, 2016
| Therapeutic class |
Share: 2016 Sales (%) |
Price change: 2015 to 2016 (%) |
Contribution: change in PMPI (%) |
| A: Alimentary tract and metabolism |
11.1 |
-1.30 |
-0.14 |
| B: Blood and blood forming organs |
5.7 |
-0.91 |
-0.05 |
| C: Cardiovascular system |
5.4 |
0.36 |
0.02 |
| D: Dermatologicals |
0.8 |
0.83 |
0.01 |
| G: Genito-urinary system and sex hormones |
2.6 |
-3.46 |
-0.09 |
| H: Systemic hormonal preparations |
0.5 |
0.95 |
0.00 |
J: General antiinfectives for systemic use and
P: Antiparasitic products* |
14.4 |
0.49 |
0.07 |
| L: Antineoplastics and immunomodulating agents |
33.2 |
-2.09 |
-0.70 |
| M: Musculo-skeletal system |
2.6 |
0.27 |
0.01 |
| N: Nervous system |
10.0 |
0.46 |
0.05 |
| R: Respiratory system |
8.0 |
-1.61 |
-0.13 |
| S: Sensory organs |
5.2 |
0.39 |
0.02 |
| V: Various |
0.6 |
-1.30 |
-0.01 |
| All therapeutic classes |
100.0† |
-0.5007 |
-0.5007 |
† Values in this column may not add to 100.0 due to rounding.
* These groups have been combined for reasons of confidentiality.
Source: PMPRB
Price Change by Class of Customer
Figure 8 presents average rates of price change by class of customer.Footnote 11 These results were obtained by applying the PMPI methodology separately to sales data for hospital, pharmacy and wholesale customers.Footnote 12 The 2016 rates of price change for these classes were, respectively, -4.7%, 4.3% and -0.3%.
Figure 8. Annual Rate of Change (%), Patented Medicines Price Index (PMPI), by Class of Customer, 2013–2016
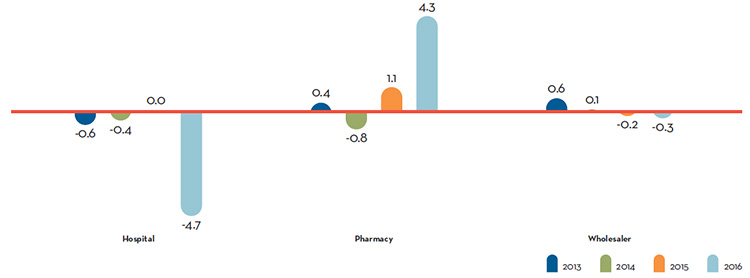
Source: PMPRB
Figure description
This is a bar graph depicting average rates of price change by class of customer, in percent. In 2013, the average rate of price change for hospitals was -0.6. For pharmacies, it was 0.4. For wholesalers, it was 0.6. 2014: Hospital -0.4, Pharmacy -0.8, Wholesaler 0.1; 2015: Hospital 0.0, Pharmacy 1.1, Wholesaler -0.2; 2016: Hospital -4.7, Pharmacy 4.3, Wholesaler -0.3.
Price Change by Province/Territory
Figure 9 presents average annual rates of price change by province/territory, obtained by applying the PMPI methodology to sales data segregated by the province/territory in which the sale occurred. These results indicate that, between 2015 and 2016, the average transaction prices of patented drug products in Quebec and Ontario fell in wholesale and hospital customer classes.
Figure 9. Annual Rate of Price Change, by Province/Territory* and Class of Customer, 2016
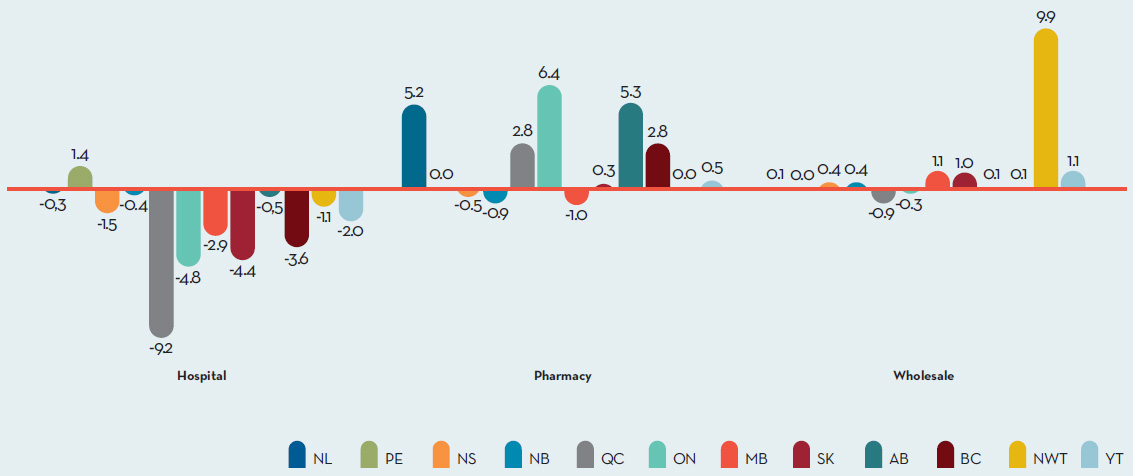
*Values for Nunavut are included in the Northwest Territories (NWT).
Source: PMPRB
Figure description
This is a bar graph depicting average annual rates of price change by province/territory and class of customer in between 2015 and 2016.
For hospitals:
- Newfoundland -0.3
- PEI 1.4
- Nova Scotia -1.5
- New Brunswick -0.4
- Quebec -9.2
- Ontario -4.8
- Manitoba -2.9
- Saskatchewan -4.4
- Alberta -0.5
- British Columbia -3.6
- Northwest Territories -1.1
- Yukon -2.0
For pharmacies:
- Newfoundland 5.2
- PEI 0.0
- Nova Scotia -0.5
- New Brunswick 10.9
- Quebec 2.8
- Ontario 6.4
- Manitoba -1.0
- Saskatchewan 0.3
- Alberta 5.3
- British Columbia 2.8
- Northwest Territories 0.0
- Yukon 0.5
For wholesalers:
- Newfoundland 0.1
- PEI 0.0
- Nova Scotia 0.4
- New Brunswick 0.4
- Quebec -0.9
- Ontario -0.3
- Manitoba 1.1
- Saskatchewan 1.0
- Alberta 0.1
- British Columbia 0.1
- Northwest Territories 9.9
- Yukon Territories 1.1.
Price Behaviour after Introduction
Does the price of a typical patented drug product change much in the years after it enters the Canadian market? To answer this question, Figure 10 provides the average ratio of the 2016 price to introductory price (the price at which the drug product was sold in its first year on the Canadian market).
Figure 10. Average Ratio of 2016 Price to Introductory Price, by Year of Introduction
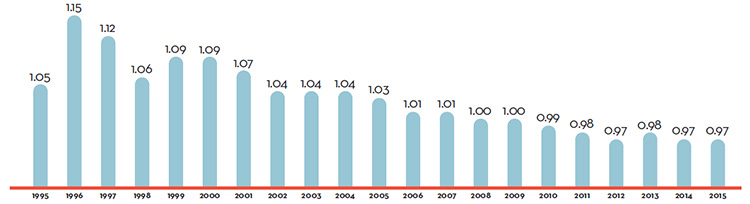
Figure description
This is a bar graph that depicts the average ratio of the 2016 price of a typical patented drug product to the introductory price (the price at which the drug product was sold in its first year on the Canadian market).
In 1995, the average ratio between the 2016 price to the introductory price is 1.05. In 1996, 1.15; 1997, 1.12; 1998, 1.06; 1999, 1.09; 2000, 1.09; 2001, 1.07; 2002, 1.04; 2003, 1.04; 2004, 1.04; 2005, 1.03; 2006, 1.01; 2007, 1.01; 2008, 1.00; 2009, 1.00; 2010, 0.99; 2011, 0.98; 2012, 0.97; 2013, 0.98; 2014, 0.97; 2015, 0.97.
The results in Figure 10 imply a consistent trend for prices to remain stable early in the life cycle, and then to gradually rise by a small amount, year-over-year, afterwards. This is consistent with the effect of the PMPRB’s CPI methodology.Footnote 13 For example; the prices of products introduced a decade ago are only 3% higher in 2016.
Price Change by Country
In accordance with the Act and the Regulations, patentees must report publicly available prices of patented drug products for seven foreign comparator countries (“PMPRB7”): France, Germany, Italy, Sweden, Switzerland, the United Kingdom and the United States.
The PMPRB uses this information to:
- conduct international price comparison tests (as specified in its Guidelines)
- compare the Canadian prices of patented drug products to those prevailing in other countries
Figure 11 gives the average annual rates of price change for Canada and each of the seven comparator countries. These results were obtained by applying the PMPI methodology (with weights based on Canadian sales patterns) to the international price data that patentees have submitted to the PMPRB. Note that results for the United States are based on prices that incorporate prices from the US Federal Supply Schedule (FSS).Footnote 14
Figure 11. Annual Average Rates of Price Change, Canada and Comparator Countries, 2016
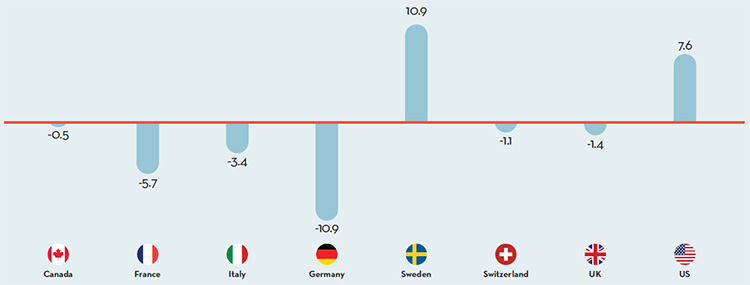
Figure description
This is a bar graph depicting the average annual rates of price change for Canada and each of the seven comparator countries, in percent. In Canada, the average annual rate of price change was -0.5. France, -5.7; Italy, -3.4; Germany, -10.9; Sweden, 10.9; Switzerland, -1.1; UK, -1.4; US, 7.6.
The results in Figure 11 indicate that in 2016, the United States saw prices rise at an average rate of 7.6%. While prices in France, Italy, Switzerland, United Kingdom and Germany declined. These results are consistent with a long-term tendency for patented medicine prices to slowly fall over time in most comparable countries (the exception being the United States).
The foreign market results are based on publicly available ex-factory price information (generally for the retail customer class) submitted by patentees to the PMPRB. The Canadian rate of change, however, is based on the actual average transaction prices and is net of rebates and discounts provided by manufacturers to their direct customers.
Comparison of Canadian Prices to Foreign Prices
Tables 12 and 13 provide detailed statistics comparing the foreign prices of patented drug products to their Canadian prices. Each table provides two sets of average price ratios. These are differentiated according to the method by which foreign prices were converted to their Canadian dollar equivalents. The tables also give the numbers of drug products (DINs) and the volume of sales encompassed by each reported price ratio.Footnote 15
The average price ratios given in Tables 12 and 13 are salesweighted arithmetic means of price ratios obtained for individual drug products, with weights based on Canadian sales patterns. Average price ratios constructed in this way provide exact answers to questions of the following type:
How much more/less would Canadians have paid for
the patented drug products they purchased in 2016 had
they paid Country X prices rather than Canadian prices?
For example, Table 12 states that the 2016 average France-to-Canada price ratio was 0.77. This means Canadians would have paid 23% less for the patented drug products they purchased in 2016 had they bought these products at French prices.
For many years, the PMPRB has reported average foreign-to-Canadian price ratios with foreign prices converted to their Canadian dollar equivalents by means of market exchange rates. (More exactly, the 36-month moving averages of market rates the PMPRB normally uses in applying its Guidelines.) Table 12 also reports foreign-to-Canadian price ratios with currency conversion at purchasing power parity (PPP). The PPP between any two countries measures their relative costs of living expressed in units of their own currencies. In practice, cost of living is determined by pricing out a standard “basket” of goods and services at the prices prevailing in each country.
Because PPPs are designed to represent relative costs of living, they offer a simple way to account for differences in overall national price levels when comparing individual prices, incomes and other monetary values across countries. When applied to the calculation of average foreign-to-Canadian price ratios they produce statistics answering questions of this type:
How much more/less consumption of other goods
and services would Canadians have sacrificed for the
patented drug products they purchased in 2016 had
they lived in Country X?
Questions of this type cannot be answered by simply comparing drug prices. Rather, one must first calculate what each price represents in terms of goods and services foregone. PPPs are designed for such purposes.
Bilateral Comparisons
Table 12 provides bilateral comparisons of prices in each of the PMPRB’s seven comparator countries to corresponding Canadian prices. Focusing on the results with currency conversion at market exchange rates, it appears that, as in previous years, Canadian prices were typically within the range of prices observed among the comparator countries. Prices in France, Italy, the United Kingdom and Sweden were appreciably lower than Canadian prices, while those in Germany and Switzerland were higher. As in previous years, prices reported for the United States were much higher than prices in Canada or any other comparator country.
Table 12. Average Foreign-to-Canadian Price Ratios, Bilateral Comparisons, 2016
At Market Exchange Rates
|
Canada |
France |
Italy |
Germany |
Sweden |
Switzerland |
United Kingdom |
United States |
| Average price ratio 2016 |
1.00 |
0.77 |
0.92 |
1.09 |
0.95 |
1.09 |
0.99 |
3.08 |
| Average price ratio 2015 |
1.00 |
0.75 |
0.87 |
1.16 |
0.94 |
1.00 |
0.92 |
2.70 |
At Purchasing Power Parities
|
Canada |
France |
Italy |
Germany |
Sweden |
Switzerland |
United Kingdom |
United States |
| Average price ratio 2016 |
1.00 |
0.83 |
1.09 |
1.22 |
0.84 |
0.87 |
0.97 |
3.15 |
| Average price ratio 2015 |
1.00 |
0.80 |
0.97 |
1.27 |
0.81 |
0.79 |
0.92 |
2.95 |
| Number of patented drug products 2016 |
1,419 |
742 |
888 |
1,011 |
853 |
887 |
973 |
1,119 |
| Sales ($millions) |
15,496.01 |
9,399.96 |
12,528.09 |
13,381.66 |
12,163.97 |
12,961.69 |
13,141.21 |
14,228.95 |
Source: PMPRB
It is important to note that it is not always possible to find a matching foreign price for each and every patented drug product sold in Canada. Table 12 displays how often an international price comparison was available for each of the comparator countries. For example, out of 1,419 patented drug products under the PMPRB’s jurisdiction in 2016, a publicly available ex-factory price for France was available 52.3% of the time, whereas for the US the number was 78.9%. Given the integrated nature of the Canadian and US supply chain, it is not uncommon for the US to be the only other country for which a comparator price to a product sold in Canada is available, in which case it is deemed to constitute the international median price as per the PMPRB’s methodology.
Average price ratios obtained with currency conversion at PPPs tell the same story. When international differences in cost of living are accounted for, it appears Canadians incurred a larger consumption cost for the patented drug products they purchased in 2016 than did residents of every other comparator country except Germany, Italy and the United States.
Figure 12 puts these results in historical perspective. In 2005, Canadian prices were, on average, approximately equal to or below corresponding prices in all comparators other than Italy. By 2016, Canadian prices were decidedly above prices in the United Kingdom, France, Italy and Sweden.
Figure 12. Average Foreign-to-Canadian Price Ratios, 2005, 2016
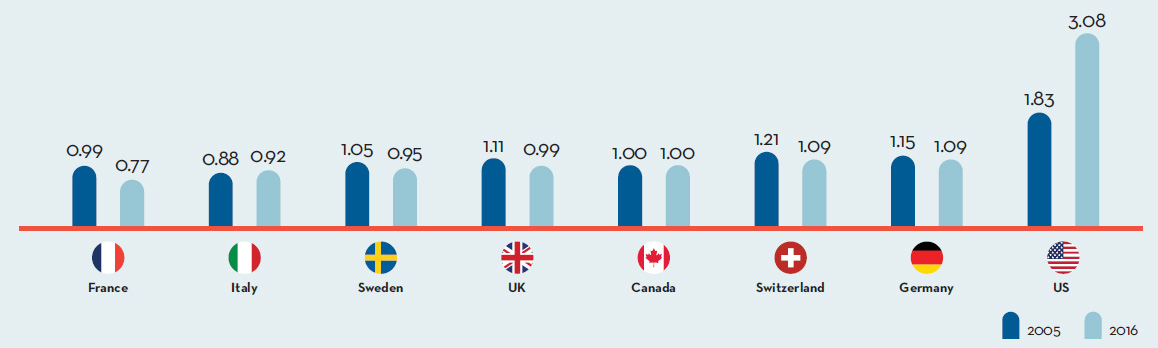
Figure description
This is a bar graph depicting the average foreign-to-Canadian price ratios in 2005 and 2016.
- France — 2005: 0.99, 2016: 0.77
- Italy — 2005: 0.88, 2016: 0.92
- Sweden — 2005: 1.05, 2016: 0.95
- United Kingdom — 2005: 1.11, 2016: 0.99
- Canada — 2005: 1.00, 2016: 1.00
- Switzerland — 2005: 1.21, 2016: 1.09
- Germany — 2005: 1.15, 2016: 1.09
- United States — 2005: 1.83, 2016: 3.08
If the patented medicine is being sold in one or more of the comparator countries (PMPRB7), the patentee must report the publicly available ex-factory prices to the PMPRB for each class of customer.Footnote 16 In order to assess how Canada compares to a basket of countries beyond the PMPRB7, Figure 13 uses Canadian and international prices reported in the IMS AG MIDAS™ database at the ex-factory manufacturer level, reflecting all sales to the pharmacy and hospital sectors.
The international price comparisons reported in Figure 13 provide a bilateral price comparison using all countries in the Organisation for Economic Co-operation and Development (OECD) available in IMS AG’s MIDAS™. The average foreign-to-Canadian price ratios are constructed using exactly the same approach employed to produce the ratios presented in Figure 12. These are Canadian sales-weighted arithmetic averages of the corresponding foreign-to-Canadian price ratios for individual drugs.Footnote 17 As shown below, median OECD prices are on average approximately 20% below prices in Canada, which are third highest among the 31 countries and on par with Germany. Notably, the top three priced countries are now the US, Switzerland and Canada.
Figure 13. Average Foreign-to-Canadian Price Ratios, Patented Drugs, OECD, 2016
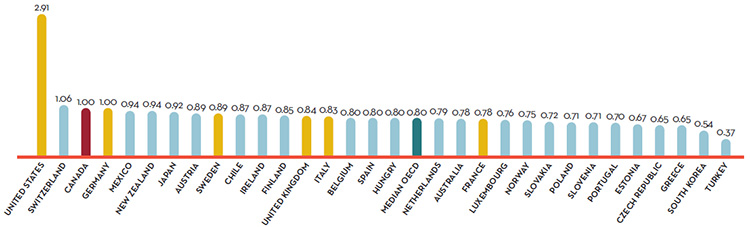
Source: MIDAS™ database, 2005-2016, IMS AG. All rights reserved.
Figure description
This is a bar graph depicting the average foreign-to-Canadian price ratios of patented drugs in 2016 for OECD countries, using Canadian and international prices reported in the IMS AG MIDAS™ database. United States: 2.91; Switzerland: 1.06; Canada: 1.00; Germany: 1.00; Mexico: 0.94; New Zealand: 0.94; Japan: 0.92; Austria: 0.89; Sweden: 0.89; Chile: 0.87; Ireland: 0.87; Finland: 0.85; United Kingdom: 0.84; Italy: 0.83; Belgium: 0.80; Spain: 0.80; Hungary: 0.80; Median for the OECD: 0.80; Netherlands: 0.79; Australia: 0.78; France: 0.78; Luxembourg: 0.76; Norway: 0.75; Slovakia: 0.72; Poland: 0.71; Slovenia: 0.71; Portugal: 0.70; Estonia: 0.67; Czech Republic: 0.65; Greece: 0.65; South Korea: 0.54; Turkey: 0.37.
Multilateral Price Comparisons
Table 13 provides average foreign-to-Canadian price ratios using several multilateral measures of foreign prices. The median international price (MIP) is the median of prices observed among the seven comparator countries. Other multilateral price ratios compare the minimum, maximum and simple mean of foreign prices to their Canadian counterparts.
Table 13. Average Foreign-to-Canadian Price Ratios, Multilateral Comparisons, 2016
|
Median |
Minimum |
Maximum |
Mean |
| Average price ratio at market exchange rates |
1.25 |
0.99 |
3.01 |
1.49 |
| Average price ratio at purchasing power parities |
1.24 |
0.98 |
3.09 |
1.51 |
| Number of patented drug products |
1,313 |
1,313 |
1,313 |
1,313 |
| Sales ($millions) |
15,085.18 |
15,085.18 |
15,085.18 |
15,085.18 |
Source: PMPRB
Focusing again on results at market exchange rates, the average MIP-to-Canadian price ratio stood at 1.25 in 2016. (The corresponding value for 2015 was 1.18.) Note that mean foreign prices produce higher foreign-to-Canadian price ratios than do MIPs. This is explained by the influence of US prices, which are typically much higher than prices elsewhere. Although US prices nearly always figure importantly in determining mean foreign price, this is less so when it comes to median international prices. Nevertheless, the US does exercise a significant influence over the average ratio of median international prices relative to Canadian prices because of the not infrequent phenomenon mentioned in the previous section, whereby the US is the only country for which an ex-factory price for a patented drug product sold in Canada is available.
Figure 14 puts these results in historical perspective, giving a history of the average MIP-to-Canadian price ratios from 2001 to 2016. Although there has been considerable movement in the ratio over this period, it has remained above parity.
Figure 14. Average Ratio of Median International Price (MIP) to Canadian Price, at Market Exchange Rates, 2001–2016
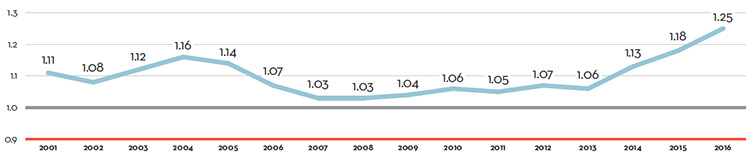
Figure description
This is a line graph depicting the history of the average MIP-to-Canadian price ratios from 2001 to 2016. In 2001, the average ratio of Median International Price (MIP) to Canadian average transaction price, at market exchange rates, was 1.11. In 2002: 1.08; 2003: 1.12; 2004: 1.16; 2005: 1.14; 2006: 1.07; 2007: 1.03; 2008: 1.03; 2009: 1.04; 2010: 1.06; 2011: 1.05; 2012: 1.07; 2013: 1.06; 2014: 1.13; 2015: 1.18; 2016: 1.25.
Figure 15 provides alternate results for the average MIP-to-Canadian price ratio at market exchange rates in 2016. To address the point that Canadian prices are national average transaction prices whereas foreign prices are list prices, a list price to list price ratio is also calculated. Using this method, the average ratio decreases from 1.25 to 1.09. It is important to keep in mind that non-transparent rebates provided to payers are currently not captured in these data.
To account for the large impact of US prices in determining the median foreign price, a ratio excluding the US and a ratio including at least five countries in the calculation of the median are also provided in Figure 15. With these restrictions, the average MIP-to-Canadian price ratio drops to 0.89 and 0.93, respectively, suggesting that Canadian list prices are on average 7%-11% higher than median foreign list prices. In many of the comparator countries, discounts off list prices are available to all payers, both public and private. By contrast, a large portion of the Canadian market pay list prices or close to list prices. Furthermore, it should be noted that these are average ratios—some patentees charge Canadian consumers less than median international prices, while others charge more. For patentee level median-to-Canadian price ratios, please refer to Table 24 in Appendix 3 of this report.
Figure 15. Average Ratio of Median International Price (MIP) to Canadian Price, at Market Exchange Rates, 2016
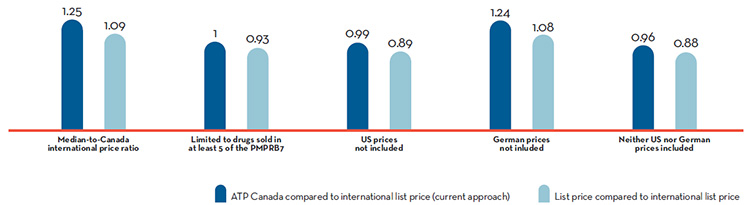
Figure description
This is a bar graph depicting the average ratio of Median International Price (MIP) to Canadian price at market exchange rates, using data from 2016.
|
Canada at N-ATP |
Canada at List Price |
| International median for PMPRB7 |
1.25 |
1.09 |
| Median limited to drugs sold in at least 5 of the PMPRB7 |
1.00 |
0.93 |
| Without US |
0.99 |
0.89 |
| Without Germany |
1.24 |
1.08 |
| Without Germany and US |
0.96 |
0.88 |
Figure 16 offers more detail on the product-level MIP-to-Canadian ratios underlying the averages reported in Table 13. This figure distributes the 2016 sales of each patented drug product according to the value of its MIP-to-Canadian price ratio (more exactly, according to the range into which the ratio fell).Footnote 18 These results show substantial dispersion in product-level price ratios: while patented drug products with MIP-to-Canadian price ratios between 0.90 and 1.10 accounted for 37.3% of sales, those with ratios less than 0.90 accounted for 29.2% of sales, and products with ratios exceeding 1.10 accounted for 33.6%.
In 2016, approximately 50% of Canadian patented drug products were priced above the median international level.Footnote 19 Table 14 shows which therapeutic categories in particular are priced above the median international levels in Canada. Drugs that share the fourth level ATC (“ATC4”)Footnote 20 are grouped to identify distinct chemical/pharmacological/ therapeutic subgroups, allowing for a calculation of the average MIP-to-Canadian price ratios among drugs that may be used to treat the same conditions. Table 14 identifies the top 10 ATC4s in 2016 in which the difference between Canadian and median prices had the largest effect on Canadian patented medicine spending. For example, had Canadian prices been in line with the international median for these classes of drugs in 2016, sales in Canada would have been reduced by $794 million (an average reduction of 16% for these ATC4s). Of the 123 DINs classified into these 10 ATC4s, over 68% were priced above the median international price.
Figure 16. Range Distribution, Sales, by MIP-to-Canadian Price Ratio, 2016
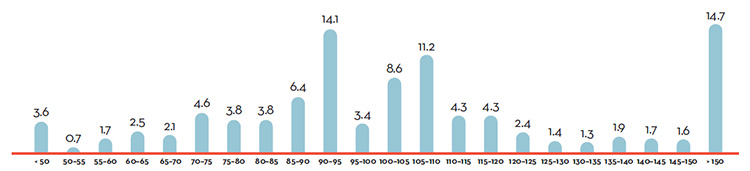
Figure description
This is a bar graph depicting the distribution of 2016 sales of each patented drug product according to the value of its MIP-to-Canadian price ratio (more exactly, according to the range into which the ratio fell).
‹ 0.50: 3.6; 0.50 to 0.55: 0.7; 0.55 to 0.60: 1.7; 0.60 to 0.65: 2.5; 0.65 to 0.70: 2.1; 0.70 to 0.75: 4.6; 0.75 to 0.80: 3.8; 0.80 to 0.85: 3.8; 0.85 to 0.90: 6.4; 0.90 to 0.95: 14.1; 0.95 to 1.00: 3.4; 1.00 to 1.05: 8.6; 1.05 to 1.10: 11.2; 1.10 to 1.15: 4.3; 1.15 to 1.20: 4.3; 1.20 to 1.25: 2.4; 1.25 to 1.30: 1.4; 1.30 to 1.35: 1.3; 1.35 to 1.40: 1.9; 1.40 to 1.45: 1.7; 1.45 to 1.50: 1.6; › 1.50: 14.7.
Table 14. Top-10 ATC4s by Total Revenues Greater than Median International Prices, 2016
| Description |
ATC4 |
# of companies |
Total # of Chemicals in ATC4 (# currently under patent)Footnote 21 |
Total Patented DINS |
Patented DINS greater than median price |
2016 Net Revenue for Patented DINS ($) |
Patented DINS ATC4 Share of 2016 Revenues |
MIP-to-Canada ratio (min 5) of Patented DINS |
$ Impact of Difference on patented drugs in 2016 ($) |
| Adrenergics in combination with corticosteroids or other drugs excluding anticholinergics |
R03AK |
3 |
5 (5) |
11 |
9 |
$543,451,835.08 |
3.50% |
61% |
$199,055,072.31 |
| Antineovascularisation agents |
S01LA |
2 |
2 (2) |
3 |
3 |
$593,565,286.65 |
3.80% |
84% |
$128,113,966.66 |
| Other antidepressants |
N06AX |
7 |
7 (6) |
11 |
5 |
$234,397,100.29 |
1.50% |
50% |
$78,362,223.95 |
| Tumor necrosis factor alpha inhibitors |
L04AB |
3 |
4 (3) |
7 |
1 |
$1,074,437,530.77 |
6.90% |
88% |
$70,207,891.05 |
| DPP-4 inhibitors |
A10BH |
4 |
4 (4) |
9 |
9 |
$267,114,372.22 |
1.70% |
73% |
$63,293,437.50 |
| Glucocorticoids |
R03BA |
9 |
10 (6) |
15 |
10 |
$182,585,300.05 |
1.20% |
82% |
$54,323,337.35 |
| Combinations of oral blood glucose lowering drugs |
A10BD |
6 |
13 (13) |
31 |
19 |
$242,486,725.25 |
1.50% |
68% |
$52,858,775.75 |
| Selective immunosuppressants |
L04AA |
18 |
22 (16) |
28 |
21 |
$1,408,148,492.13 |
9.10% |
99% |
$49,815,389.90 |
| Colony stimulating factors |
L03AA |
3 |
4 (4) |
4 |
3 |
$188,089,000.34 |
1.20% |
64% |
$49,495,790.86 |
| Insulins and analogues for injection, long-acting |
A10AE |
3 |
4 (2) |
4 |
4 |
$256,522,336.48 |
1.70% |
79% |
$48,036,062.66 |
Source: PMPRB
Utilization of Patented Drug Products
The price and sales data used to calculate the PMPI also allow the PMPRB to examine trends in the quantities of patented drug products sold in Canada. The PMPRB maintains the Patented Medicines Quantity Index (PMQI) for this purpose. Figure 17 provides average rates of utilization growth, as measured by the PMQI, from 1988 through 2016. These results confirm that in recent years, growth in the utilization of patented drug products has been the primary source of rising sales, with rates of utilization growth roughly tracking sales growth. This tracking pattern continued in 2016, with utilization of patented drug products, on average, increasing by 8.7% between 2015 and 2016 and sales increasing by 2.6%.
Figure 17. Annual Rate of Change (%), Patented Medicines Quantity Index (PMQI), 1988-2016
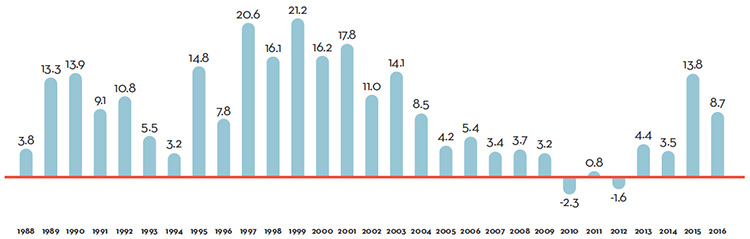
Figure description
This is a bar graph depicting average rates of utilization growth, as measured by the Patented Medicines Quantity Index (PMQI), from 1988 through 2016, in percent.
1988: 3.8; 1989: 13.3; 1990: 13.9; 1991: 9.1; 1992: 10.8; 1993: 5.5; 1994: 3.2; 1995: 14.8; 1996: 7.8; 1997: 20.6; 1998: 16.1; 1999: 21.2; 2000: 16.2; 2001: 17.8; 2002: 11.0; 2003: 14.1; 2004: 8.5; 2005: 4.2; 2006: 5.4; 2007: 3.4; 2008: 3.7; 2009: 3.2; 2010: -2.3; 2011: 0.8; 2012: -1.6; 2013: 4.4; 2014: 3.5; 2015: 13.8; 2016: 8.7.
Utilization Growth by Therapeutic Class
Table 15 provides average rates of utilization growth among patented drug products at the level of major therapeutic classes. The results in this table were obtained by applying the PMQI methodology to data segregated by ATC Level I class. As in Table 11, the last column provides an approximate decomposition of overall PMQI change into contributions attributable to each therapeutic class.
In 2016, levels of utilization increased in ten therapeutic classes. Increased consumption of various, blood and blood forming organs, systemic hormonal preparations, and antineoplastics and immunomodulating agents accounted for most of the growth in overall utilization.
Table 15. Change in the Patented Medicines Quantity Index (PMQI), by Major Therapeutic Class, 2016
| Therapeutic class |
Share: 2016 sales (%) |
Quantity change: 2015-2016 (%) |
Contribution: change in PMQI (%) |
| A: Alimentary tract and metabolism |
11.1 |
7.44 |
0.83 |
| B: Blood and blood forming organs |
5.7 |
15.56 |
0.89 |
| C: Cardiovascular system |
5.4 |
4.77 |
0.26 |
| D: Dermatologicals |
0.8 |
-2.36 |
-0.02 |
| G: Genito-urinary system and sex hormones |
2.6 |
-6.46 |
-0.17 |
| H: Systemic hormonal preparations |
0.5 |
12.48 |
0.06 |
J: General antiinfectives for systemic use and
P: Antiparasitic products* |
14.4 |
6.81 |
0.98 |
| L: Antineoplastics and immunomodulating agents |
33.2 |
12.36 |
4.10 |
| M: Musculo-skeletal system |
2.6 |
8.35 |
0.22 |
| N: Nervous system |
10.0 |
-2.24 |
-0.22 |
| R: Respiratory system |
8.0 |
2.71 |
0.22 |
| S: Sensory organs |
5.2 |
2.71 |
0.14 |
| V: Various |
0.6 |
16.80 |
0.10 |
| All therapeutic classes |
100.0† |
8.7 |
8.7 |
† Values in this column may not add to 100.0 due to rounding.
* These groups have been combined for reasons of confidentiality.
Source: PMPRB
Canadian Drug Expenditures in the Global Context
IMS HealthFootnote 22 regularly reports on drug sales across a large number of countries. Based on sales data from this source, Figure 18 provides shares of global sales for Canada and each of the seven comparator countries that the PMPRB considers in conducting its price reviews (PMPRB7).Footnote 23 The Canadian market accounted for 1.9% of the global market in 2016.
Figure 18. Distribution of Drug Sales Among Major National Markets, 2016
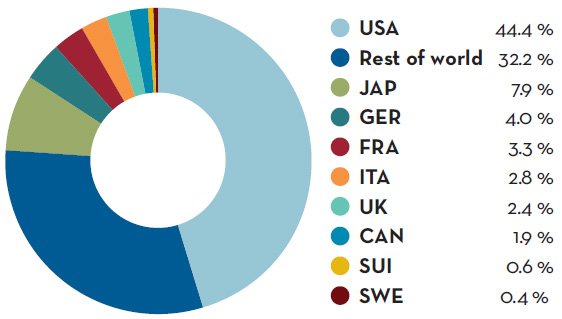
Source: Midas™ database, 2005-2016, IMS AG. All rights reserved.Footnote 24
Figure description
This is a pie chart depicting the distribution of drug sales among major global markets in 2016.
The United States market accounted for 44.4% of the global market in 2016. Japan: 7.9%; Germany: 4.0%; France: 3.3%; Italy: 2.8%; U.K.: 2.4%; Canada: 1.9%; Switzerland: 0.6%; Sweden: 0.4%; the rest of the world: 32.2%.
Figure 19 provides Canada’s share of global sales for 2005 to 2016. The Canadian share has remained between 1.9% and 2.7% throughout this period. Though 1.9% is at the low end for Canada’s average share of global sales in recent years, and marks the sixth year in a row that Canada’s share has fallen, this trend is driven by rapid price increases in the United States, which grew the US share from 40.4% in 2014 to 44.4% in 2016, resulting in declining shares for all other major countries.
Figure 19. Canada’s Share of Drug Sales, 2005–2016
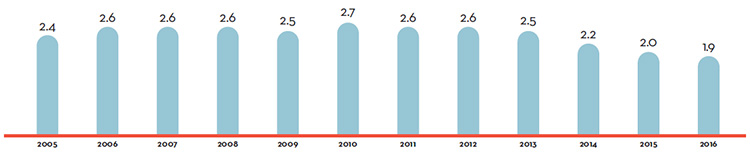
Source: Midas™ database, 2005-2016, IMS AG. All rights reserved.Footnote 26
Figure description
This is a bar graph depicting Canada’s share of global sales for 2005 to 2016, in percent.
2005: 2.4; 2006: 2.6; 2007: 2.6; 2008: 2.6; 2009: 2.5; 2010: 2.7; 2011: 2.6; 2012: 2.6; 2013: 2.5; 2014: 2.2; 2015: 2.0; 2016:1.9.
Figure 20 gives the average annual rate of growth in total drug sales for Canada and the seven comparator countries, individually and collectively (PMPRB7). From 2005 to 2016, drug sales in Canada rose at an average annual rate of approximately 4.1%. This is less than the average rate of growth in drug sales among the seven comparator countries over the same period, though as is clear from the figure, this growth rate is heavily skewed by the influence of US sales on the total revenues of the PMPRB7.
Figure 20. Average Rate of Growth (%), Drug Sales, at Constant 2016 Market Exchange Rates, by Country, 2005–2016
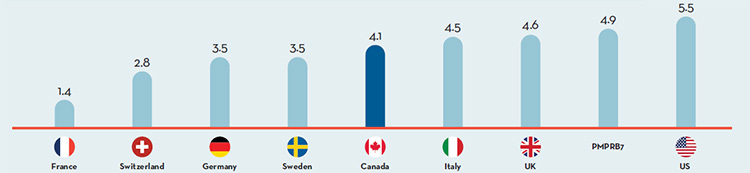
Source: Midas™ database, 2005-2016, IMS AG. All rights reserved.Footnote 27
Figure description
This is a bar graph depicting the annual rate of growth in total drug sales, in percent, for Canada and the seven comparator countries, individually and collectively (PMPRB7), between 2005 and 2016.
France: 1.4; Switzerland: 2.8; Germany: 3.5; Sweden: 3.5; Canada: 4.1; Italy: 4.5; UK: 4.6; PMPRB7: 4.9; US: 5.5.
Figure 21 compares rates of year-over-year growth in drug sales in Canada and the comparator countries combined (PMPRB7). In 2016, for the seventh consecutive year, sales grew at a slower rate in Canada than the PMPRB7 total. As identified in the discussion of Figures 14 and 15, however, the presence of the US skews these results. Accordingly, the median of the PMPRB7 expenditure growth rate has been added to this figure, showing that Canadian expenditure growth rate has tracked the PMPRB7 expenditure growth rate quite closely since 2010.
Figure 21. Average Annual Rate of Change in Drug Sales, at Constant 2016 Market Exchange Rates, Canada and Comparator Countries, 2006–2016
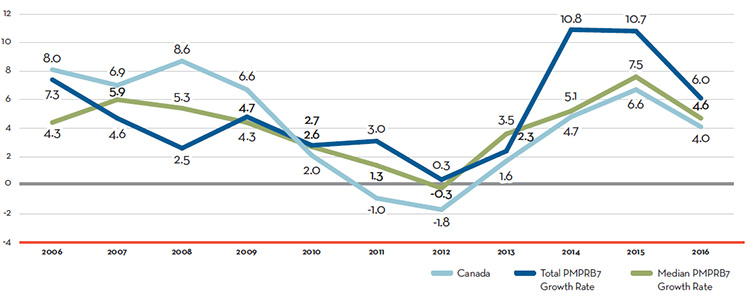
Source: Midas™ database, 2005-2016, IMS AG. All rights reserved.Footnote 28
Figure description
Figure 21 is a line graph comparing the average annual rate of change in drug sales, at constant 2016 market exchange rates, between Canada, the comparator countries combined (PMPRB7) and the median of the PMPRB7 expenditure growth rate, between 2006 and 2016.
2006: Canada 8.0, Comparators 7.3, Median of Comparators 4.3;
2007: Canada 6.9, Comparators 4.6, Median of Comparators 5.9;
2008: Canada 8.6, Comparators 2.5, Median of Comparators 5.3;
2009: Canada 6.6, Comparators 4.7, Median of Comparators 4.3;
2010: Canada 2.0, Comparators 2.7, Median of Comparators 2.6;
2011: Canada -1.0, Comparators 3.0, Median of Comparators 1.3;
2012: Canada -1.8, Comparators 0.3, Median of Comparators -0.3;
2013: Canada 1.6, Comparators 2.3, Median of Comparators 3.5;
2014: Canada 4.7, Comparators 10.8, Median of Comparators 5.1;
2015: Canada 6.6, Comparators 10.7, Median of Comparators 7.5;
2016: Canada 4.0, Comparators 6.0, Median of Comparators 4.6.
The proportion of national income allocated to the purchase of drug products provides another way to compare drug costs across countries.Footnote 25 Figure 22 gives drug expenditures as a share of Gross Domestic Product (GDP) for Canada and the seven comparator countries based on data for 2014. Drug expenditures absorbed between 1.0% and 2.0% of the GDP in the seven comparators. The Canadian value (1.7%) lies near the upper end of this range.
Figure 22. Drug Expenditures as a Share of GDP, 2014
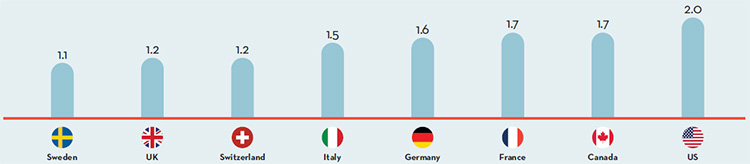
Figure description
This is a bar graph depicting drug expenditures as a share of Gross Domestic Product (GDP), in percent, for Canada and the seven comparator countries based on data for 2014.
Sweden: 1.1; United Kingdom: 1.2; Switzerland: 1.2; Italy: 1.5; Germany: 1.6; France: 1.7; Canada: 1.7; United States: 2.0.
Table 16 provides a historical perspective on the expenditures-to-GDP ratio. In 2005, Canada’s ratio was fourth highest of the PMPRB7. Since that time, Canada’s ratio has risen, while the ratios of three other countries (France, Italy and Sweden) have declined. In 2014, Canada once again had the second highest drug spending per capita among the PMPRB7 (again behind only the United States), 18% higher than the median of these countries.
Table 16. Drug Expenditures as a Share of GDP, 2014Footnote 30
|
Share: Drug Expenditures/GDP 2014 (%) |
Share: Drug Expenditures/GDP 2005 (%) |
Growth: GDP 2005-2014 (%) |
Drug Spending Per Capita 2005
($US PPP) |
Drug Spending Per Capita 2014
($US PPP) |
| Canada |
1.72 |
1.64 |
37.8 |
593 |
772 |
| France |
1.67 |
1.79 |
38.5 |
545 |
656 |
| Germany |
1.60 |
1.58 |
43.6 |
509 |
741 |
| Italy |
1.55 |
1.70 |
28.3 |
505 |
544 |
| Sweden |
1.08 |
1.15 |
45.2 |
396 |
489 |
| Switzerland |
1.23 |
1.09 |
72.1 |
427 |
730 |
| United Kingdom |
1.21 |
1.00 |
25.8 |
NA |
485 |
| United States |
2.04 |
1.88 |
32.8 |
832 |
1,112 |
Source: OECD
Table 17 gives the composition of patentees’ sales by therapeutic class for Canada and the seven comparator countries, individually and as an aggregate (PMPRB7).Footnote 29 The results imply a remarkable degree of similarity across countries.
Table 17. Distribution of Drug Sales (%) by Major Therapeutic Class for Canada and Comparator Countries, 2016
| Therapeutic Class |
Canada |
PMPRB7 |
France |
Italy |
Germany |
Sweden |
Switzerland |
United Kingdom |
United States |
| A: Alimentary tract and metabolism |
12.9 |
14.6 |
9.9 |
9.9 |
10.9 |
9.7 |
10.9 |
10.8 |
15.8 |
| B: Blood and blood-forming organs |
4.5 |
5.2 |
7.8 |
8.2 |
7.3 |
8.6 |
5.7 |
5.5 |
4.6 |
| C: Cardiovascular system |
9.6 |
6.8 |
8.6 |
10.1 |
7.3 |
4.5 |
9.6 |
6.6 |
6.5 |
| D: Dermatologicals |
2.7 |
2.7 |
2.2 |
1.7 |
2.7 |
2.5 |
3.3 |
2.6 |
2.8 |
| G: Genito-urinary system and sex hormones |
4.5 |
4.2 |
2.9 |
3.4 |
3.0 |
3.9 |
4.2 |
3.6 |
4.5 |
| H: Systemic hormonal preparations |
1.3 |
2.4 |
2.2 |
1.8 |
2.0 |
2.3 |
1.6 |
3.0 |
2.5 |
| J: General antiinfectives for systemic use |
9.4 |
12.9 |
13.4 |
21.3 |
10.8 |
12.2 |
11.5 |
11.7 |
12.6 |
| L: Antineoplastics and immunomodulating agents |
19.5 |
20.4 |
22.1 |
17.7 |
23.8 |
23.5 |
21.5 |
20.4 |
20.1 |
| M: Musculo-skeletal system |
3.2 |
3.2 |
2.9 |
3.2 |
3.9 |
3.7 |
5.5 |
2.5 |
3.2 |
| N: Nervous system |
17.3 |
14.9 |
13.2 |
10.7 |
13.1 |
16.3 |
15.5 |
16.8 |
15.3 |
| P: Antiparasitic products |
0.2 |
0.2 |
0.2 |
0.0 |
0.2 |
0.1 |
0.1 |
0.1 |
0.2 |
| R: Respiratory system |
7.2 |
6.7 |
6.0 |
5.0 |
6.7 |
6.9 |
5.9 |
8.5 |
6.8 |
| S: Sensory organs |
4.2 |
2.5 |
3.3 |
2.0 |
3.1 |
3.1 |
4.1 |
4.4 |
2.3 |
| V: Various |
3.6 |
3.2 |
5.3 |
5.0 |
5.3 |
2.7 |
0.7 |
3.6 |
2.8 |
| All therapeutic classes† |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
† Values may not add to 100.0 due to rounding.
Source: MIDAS™, 2005–2016, IMS AG. All rights reserved.Footnote 31
The National Prescription Drug Utilization Information System: Supporting Health-Care Decision Making in Canada
How medications are used—where, by whom and for what—has an impact on the amount that we spend on drugs. The PMPRB contributes to Canada’s understanding of drug usage through the National Prescription Drug Utilization Information System (NPDUIS) initiative, generating comprehensive, accurate information to help guide decision making and support continued sustainability of our pharmaceutical system.
Background
The National Prescription Drug Utilization Information System (NPDUIS) is a research initiative established by federal, provincial, and territorial Ministers of Health in September 2001. It is a partnership between the PMPRB and the Canadian Institute for Health Information (CIHI). The PMPRB conducts critical analyses of price, utilization and cost trends for patented and non-patented prescription drugs under the NPDUIS at the request of the Minister of Health pursuant to section 90 of the Patent Act.
Its purpose is to provide policy makers and public drug plan managers with critical analyses of price, utilization and cost trends, so that Canada’s health care systems has more comprehensive and accurate information on how prescription drugs are being used and on sources of cost increases.
The NPDUIS Advisory Committee, composed of representatives from public drug plans in British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, New Brunswick, Nova Scotia, Prince Edward Island, Newfoundland and Labrador, Yukon, and Health Canada, advises the PMPRB on its research agenda and on individual studies. The Committee also includes observers from CIHI, the Canadian Agency for Drugs and Technologies in Health (CADTH), the Ministère de la Santé et des Services sociaux Québec, and the pan-Canadian Pharmaceutical Alliance (pCPA) Office.
Highlights
Since the last Annual Report, through the NPDUIS initiative, the PMPRB has released three analytical reports and six posters.
Published Reports:
- Market Intelligence Report: Biologic Response Modifier Agents, 2015 (October 2016)
- Meds Entry Watch, 2015 (April 2017)
- CompassRx: Annual Public Drug Plan Expenditure Report, 3rd Edition (May 2017)
Poster Presentations:
- The Canadian Market for Biologic Response Modifier Agents, 2015
- Drug Cost Drivers in Canadian Public Drug Plans, 2015/16
- Generic Drugs in Canada, 2015
- A Review of Public Coverage of CDR Reviewed Drugs
- A Review of Public and Private Coverage of iJODR/pCODR Reviewed Drugs
- Potential Savings from Biosimilars in Canada
In addition, the NPDUIS conducted a number of ad-hoc studies at the request of the NPDUIS participating jurisdictions.
The PMPRB continued to support and strengthen its NPDUIS engagement activities by regularly consulting with the NPDUIS Advisory Committee, participating in conferences and stakeholder committees, hosting information exchange sessions with researchers, and organizing information sessions with interested stakeholders to share the results of the analytical studies.
Synopses of the three most recent reports are provided in this report.
Market Intelligence Report: Biologic Response Modifier Agents, 2015
The recently launched NPDUIS Market Intelligence Report analytical series provides drug pricing and utilization information on specific therapeutic market segments of importance to Canadians.
The first report in this series explores the market impact of biologic disease-modifying antirheumatic drugs (DMARDs), which are used in the treatment of chronic inflammatory diseases such as rheumatoid arthritis Crohn’s disease, ulcerative colitis and psoriasis. The report explores the utilization, market shares, pricing, and treatment costs from a national and international perspective, with a retrospective look at recent trends. It also describes the drug portfolio of the manufacturers operating in this space and identifies opportunities for potential cost savings based on international and domestic market trends.
Key Findings
- Biologic DMARDs accounted for 10.3% of the Canadian pharmaceutical market in 2015. This market share was higher than in almost all the PMPRB7 countries.
- The growth in Canadian sales of biologic anti-inflammatory drugs has nearly doubled since 2010, reaching $2.2 billion in 2015.
- The top-selling biologic, Remicade, accounted for nearly 40% of the Canadian market for biologic DMARDs; the market share was much lower in the PMPRB7 countries, ranging from 12% to 23% in 2015, with a median list price 25% less than in Canada. This price difference translates into $224 million in drug sales or 1.0% of the entire Canadian pharmaceutical market.
- While the price of Inflectra, a biosimilar of Remicade, in Canada is in line with the average international level, the uptake in sales in 2015 was relatively modest. If the use of the biosimilar in Canada had mirrored the median use in the OECD (Organisation for Economic Co-operation and Development) countries (10.1%), it would have translated into a $41.7 million reduction in drug expenditures in 2015.
- There has been a growing gap between Canadian and foreign prices for biologic DMARDs, as Canadian prices have been slowly increasing at a rate lower than the rate of inflation, while the prices in the PMPRB7 countries (except in the US market) have been flat or declining.
Trends in prices for biologic DMARDs
Canada versus PMPRB7*, 2011 to 2015
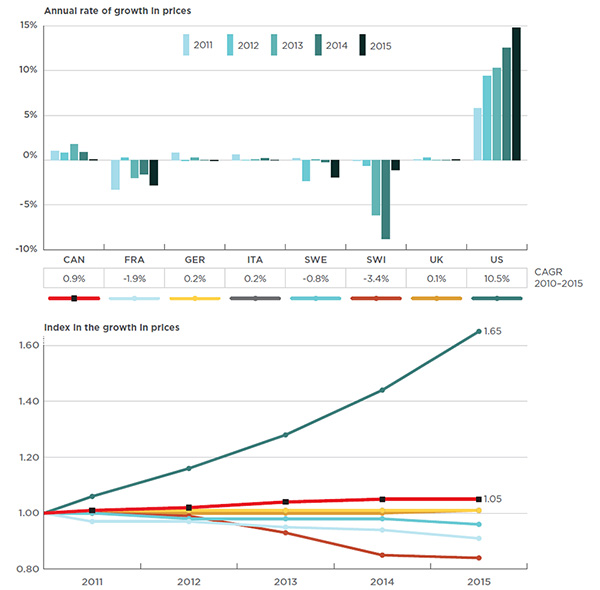
*France, Germany, Italy, Sweden, Switzerland, the United Kingdom and the United States.
Source: MIDAS™ Database, 2011 to 2015, IMS AG. All rights reserved.
Figure description
These two graphs give the trends in prices for biologic DMARDs in Canada and the PMPRB7 countries from 2011 to 2015.
The first graph is a bar graph depicting the annual growth rate in price for each country.
blank
| |
2011 |
2012 |
2013 |
2014 |
2015 |
Compound annual growth rate 2010–2015 |
| Canada |
1.0% |
0.8% |
1.8% |
0.9% |
0.1% |
0.9% |
| France |
-3.3% |
0.3% |
-2.0% |
-1.6% |
-2.8% |
-1.9% |
| Germany |
0.8% |
-0.1% |
0.3% |
0.0% |
-0.1% |
0.2% |
| Italy |
0.6% |
0.0% |
0.1% |
0.2% |
0.0% |
0.2% |
| Sweden |
0.2% |
-2.3% |
0.1% |
-0.2% |
-1.9% |
-0.8% |
| Switzerland |
-0.1% |
-0.6% |
-6.1% |
-8.8% |
-1.1% |
-3.4% |
| United Kingdom |
0.1% |
0.3% |
0.0% |
0.0% |
0.1% |
0.1% |
| United States |
5.8% |
9.4% |
10.3% |
12.5% |
14.8% |
10.5% |
The second line graph gives the corresponding indexed annual growth in price with the 2010 price for each country set to one.
blank
| |
2010 |
2011 |
2012 |
2013 |
2014 |
2015 |
| Canada |
1.00 |
1.01 |
1.02 |
1.04 |
1.05 |
1.05 |
| France |
1.00 |
0.97 |
0.97 |
0.95 |
0.94 |
0.91 |
| Germany |
1.00 |
1.01 |
1.01 |
1.01 |
1.01 |
1.01 |
| Italy |
1.00 |
1.01 |
1.01 |
1.01 |
1.01 |
1.01 |
| Sweden |
1.00 |
1.00 |
0.98 |
0.98 |
0.98 |
0.96 |
| Switzerland |
1.00 |
1.00 |
0.99 |
0.93 |
0.85 |
0.84 |
| United Kingdom |
1.00 |
1.00 |
1.00 |
1.00 |
1.00 |
1.01 |
| United States |
1.00 |
1.06 |
1.16 |
1.28 |
1.44 |
1.65 |
Pricing of biosimilars for Remicade
Canada versus PMPRB7* and other OECD† countries, 2015
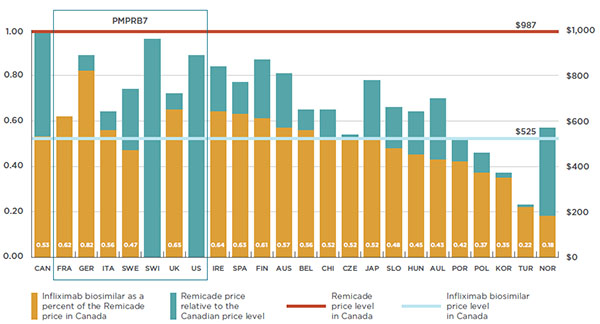
*France, Germany, Italy, Sweden, Switzerland, the United Kingdom and the United States.
†Ireland, Spain, Finland, Austria, Belgium, Chili, the Czech Republic, Japan, Slovakia, Hungry, Australia, Portugal, Poland, South Korea, Turkey and Norway.
Source: MIDAS™ Database, January–December 2015, IMS AG. All rights reserved.
Figure description
This bar graph illustrates the pricing of the biosimilar for Remicade in Canada versus the PMPRB7 and select Organisation for Economic Co-operation and Development countries in 2015. The Remicade price level in Canada – $978 – is given as one. The biosimilar price in Canada is $525.
blank
|
Infliximab biosimilar as a percent of the Remicade price in Canada |
Remicade price above the price of the infliximab biosimilar |
| Canada |
0.53 |
0.47 |
| France |
0.62 |
0.00 |
| Germany |
0.82 |
0.07 |
| Italy |
0.56 |
0.08 |
| Sweden |
0.47 |
0.27 |
| Switzerland |
– |
0.96 |
| United Kingdom |
0.65 |
0.07 |
| United States |
– |
0.89 |
| Ireland |
0.64 |
0.20 |
| Spain |
0.63 |
0.14 |
| Finland |
0.61 |
0.26 |
| Austria |
0.57 |
0.24 |
| Belgium |
0.56 |
0.09 |
| Chili |
0.52 |
0.13 |
| Czech Republic |
0.52 |
0.02 |
| Japan |
0.52 |
0.26 |
| Slovakia |
0.48 |
0.18 |
| Hungry |
0.45 |
0.19 |
| Australia |
0.43 |
0.27 |
| Portugal |
0.42 |
0.10 |
| Poland |
0.37 |
0.09 |
| South Korea |
0.35 |
0.02 |
| Turkey |
0.22 |
0.01 |
| Norway |
0.18 |
0.39 |
Meds Entry Watch, 2015
The Meds Entry Watch is a new NPDUIS annual publication that explores the market entry dynamics of new drugs launched in Canadian and international markets, including their availability, launch sequence, market penetration, sales and prices.
The first edition of this report includes a retrospective analysis of the new drugs launched over a six-year period from 2009 to 2014, as well as an early analysis of the drugs launched in 2015. The next edition of the Meds Entry Watch will build on this analysis and provide additional up-to-date information on new drugs launched in 2016.
Key Findings
- On average, 35 new active substances were launched each year between 2009 and 2014, for a total of 210. By the last quarter of 2015, the sales of these drugs accounted for 21.8% of the total brand-name pharmaceutical market in Canada and the PMPRB7.
- The availability of new drugs in Canada was similar to that in the international markets analyzed, with more than half the new drugs launched between 2009 and 2014 accounting for 97% of total domestic and foreign new drug sales in the last quarter of 2015.
- After an initial international launch, it took an average of 11 months for a new drug to be made available in Canada, which is well within the international norm; the 10 top-selling drugs had an even shorter average launch lag time of 3 months.
- The share of new products designated as orphan drugs increased in the countries analyzed from 17% in 2009 to 43% in 2015, with international list prices of 24 out of 35 new drugs launched in 2015 found to be in the hundreds or thousands of dollars.
- New direct-acting antiviral treatments for hepatitis C accounted for 25% of new drug sales in the countries analyzed in the last quarter of 2015.
Share of NASs launched by OECD country, Q4-2015
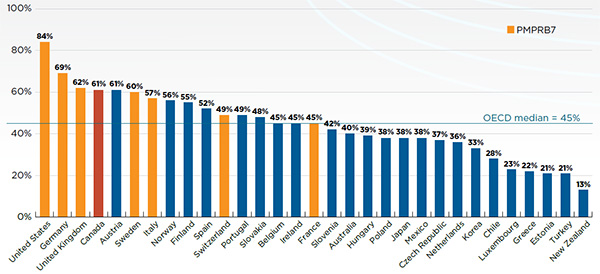
Note: New active substances (NASs) launched between 2009 and 2014 in Canada and the PMPRB7. A NAS was considered to be launched in a particular country once sales data was available in MIDAS™. For the Canadian market, Health Canada’s Drug Product Database and the Notice of Compliance database were cross-referenced to ensure that all NASs authorized for sale in Canada were accurately identified.
Source: MIDAS™ Database, October‒December 2015, IMS AG. All rights reserved.
Figure description
This bar graph depicts the share of new active substances launched by each country in the Organisation for Economic Co-operation and Development as of the fourth quarter of 2015.
blank
| Country |
Share of new active substances launched |
| United States |
84% |
| Germany |
69% |
| United Kingdom |
62% |
| Canada |
61% |
| Austria |
61% |
| Sweden |
60% |
| Italy |
57% |
| Norway |
56% |
| Finland |
55% |
| Spain |
52% |
| Switzerland |
49% |
| Portugal |
49% |
| Slovakia |
48% |
| Belgium |
45% |
| Ireland |
45% |
| France |
45% |
| Slovenia |
42% |
| Australia |
40% |
| Hungary |
39% |
| Poland |
38% |
| Japan |
38% |
| Mexico |
38% |
| Czech Republic |
37% |
| Netherlands |
36% |
| Korea |
33% |
| Chile |
28% |
| Luxembourg |
23% |
| Greece |
22% |
| Estonia |
21% |
| Turkey |
21% |
| New Zealand |
13% |
| Organisation for Economic Co-operation and Development median |
45% |
Number of NASs by year of launch, Canada and the PMPRB7*, 2009 to 2015
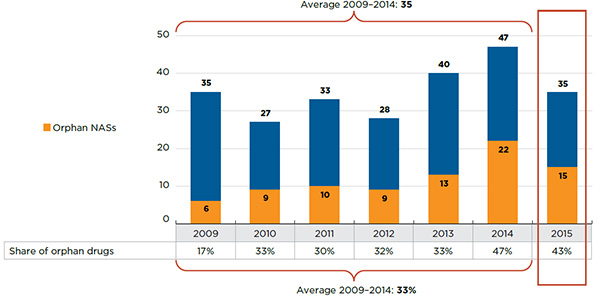
Note: New active substances (NASs) launched between 2009 and 2015. A NAS was considered to be launched in a particular country once sales data was available in MIDAS™. For this analysis, launches in the Canadian market were solely based on available MIDAS™ sales.
*France, Germany, Italy, Sweden, Switzerland, United Kingdom and the United States.
Source: MIDAS™ Database, 2009 to 2015, IMS AG. All rights reserved.
Figure description
This stacked bar graph depicts the number of new active substances by year of launch for Canada and the PMPRB comparator countries from 2009 to 2015. The number and share of drugs with an orphan indication is also given. Between 2009 and 2014, the average number of new active substances was 35 and 33% were orphan drugs.
blank
| |
2009 |
2010 |
2011 |
2012 |
2013 |
2014 |
2015 |
| Total new active substances |
35 |
27 |
33 |
28 |
40 |
47 |
35 |
| Orphan new active substances |
6 |
9 |
10 |
9 |
13 |
22 |
15 |
| Share of orphan new active substances |
17% |
33% |
30% |
32% |
33% |
47% |
43% |
CompassRx, 3rd Edition, Annual Public Drug Plan Expenditure Report, 2015/16
CompassRx is a flagship NPDUIS annual report that monitors and analyzes the major factors driving changes in prescription drug expenditures in public drug plans in Canada. The 2015/16 edition of this report identifies developing trends in drug use, demographics and pricing based on the results presented in previous publications.
Key Findings
- Prescription drug expenditures in the Canadian public drug plans totaled $11.3 billion in 2015–16, a $1 billion increase over the previous year.
- Drug costs, which accounted for three quarters of these expenditures, saw a 12% increase, with an 18.8% rise in the patented market segment.
- Patented drugs, the largest market segment at 58.5%, grew at a rate of 18.8% in 2015–16. Patented drugs exceeding $10,000 in annual treatment costs grew by 60.5%, accounting for 27.6% of drug costs, but were used by less than 1% of public drug plan beneficiaries.
- While new and curative treatments for hepatitis C were major contributors to this growth (8% of the 12% growth in drug costs), other high-costs drugs continued to put upward pressure on drug plan costs (4.1% of the 12% growth in drug costs).
- These relatively high rates of growth signal a shift in a previous trend of low growth marked by important savings from the “patent cliff” and generic drug price reforms, with the savings potential of these factors gradually diminishing in recent years and only partially counterbalancing cost pressures from higher-cost drugs in 2015–16.
- Drug costs were the largest component of total expenditures, accounting for nearly three quarters (74.7%) in 2015-16, followed by dispensing costs (21.8%), and reported markups (3.5%).
Drug cost drivers, NPDUIS public drug plans*, 2012/13 to 2015/16
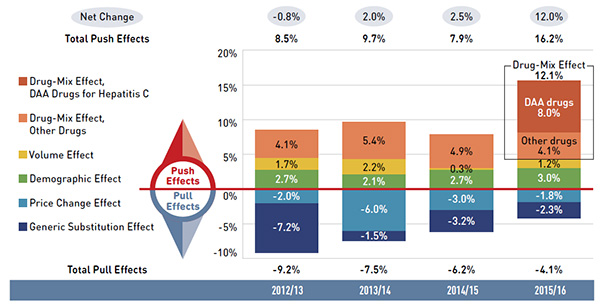
Note: Values may not add to totals due to rounding and the cross effect. Results for 2012/13 do not capture the data for the British Columbia and Newfoundland and Labrador provincial public drug plans.
*British Columbia, Alberta, Saskatchewan, Manitoba, Ontario, New Brunswick, Nova Scotia, Prince Edward Island, Newfoundland and Labrador, and the Non-Insured Health Benefits Program.
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure description
This bar graph and adjacent tables describe the factors or effects that impacted the rates of change in drug costs in the NPDUIS public drug plans for each year from 2012/13 to 2015/16. Each bar is broken out into the following factors - demographic, volume, drug mix, price change and generic substitution - and a percent change is assigned to each. The bars are totals for all NPDUIS drug plans combined, and the effects for Hepatitis C drugs are isolated because of their high impact. Three one-line tables above and below the bar graph show the total positive or push effects and net change, (above) and negative or pull effects, (below) for each year. A color-coded table to the right of the bar graph provides written analysis for each effect.
blank
|
2012/13 |
2013/14 |
2014/15 |
2015/16 |
| Demographic |
2.7% |
2.1% |
2.7% |
3.0% |
| Volume |
1.7% |
2.2% |
0.3% |
1.2% |
| Drug Mix, Other Drugs |
4.1% |
5.4% |
4.9% |
4.1% |
| Drug Mix, Hepatitis C |
0.0% |
0.0% |
0.0% |
8.0% |
| Price Change |
-2.0% |
-6.0% |
-3.0% |
-1.8% |
| Generic Substitution |
-7.2% |
-1.5% |
-3.2% |
-2.3% |
| Total pull effects |
-9.2% |
-7.5% |
-6.2% |
-4.1% |
| Total push effects |
8.5% |
9.7% |
7.9% |
16.2% |
| Net change |
-0.8% |
2.0% |
2.5% |
12.0% |
Overview of Drug Cost Drivers
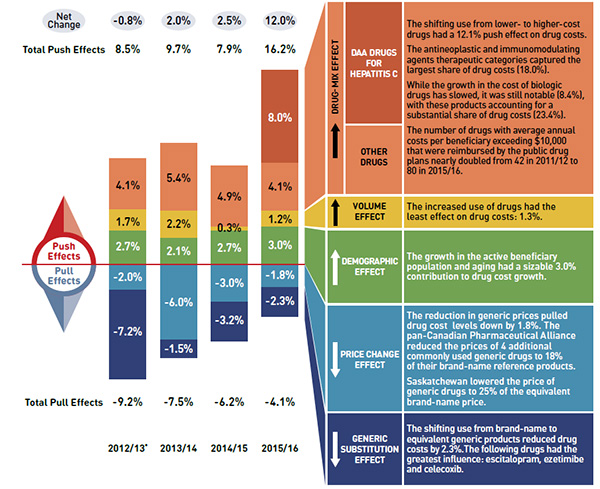
* Results for 2012/13 do not capture the data for the British Columbia and Newfoundland and Labrador provincial public drug plans.
Note: Values may not add to totals due to rounding and the cross effect.
Data source: National Prescription Drug Utilization Information System Database, Canadian Institute for Health Information.
Figure description
This bar graph and adjacent tables describe the factors or effects that impacted the rates of change in drug costs in the NPDUIS public drug plans for each year from 2012/13 to 2015/16. Each bar is broken out into the following factors - demographic, volume, drug mix, price change and generic substitution - and a percent change is assigned to each. The bars are totals for all NPDUIS drug plans combined, and the effects for Hepatitis C drugs are isolated because of their high impact. Three one-line tables above and below the bar graph show the total positive or push effects and net change, (above) and negative or pull effects, (below) for each year. A color-coded table to the right of the bar graph provides written analysis for each effect.
blank
|
2012/13 |
2013/14 |
2014/15 |
2015/16 |
| Demographic |
2.7% |
2.1% |
2.7% |
3.0% |
| Volume |
1.7% |
2.2% |
0.3% |
1.2% |
| Drug Mix, Other Drugs |
4.1% |
5.4% |
4.9% |
4.1% |
| Drug Mix, Hepatitis C |
0.0% |
0.0% |
0.0% |
8.0% |
| Price Change |
-2.0% |
-6.0% |
-3.0% |
-1.8% |
| Generic Substitution |
-7.2% |
-1.5% |
-3.2% |
-2.3% |
| Total pull effects |
-9.2% |
-7.5% |
-6.2% |
-4.1% |
| Total push effects |
8.5% |
9.7% |
7.9% |
16.2% |
| Net change |
-0.8% |
2.0% |
2.5% |
12.0% |
Research Agenda
The NPDUIS research agenda for the two upcoming fiscal years includes the following analytical studies:
- CompassRx, 4th Edition, 2016/17
- Meds Entry Watch, 2nd Edition, 2016
- Market Intelligence Report, 2nd Edition, 2016
- Generics360, 2016
- Private Drug Plans in Canada – Part 2: Cost Driver Analysis, 2015
- Private Drug Plans in Canada – Part 3: High-Cost Drugs and Beneficiaries, 2015
- Potential Savings from Biosimilars in Canada
- The Canadian Drug Reimbursement Landscape: A Review of Public and Private Markets
Additional research topics may be pursued based on consultation with the NPDUIS Advisory Committee.
Analysis of Research and Development Expenditures: R&D Investment Falling Short of Target
Innovation is vital to advancing health care. In part, the provisions of Canada’s Patent Act are intended to foster an investment climate favorable to pharmaceutical research and development (R&D) in Canada. However, the percentage of R&D-to-sales by pharmaceutical patentees in Canada has been falling since the late 1990’s and has been under the agreed-upon target of 10% since 2003. In 2016, it was at 4.4% for all patentees and 4.9% for members of Innovative Medicines Canada.
4.4% R&D-to-Sales Ratio
The R&D-to-Sales Ratio for all Patentees was 4.4% in 2016. This represents a 62% decrease from a peak of 11.7% in 1995.
The PMPRB7 Average R&D Ratio is 5 X Greater than Canada
The R&D-to-Sales ratio obtained by aggregating R&D spending and sales across all seven comparator countries was 22.4%, more than five times Canada's.
Analysis of Research and Development Expenditures
The Act mandates the PMPRB to monitor and report on pharmaceutical R&D spending. This chapter provides key statistics on the current state of pharmaceutical R&D investment in Canada.
Data Sources
The statistical results presented in this report were entirely derived from data that patentees submitted to the PMPRB.
The Act requires each patentee to report its total gross revenues from sales of all drugs for human or veterinary use (including revenues from sales of non-patented drug products and from licensing agreements) and R&D expenditures in Canada related to medicines (both patented and non-patented for human or veterinary use). Patentees transmit this information to the PMPRB by means of its Form 3 (Revenues and Research and Development Expenditures Provided Pursuant to subsection 88(1) of the Patent Act).
The Patented Medicines Regulations (Regulations) require that each submitted Form 3 be accompanied by a certificate stating the information it contains is “true and correct”. The Board does not audit Form 3 submissions, but it does review submitted data for anomalies and inconsistencies, seeking corrections or clarifications from patentees where necessary. To confirm that PMPRB staff has correctly interpreted the data submitted, each patentee is given the opportunity to review and confirm the accuracy of its own R&D-to-sales ratio before that ratio is published.
Failure to File R&D Expenditures (Form 3)
It is a patentee’s responsibility to ensure a complete and accurate Form 3 is filed within the time frame set out in the Regulations. If a patentee fails to meet these filing requirements, the Board may issue an Order demanding compliance. There were no such Board Orders issued for the 2016 reporting period.
Coverage
Note that companies without sales of patented medicines do not need to report their R&D expenditures to the PMPRB. This has two implications.
First, the statistical results reported here should not be taken to cover all pharmaceutical research conducted in Canada. For example, a company may sell only non-patented drug products but may still perform considerable research in Canada. Similarly, a company may conduct research and have no product sales at all.Footnote 32 The results presented below will not reflect the R&D expenditures of firms in either situation.
Second, as new patented drug products come onto the Canadian market and existing patents expire, the number and identity of companies required to file R&D data may change from year to year. A total of 78 companies reported on their R&D activity in 2016. Of these, 32 were members of Innovative Medicines Canada.
Definition of Sales Revenues
For reporting purposes, sales revenues are defined as total gross revenues from sales in Canada of all drug products and from licensing agreements (e.g., royalties and fees accruing to the patentee related to sales in Canada by licensees).
Definition of R&D Expenditures
Pursuant to section 6 of the Regulations, patentees are required to report R&D expenditures that would have qualified for an investment tax credit in respect to scientific research and experimental development (SR&ED) under the provisions of the Income Tax Act that came into effect on December 1, 1987.Footnote 33 By this definition, R&D expenditures may include current expenditures, capital equipment costs and allowable depreciation expenses. Market research, sales promotions, quality control or routine testing of materials, devices or products and routine data collection are not eligible for an investment tax credit and, therefore, are not to be included in the R&D expenditures reported by patentees.
Total Sales Revenues and R&D Expenditures
Table 18 provides an overview of reported sales revenues and R&D expenditures over the period 1988 through 2016.
Patentees reported total 2016 sales revenues of $20.8 billion, an increase of 5.9% from 2015. Sales revenues reported by Innovative Medicines Canada members were $15.6 billion, accounting for 75% of the total. (Less than 1% of reported sales revenues were generated by licensing agreements.)
Patentees reported R&D expenditures of $918.2 million in 2016, an increase of 5.7% over 2015. Innovative Medicines Canada members reported R&D expenditures of $769.9 million in 2016, an increase of 0.3% over last year. Innovative Medicines Canada members accounted for 83.9% of all reported R&D expenditures in 2016.
R&D-to-Sales Ratios
Table 18 and Figure 23 also provide ratios of R&D expenditures to sales revenues. It should be noted in this context that, with the adoption of the 1987 amendments to the Act, Innovative Medicines Canada made a public commitment to increase its members’ annual R&D expenditures to 10% of sales revenues by 1996.Footnote 34 This level of R&D expenditure was reached by 1993, with the ratio exceeding 10% in some years.
Figure 23. R&D-to-Sales Ratio, Pharmaceutical Patentees, 1988–2016
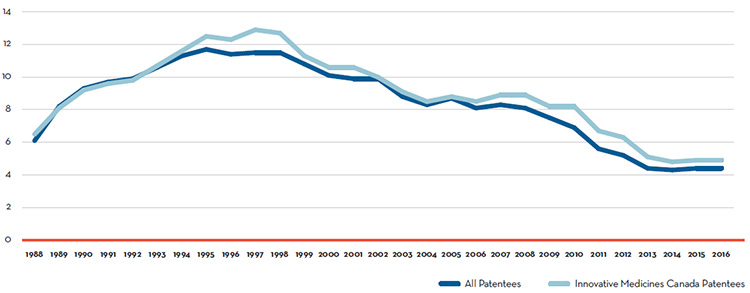
Figure description
This is a line graph comparing the R&D-to-sales ratio of all patentees vs. Innovative Medicines Canada patentees based on data collected between 1988 and 2016.
Innovative Medicines Canada Patentees: 1988, 6.5; 1989, 8.1; 1990, 9.2; 1991, 9.6; 1992, 9.8; 1993, 10.7; 1994, 11.6; 1995, 12.5; 1996, 12.3; 1997, 12.9; 1998, 12.7; 1999, 11.3; 2000, 10.6; 2001, 10.6; 2002, 10; 2003, 9.1; 2004, 8.5; 2005, 8.8; 2006, 8.5; 2007, 8.9; 2008, 8.9; 2009, 8.2; 2010, 8.2; 2011, 6.7; 2012, 6.3; 2013, 5.1; 2014, 4.8; 2015, 4.9; 2016, 4.9.
All Patentees: 1988, 6.1; 1989, 8.2; 1990, 9.3; 1991, 9.7; 1992, 9.9; 1993, 10.6; 1994, 11.3; 1995, 11.7; 1996, 11.4; 1997, 11.5; 1998, 11.5; 1999, 10.8; 2000, 10.1; 2001, 9.9; 2002, 9.9; 2003, 8.8; 2004, 8.3; 2005, 8.7; 2006, 8.1; 2007, 8.3; 2008, 8.1; 2009, 7.5; 2010, 6.9; 2011, 5.6; 2012, 5.2; 2013, 4.4; 2014, 4.3; 2015, 4.4; 2016, 4.4.
The ratio of R&D expenditures to sales revenues among all patentees was 4.4% in 2016, unchanged from 4.4% in 2015. The overall R&D-to-sales ratio has been less than 10% for the past 16 consecutive years.
The corresponding R&D-to-sales ratio for members of Innovative Medicines Canada was 4.9% in 2016, unchanged from 4.9% in 2015.Footnote 35 The Innovative Medicines Canada ratio has been less than 10% for the past 14 consecutive years.
Table 23 in Appendix 3 provides details on the range of 2016 R&D-to-sales ratios. Of the 78 companies reporting in 2016, 89.7% had R&D-to-sales ratios below 10%.
Table 18. Total R&D Expenditures and R&D-to-Sales Ratios of Reporting Companies, 1988–2016
| Year |
All patentees |
Innovative Medicines Canada |
R&D-to-sales ratio: all patentees (%) |
R&D-to-sales ratio: Innovative Medicines Canada patentees (%) |
| Number of companies reporting |
R&D expenditures by all patentees ($millions) |
Change from previous year (%) |
Sales revenues ($millions) |
Change from previous year (%) |
R&D expenditures by Innovative Medicines Canada patentees ($millions) |
Change from previous year (%) |
Sales revenues by Innovative Medicines Canada patentees ($millions) |
Change from previous year (%) |
| 2016 |
78 |
918.2 |
5.7 |
20,855.7 |
5.9 |
769.9 |
0.3 |
15,599.9 |
0.2 |
4.4 |
4.9 |
| 2015 |
77 |
869.1 |
9.7 |
19,693.3 |
6.7 |
767.4 |
7.8 |
15,565.1 |
4.7 |
4.4 |
4.9 |
| 2014 |
75 |
792.2 |
-0.8 |
18,455.1 |
1.0 |
711.7 |
2.0 |
14,861.1 |
9.2 |
4.3 |
4.8 |
| 2013 |
81 |
798.3 |
-14.7 |
18,268.1 |
1.4 |
697.5 |
-15.4 |
13,614.8 |
3.4 |
4.4 |
5.1 |
| 2012 |
85 |
936.1 |
-5.6 |
18,021.1 |
1.3 |
824.1 |
-8.6 |
13,162.8 |
-2.1 |
5.2 |
6.3 |
| 2011 |
79 |
991.7 |
-15.8 |
17,798.8 |
4.7 |
901.2 |
-9.9 |
13,446.1 |
10.7 |
5.6 |
6.7 |
| 2010 |
82 |
1,178.2 |
-7.4 |
17,000.0 |
-0.3 |
1,000.2 |
-11.7 |
12,149.0 |
-11.8 |
6.9 |
8.2 |
| 2009 |
81 |
1,272.0 |
-2.9 |
17,051.9 |
4.5 |
1,132.9 |
-3.4 |
13,780.0 |
4.6 |
7.5 |
8.2 |
| 2008 |
82 |
1,310.7 |
-1.1 |
16,316.7 |
2.0 |
1,172.2 |
-1.0 |
13,178.2 |
-1.4 |
8.1 |
8.9 |
| 2007 |
82 |
1,325.0 |
9.5 |
15,991.0 |
7.3 |
1,184.4 |
24.8 |
13,359.8 |
20.0 |
8.3 |
8.9 |
| 2006 |
72 |
1,210.0 |
-1.9 |
14,902.0 |
4.7 |
949.0 |
-8.8 |
11,131.2 |
-5.8 |
8.1 |
8.5 |
| 2005 |
80 |
1,234.3 |
5.5 |
14,231.3 |
0.5 |
1,040.1 |
3.9 |
11,821.4 |
0.0 |
8.7 |
8.8 |
| 2004 |
84 |
1,170.0 |
-2.0 |
14,168.3 |
4.0 |
1,000.8 |
0.8 |
11,819.0 |
8.8 |
8.3 |
8.5 |
| 2003 |
83 |
1,194.3 |
-0.4 |
13,631.1 |
12.8 |
992.9 |
-3.6 |
10,865.7 |
5.2 |
8.8 |
9.1 |
| 2002 |
79 |
1,198.7 |
13.0 |
12,081.2 |
12.5 |
1,029.6 |
10.1 |
10,323.8 |
16.8 |
9.9 |
10.0 |
| 2001 |
74 |
1,060.1 |
12.6 |
10,732.1 |
15.3 |
935.2 |
14.7 |
8,835.4 |
14.3 |
9.9 |
10.6 |
| 2000 |
79 |
941.8 |
5.3 |
9,309.6 |
12.0 |
815.5 |
4.0 |
7,728.8 |
11.6 |
10.1 |
10.6 |
| 1999 |
78 |
894.6 |
12.0 |
8,315.5 |
19.2 |
784.3 |
9.9 |
6,923.4 |
22.8 |
10.8 |
11.3 |
| 1998 |
74 |
798.9 |
10.2 |
6,975.2 |
10.9 |
713.7 |
8.6 |
5,640.2 |
10.6 |
11.5 |
12.7 |
| 1997 |
75 |
725.1 |
9.0 |
6,288.4 |
7.4 |
657.4 |
10.3 |
5,098.2 |
4.9 |
11.5 |
12.9 |
| 1996 |
72 |
665.3 |
6.4 |
5,857.4 |
9.9 |
595.8 |
6.5 |
4,859.5 |
8.7 |
11.4 |
12.3 |
| 1995 |
71 |
625.5 |
11.5 |
5,330.2 |
7.5 |
559.5 |
9.8 |
4,468.8 |
1.4 |
11.7 |
12.5 |
| 1994 |
73 |
561.1 |
11.4 |
4,957.4 |
4.4 |
509.5 |
10.4 |
4,407.2 |
2.0 |
11.3 |
11.6 |
| 1993 |
70 |
503.5 |
22.1 |
4,747.6 |
14.0 |
461.4 |
24.0 |
4,321.4 |
14.4 |
10.6 |
10.7 |
| 1992 |
71 |
412.4 |
9.6 |
4,164.4 |
6.9 |
372.1 |
9.0 |
3,778.4 |
6.5 |
9.9 |
9.8 |
| 1991 |
65 |
376.4 |
23.2 |
3,894.8 |
18.1 |
341.4 |
24.7 |
3,546.9 |
19.5 |
9.7 |
9.6 |
| 1990 |
65 |
305.5 |
24.8 |
3,298.8 |
11.0 |
273.8 |
25.8 |
2,967.9 |
10.5 |
9.3 |
9.2 |
| 1989 |
66 |
244.8 |
47.4 |
2,973.0 |
9.4 |
217.6 |
34.7 |
2,685.5 |
7.3 |
8.2 |
8.1 |
| 1988 |
66 |
165.7 |
— |
2,718.0 |
— |
161.5 |
— |
2,502.3 |
— |
6.1 |
6.5 |
Source: PMPRB
Current Expenditures by Type of Research
Table 19 and Figure 24 (as well as Figure 26 in Appendix 3) provide information on the allocation of 2016 current R&D expendituresFootnote 36 among basic and applied research and other qualifying R&D.Footnote 37 Patentees reported spending $105.9 million on basic research in 2016, representing 12.6% of current R&D expenditures and an increase of 3.6% over the previous year. Patentees reported spending $500.9 million on applied research, representing 59.5% of current R&D expenditures. Clinical trials accounted for 72.0% of applied research expenditures.
Table 19. Current R&D Expenditures by Type of Research, 2016 and 2015
| Type of research |
Expenditures:
2016
($millions) |
Share:
2016
(%) |
Expenditures:
2015
($millions) |
Share:
2015
(%) |
Annual
change in
expenditures
(%) |
| Basic |
105.9 |
12.6 |
102.2 |
12.9 |
3.6 |
| Chemical |
72.1 |
8.6 |
66.4 |
8.4 |
8.6 |
| Biological |
33.8 |
4.0 |
35.8 |
4.5 |
-5.6 |
| Applied |
500.9 |
59.5 |
456.2 |
57.7 |
9.7 |
| Manufacturing process |
79.7 |
9.5 |
58.0 |
7.3 |
36.7 |
| Pre-clinical Trial I |
37.2 |
4.4 |
40.4 |
5.1 |
-7.9 |
| Pre-clinical Trial II |
24.6 |
2.9 |
26.7 |
3.4 |
-7.9 |
| Clinical Trial Phase I |
49.4 |
5.9 |
25.1 |
3.2 |
96.8 |
| Clinical Trial Phase II |
68.1 |
8.1 |
67.1 |
8.5 |
1.5 |
| Clinical Trial Phase III |
241.9 |
28.8 |
238.9 |
30.2 |
1.3 |
| Other qualifying R&D |
234.9 |
27.9 |
231.7 |
29.3 |
1.4 |
| Total |
841.7 |
100.0† |
790.1 |
100.0† |
6.5 |
† Values in this column may not add due to rounding
Source: PMPRB
Figure 24. Current R&D Expenditures by Type of Research, 1988–2016
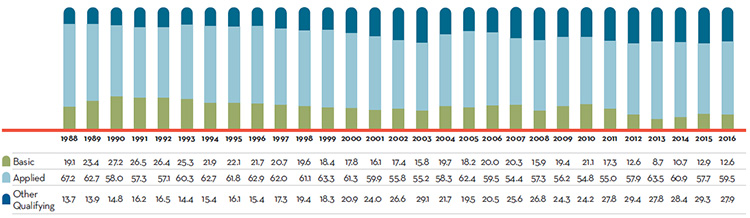
Figure description
This is a vertical stacked bar graph that depicts current R&D expenditures by type of research, based on data collected between 1988 and 2016, in percent. In 1988, basic types of research accounted for 19.1% of R&D expenditures. Applied types of research accounted for 67.2% of R&D expenditures. Other qualifying types of research accounted for 13.7% of R&D expenditures.
- 1989: Basic 23.4, Applied 62.7, Other Qualifying 13.9;
- 1990: Basic 27.2, Applied 58.0, Other Qualifying 14.8;
- 1991: Basic 26.5, Applied 57.3, Other Qualifying 16.2;
- 1992: Basic 26.4, Applied 57.1, Other Qualifying 16.5;
- 1993: Basic 25.3, Applied 60.3, Other Qualifying 14.4;
- 1994: Basic 21.9, Applied 62.7, Other Qualifying 15.4;
- 1995: Basic 22.1, Applied 61.8, Other Qualifying 16.1;
- 1996: Basic 21.7, Applied 62.9, Other Qualifying 15.4;
- 1997: Basic 20.7, Applied 62.0, Other Qualifying 17.3;
- 1998: Basic 19.6, Applied 61.1, Other Qualifying 19.4;
- 1999: Basic 18.4, Applied 63.3, Other Qualifying 18.3;
- 2000: Basic 17.8, Applied 61.3, Other Qualifying 20.9;
- 2001: Basic 16.1, Applied 59.9, Other Qualifying 24.0;
- 2002: Basic 17.4, Applied 55.8, Other Qualifying 26.6;
- 2003: Basic 15.8, Applied 55.2, Other Qualifying 29.1;
- 2004: Basic 19.7, Applied 58.3, Other Qualifying 21.7;
- 2005: Basic 18.2, Applied 62.4, Other Qualifying 19.5;
- 2006: Basic 20.0, Applied 59.5, Other Qualifying 20.5;
- 2007: Basic 20.3, Applied 54.4, Other Qualifying 25.6;
- 2008: Basic 15.9, Applied 57.3, Other Qualifying 26.8;
- 2009: Basic 19.4, Applied 56.2, Other Qualifying 24.3;
- 2010: Basic 21.1, Applied 54.8, Other Qualifying 24.2;
- 2011: Basic 17.3, Applied 55.0, Other Qualifying 27.8;
- 2012: Basic 12.6, Applied 57.9, Other Qualifying 29.4;
- 2013: Basic 8.7, Applied 63.5, Other Qualifying 27.8;
- 2014: Basic 10.7 Applied 60.9, Other Qualifying 28.4;
- 2015: Basic 12.9 Applied 57.7, Other Qualifying 29.3;
- 2016: Basic 12.6 Applied 59.5, Other Qualifying 27.9
Current R&D Expenditures by Performer
Patentees report expenditures on research they conduct themselves (intramural) and research performed by other establishments, such as universities, hospitals and other manufacturers (extramural). Table 20 shows that 46.9% of 2016 current research expenditures were intramural. Research performed by other companies on behalf of patentees was 25.4% of current expenditures, while research conducted in universities and hospitals accounted for 15.6%.
Table 20. Current R&D Expenditures by R&D Performer, 2016 and 2015
| R&D performer |
Expenditures:
2016
($millions) |
Share:
2016
(%) |
Expenditures:
2015
($millions) |
Share:
2015
(%) |
Annual
change
in
expenditures
(%) |
| Intramural |
| Patentees |
394.9 |
46.9 |
390.0 |
49.4 |
1.3 |
| Extramural |
| Universities and hospitals |
131.4 |
15.6 |
134.6 |
17.0 |
-2.4 |
| Other companies |
213.6 |
25.4 |
170.3 |
21.6 |
25.5 |
| Others |
101.8 |
12.1 |
95.2 |
12.1 |
6.9 |
| Total † |
841.7 |
100.0 |
790.1 |
100.0 |
6.5 |
† Values in this row may not add due to rounding
Source: PMPRB
Total R&D Expenditures by Source of Funds
Table 21 provides information on the sources of funds used by patentees to finance their R&D activity. Internal company funds remained by far the single largest source of funding in 2016, accounting for 92.4% of total expenditures. Funds received from government amounted to 0.6% of total expenditures.
Table 21. Total R&D Expenditures by Source of Funds, 2016 and 2015
| Source of funds |
Expenditures:
2016
($millions) |
Share:
2016
(%) |
Expenditures:
2015
($millions) |
Share:
2015
(%) |
Annual
increase in
expenditures
(%) |
| Company funds |
848.5 |
92.4 |
791.7 |
91.1 |
7.2 |
| Federal/provincial governments |
5.4 |
0.6 |
8.3 |
1.0 |
-35.0 |
| Others |
64.3 |
7.0 |
69.1 |
8.0 |
-7.0 |
| Total† |
918.2 |
100.0 |
869.1 |
100.0 |
5.7 |
† Values in this row may not add due to rounding
Source: PMPRB
Current R&D Expenditures by Region
Table 22 (as well as Table 25 and Table 26 in Appendix 3) show current R&D expenditures by region. As in previous years, current expenditures were heavily concentrated in Ontario and Quebec in 2016, with these provinces accounting for 81.5% of total expenditures. While current R&D expenditures increased at a year-over-year rate of 3.2% in Western Canada, they remained unchanged in Ontario and increased by 20.0% in Quebec.
Table 22. Current R&D Expenditures by Region, 2016 and 2015
| Region |
Expenditures: 2016
($millions) |
Share: 2016
(%) |
Expenditures: 2015
($millions) |
Share: 2015
(%) |
Annual change in
expenditures
(%) |
| Atlantic provinces |
16.0 |
1.9 |
14.3 |
1.8 |
12.1 |
| Quebec |
272.6 |
32.4 |
227.1 |
28.7 |
20.0 |
| Ontario |
413.1 |
49.1 |
413.0 |
52.3 |
0.0 |
| Western provinces |
140.0 |
16.6 |
135.7 |
17.2 |
3.2 |
| Territories |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
| Total† |
841.7 |
100.0 |
790.1 |
100.0 |
6.5 |
† Values in this row may not add due to rounding
Source: PMPRB
The Global Context
Figure 25 compares Canadian pharmaceutical R&D-to-sales ratios for the years 2000 and 2014 to those in the PMPRB’s seven comparator countries.Footnote 38 Canada’s ratio stood at 10.1% in 2000. Only Italy, at 6.2%, had a lower ratio in that year, while Switzerland had the highest ratio at 102.5%.
In 2014, Canada stood at the bottom of the range at 4.3%, with Italy second lowest at 6.3%. Ratios in all other comparator countries remained well above Canada’s. The ratio obtained by aggregating R&D spending and sales across all seven comparator countries was 22.4%, more than five times Canada’s.
Figure 25. R&D-to-Sales Ratios, Canada and Comparator Countries
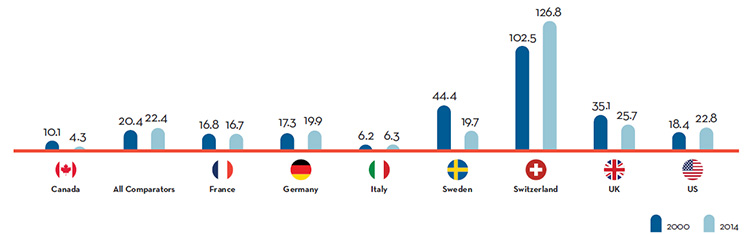
Source: PMPRB; European Federation of Pharmaceutical Industries and Associations (EFPIA): The Pharmaceutical Industry in Figures 2016, PhRMA 2016 profile
Figure description
This is a bar graph depicting R&D-to-sales ratios between Canada and comparator countries in 2000 and 2014.
2000: Canada, 10.1; All Comparators, 20.4; France, 16.8; Germany, 17.3; Italy, 6.2; Sweden, 44.4; Switzerland, 102.5; UK, 35.1; US, 18.4
2014: Canada, 4.3; All Comparators, 22.4; France, 16.7; Germany, 19.9; Italy, 6.3; Sweden, 19.7; Switzerland, 126.8; UK, 25.7; US, 22.8
The R&D-to-sales ratios represented in Figure 25 may be compared to the average bilateral price ratios reported in Table 11 (see Comparison of Canadian Prices to Foreign Prices section). Several comparator countries, which have patented drug prices that are, on average, substantially less than prices in Canada, have achieved R&D-to-sales ratios well above those in Canada.
As noted in previous years’ reports, there are a multitude of factors that drive the location of pharmaceutical R&D. These include where companies can find the best science base at reasonable cost and ready access to a quality clinical trials infrastructure. Although price levels are often cited as an important policy lever for attracting R&D, the data has not supported this link domestically or internationally.
Appendix 1: Glossary
For more detailed information and definitions please refer to the Patent Act, the Patented Medicines Regulations, the PMPRB Compendium of Policies, Guidelines and Procedures, and the Food and Drug Regulations, or contact the PMPRB.
Active Ingredient: Chemical or biological substance responsible for the claimed pharmacologic effect of a drug product.
Advance Ruling Certificate (ARC): A non-binding advance ruling certificate may be issued pursuant to subsection 98(4) of the Patent Act at the request of a patentee when the Board is satisfied that the price or proposed price of the medicine would not exceed the maximum non-excessive price under the Board's Guidelines.
ATC: Anatomical Therapeutic Chemical (ATC) classification system, developed and maintained by the World Health Organization (WHO) Collaborating Centre for Drug Statistics Methodology, divides drugs into different groups according to their site of action and therapeutic and chemical characteristics. This system is used by the PMPRB as a guide for selecting comparable medicines for purposes of price review.
Dedication of Patent: A practice whereby a patentee notifies the Commissioner of Patents that it has surrendered its rights and entitlements flowing from the patent for the benefit of the public to use and enjoy. NB: As of January 30, 1995, the Board does not recognize dedication of patent as a means to remove the medicine from its jurisdiction.
Drug Identification Number (DIN): A registration number (drug identification number) that the Health Products and Food Branch of Health Canada assigns to each prescription and non-prescription drug product marketed under the Food and Drug Regulations. A DIN uniquely identifies the following product characteristics: manufacturer; product name; active ingredient(s); strength(s) of active ingredient(s); pharmaceutical dosage form; route of administration.
Drug Product: A particular presentation of a medicine characterized by its pharmaceutical dosage form and the strength of the active ingredient(s).
Failure to File: The complete or partial failure of a patentee to comply with regulatory filing requirements pursuant to the Patent Act and the Patented Medicines Regulations.
Failure to Report: The complete failure of a patentee to have reported a patented drug product being sold in accordance with regulatory filing requirements pursuant to the Patent Act and the Patented Medicines Regulations.
Generic Product: A drug product with the same active ingredient, strength and dosage form of a brand name drug product.
License, Voluntary: A contractual agreement between a patent holder and a licensee under which the licensee is entitled to enjoy the benefit of the patent or to exercise any rights in relation to the patent for some consideration (i.e., royalties in the form of a share of the licensee’s sales).
Medicine: Any substance or mixture of substances made by any means, whether produced biologically, chemically, or otherwise, that is applied or administered in vivo in humans or in animals to aid in the diagnosis, treatment, mitigation or prevention of disease, symptoms, disorders, abnormal physical states, or modifying organic functions in humans and or animals, however administered. For greater certainty, this definition includes vaccines, topical preparations, anaesthetics and diagnostic products used in vivo, regardless of delivery mechanism (e.g., transdermal, capsule form, injectable, inhaler, etc.). This definition excludes medical devices, in vitro diagnostic products and disinfectants that are not used in vivo.
Notice of Compliance (NOC): Means a notice issued under section C.08.004 or C.08.004.01 of the Food and Drug Regulations. The issuance of an NOC indicates that a drug product meets the required Health Canada standards for use in humans or animals and that the product is approved for sale in Canada.
Patent: An instrument issued by the Commissioner of Patents in the form of letters patent for an invention that provides its holder with a monopoly limited in time, for the claims made within the patent. A patent gives its holder and its legal representatives, the exclusive right of making, constructing and using the invention and selling it to others to be used.
Patented Medicine Price Index (PMPI): The PMPI was developed by the PMPRB as a measure of average year-over-year change in the transaction prices of patented drug products sold in Canada, based on the price and sales information reported by patentees.
Patentee: As defined by subsection 79(1) of the Patent Act, “the person for the time being entitled to the benefit of the patent for that invention and includes, where any other person is entitled to exercise any rights in relation to that patent other than under a license continued by subsection 11(1) of the Patent Act Amendment Act, 1992, that other person in respect of those rights.”
Pending Patent: An application for a patent that has not yet been issued.
PMPRB7: France, Germany, Italy, Sweden, Switzerland, the United Kingdom and the United States.
Research and Development (R&D): Basic or applied research for the purpose of creating new, or improving existing, materials, devices, products or processes (e.g., manufacturing processes).
Research and Development–Applied Research: R&D directed toward a specific practical application, comprising research intended to improve manufacturing processes, pre-clinical trials and clinical trials.
Research and Development–Basic Research: R&D defined as work that advances scientific knowledge without a specific application in mind.
Research and Development–Other Qualifying: Includes eligible research and development expenditures that cannot be classified into any of the preceding categories of “type of research and development”. It includes drug regulation submissions, bioavailability studies and Phase IV clinical trials.
Research and Development Expenditures: For the purposes of the Patented Medicines Regulations, in particular Sections 5 and 6, research and development includes activities for which expenditures would have qualified for the investment tax credit for scientific research and experimental development under the Income Tax Act as it read on December 1, 1987.
Research and Development Expenditures–Current: Consist of the following non-capital expenses that are directly related to research work: (a) wages and salaries, (b) direct material, (c) contractors and subcontractors, (d) other direct costs such as factory overhead, (e) payments to designated institutions, (f) payments to granting councils, and (g) payments to other organizations. These elements are described in greater detail in the Patentees' Guide to Reporting—Form 3, available from the PMPRB Website under Regulatory Filings.
Special Access Programme (SAP): A program operated by Health Canada to give practitioners access to drugs that are not approved or otherwise available for sale in Canada.
Voluntary Compliance Undertaking (VCU): A written undertaking by a patentee to adjust its price to conform to the Board's Guidelines. A VCU represents a compromise between the PMPRB and the patentee as a result of negotiations between the parties geared towards a satisfactory resolution of an investigation initiated by Board Staff as per the Guidelines. A VCU takes into account the specific facts and underlying context of a particular case. As such, VCUs are not intended to have precedential value. The Chairperson may accept a VCU in lieu of issuing a Notice of Hearing if it is in the public interest. A VCU can also be submitted following the issuance of a Notice of Hearing. A VCU submitted at this point must be approved by the Board Hearing Panel struck to hear the matter. The Board reports publicly on all VCUs accepted by the Chairperson or the Board.
Appendix 2: Patented Drug Products First Reported to the PMPRB in 2016
blank
|
Brand Name |
Company |
DIN |
Status |
Level of therapeutic improvement/
category* |
| 1 |
AFINITOR – 7.5 mg/tablet |
Novartis Pharmaceuticals Canada Inc. |
02450267 |
Does Not Trigger Investigation |
SN |
| 2 |
ALPROLIX – 500 IU/vial |
Biogen Canada Inc. |
02422913 |
Under Review |
SN |
| 3 |
ALPROLIX – 1000 IU/vial |
Biogen Canada Inc. |
02422921 |
Under Review |
SN |
| 4 |
ALPROLIX – 2000 IU/vial |
Biogen Canada Inc. |
02422948 |
Under Review |
SN |
| 5 |
ALPROLIX – 3000 IU/vial |
Biogen Canada Inc. |
02422956 |
Under Review |
SN |
| 6 |
ARNUITY ELLIPTA – 100 mcg/dose |
GlaxoSmithKline Inc. |
02446561 |
Within Guidelines |
SN |
| 7 |
ARNUITY ELLIPTA – 200 mcg/dose |
GlaxoSmithKline Inc. |
02446588 |
Within Guidelines |
SN |
| 8 |
BELKYRA – 10 mg/milliliter |
Allergan Inc. |
02443910 |
Does Not Trigger Investigation |
B |
| 9 |
BETEFLAM – 2.25 mg/patch |
Cipher Pharmaceuticals Inc. |
02449773 |
Subject to Investigation |
SN |
| 10 |
BLINCYTO – 38.5 mcg/vial |
Amgen Canada Inc. |
02450283 |
Within Guidelines |
SN |
| 11 |
BRENZYS – 50 mg/milliliter |
Merck Canada Inc. |
02455323 |
Within Guidelines |
SN |
| 12 |
BRENZYS – 50 mg/milliliter |
Merck Canada Inc. |
02455331 |
Within Guidelines |
SN |
| 13 |
BRIDION – 100 mg/milliliter |
Merck Canada Inc. |
02451816 |
Subject to Investigation |
MI-P |
| 14 |
BRILINTA – 60 mg/tablet |
AstraZeneca Canada Inc. |
02455005 |
Within Guidelines |
SN |
| 15 |
BRIVLERA – 10 mg/tablet |
UCB Canada Inc. |
02452936 |
Within Guidelines |
SN |
| 16 |
BRIVLERA – 25 mg/tablet |
UCB Canada Inc. |
02452944 |
Within Guidelines |
SN |
| 17 |
BRIVLERA – 50 mg/tablet |
UCB Canada Inc. |
02452952 |
Within Guidelines |
SN |
| 18 |
BRIVLERA – 75 mg/tablet |
UCB Canada Inc. |
02452960 |
Within Guidelines |
SN |
| 19 |
BRIVLERA – 100 mg/tablet |
UCB Canada Inc. |
02452979 |
Within Guidelines |
SN |
| 20 |
BUTRANS – 15 mg/patch |
Purdue Pharma |
02450771 |
Under Review |
SN |
| 21 |
BYDUREON – 2 mg/dose |
AstraZeneca Canada Inc. |
02448610 |
Within Guidelines |
SN |
| 22 |
COPAXONE – 40 mg/milliliter |
Teva Canada Innovation G.P.-S.E.N.C. |
02456915 |
Under Review |
MI-S |
| 23 |
CORTIMENT – 9 mg/tablet |
Ferring Inc. |
02455889 |
Under Review |
SN |
| 24 |
COTELLIC – 20 mg/tablet |
Hoffmann-La Roche Limited |
02452340 |
Subject to Investigation |
SN |
| 25 |
DESCOVY 200/10 – 210 mg/tablet |
Gilead Sciences Canada Inc. |
02454416 |
Under Review |
SN |
| 26 |
DESCOVY 200/25 – 225 mg/tablet |
Gilead Sciences Canada Inc. |
02454424 |
Under Review |
SN |
| 27 |
ELOCTATE – 250 IU/vial |
Biogen Canada Inc. |
02430290 |
Under Review |
SN |
| 28 |
ELOCTATE – 500 IU/vial |
Biogen Canada Inc. |
02430304 |
Under Review |
SN |
| 29 |
ELOCTATE – 750 IU/vial |
Biogen Canada Inc. |
02430312 |
Under Review |
SN |
| 30 |
ELOCTATE – 1000 IU/vial |
Biogen Canada Inc. |
02430320 |
Under Review |
SN |
| 31 |
ELOCTATE – 1500 IU/vial |
Biogen Canada Inc. |
02430339 |
Under Review |
SN |
| 32 |
ELOCTATE – 2000 IU/vial |
Biogen Canada Inc. |
02430347 |
Under Review |
SN |
| 33 |
ELOCTATE – 3000 IU/vial |
Biogen Canada Inc. |
02430355 |
Under Review |
SN |
| 34 |
ENSTILAR – 0.55 mg/g |
Leo Pharma Inc. |
02457393 |
Under Review |
SN |
| 35 |
EPCLUSA 400/100 – 500 mg/tablet |
Gilead Sciences Canada Inc. |
02456370 |
Within Guidelines |
SN |
| 36 |
EVOTAZ – 450 mg/tablet |
Bristol-Myers Squibb Canada Co. |
02446731 |
Within Guidelines |
SN |
| 37 |
FETZIMA – 80 mg/capsule |
Allergan Inc. |
02440997 |
Within Guidelines |
SN |
| 38 |
FETZIMA – 120 mg/capsule |
Allergan Inc. |
02441004 |
Within Guidelines |
SN |
| 39 |
GENVOYA 200/150/150/10 – 510 mg/tablet |
Gilead Sciences Canada Inc. |
02449498 |
Subject to Investigation |
SN |
| 40 |
HUMIRA – 40 mg/syringe |
AbbVie |
02458349 |
Under Review |
SN |
| 41 |
HUMIRA – 40 mg/pen |
AbbVie |
02458357 |
Under Review |
SN |
| 42 |
IBRANCE – 75 mg/capsule |
Pfizer Canada Inc. |
02453150 |
Within Guidelines |
SN |
| 43 |
IBRANCE – 100 mg/capsule |
Pfizer Canada Inc. |
02453169 |
Within Guidelines |
SN |
| 44 |
IBRANCE – 125 mg/capsule |
Pfizer Canada Inc. |
02453177 |
Within Guidelines |
SN |
| 45 |
ICLUSIG – 45 mg/tablet |
Paladin Labs Inc. |
02437341 |
Within Guidelines |
SN |
| 46 |
INVEGA TRINZA – 175 mg/syringe |
Janssen Inc. |
02455943 |
Within Guidelines |
SN |
| 47 |
INVEGA TRINZA – 263 mg/syringe |
Janssen Inc. |
02455986 |
Within Guidelines |
SN |
| 48 |
INVEGA TRINZA – 350 mg/syringe |
Janssen Inc. |
02455994 |
Within Guidelines |
SN |
| 49 |
INVEGA TRINZA – 525 mg/syringe |
Janssen Inc. |
02456001 |
Within Guidelines |
SN |
| 50 |
INVOKAMET 150/1000 – 1150 mg/tablet |
Janssen Inc. |
02455455 |
Under Review |
SN |
| 51 |
INVOKAMET 150/500 – 650 mg/tablet |
Janssen Inc. |
02455439 |
Under Review |
SN |
| 52 |
INVOKAMET 150/850 – 1000 mg/tablet |
Janssen Inc. |
02455447 |
Under Review |
SN |
| 53 |
INVOKAMET 50/1000 – 1050 mg/tablet |
Janssen Inc. |
02455420 |
Under Review |
SN |
| 54 |
INVOKAMET 50/500 – 550 mg/tablet |
Janssen Inc. |
02455404 |
Under Review |
SN |
| 55 |
INVOKAMET 50/850 – 900 mg/tablet |
Janssen Inc. |
02455412 |
Under Review |
SN |
| 56 |
JADENU – 90 mg/tablet |
Novartis Pharmaceuticals Canada Inc. |
02452219 |
Does Not Trigger Investigation |
SN |
| 57 |
JADENU – 180 mg/tablet |
Novartis Pharmaceuticals Canada Inc. |
02452227 |
Does Not Trigger Investigation |
SN |
| 58 |
JADENU – 360 mg/tablet |
Novartis Pharmaceuticals Canada Inc. |
02452235 |
Does Not Trigger Investigation |
SN |
| 59 |
KOVALTRY – 1 N.A./unit |
Bayer Inc. |
02451441 |
Under Review |
SN |
| 60 |
KOVALTRY – 1 N.A./unit |
Bayer Inc. |
02451468 |
Under Review |
SN |
| 61 |
KOVALTRY – 1 N.A./unit |
Bayer Inc. |
02451476 |
Under Review |
SN |
| 62 |
KOVALTRY – 1 N.A./unit |
Bayer Inc. |
02451484 |
Under Review |
SN |
| 63 |
KOVALTRY – 1 N.A./unit |
Bayer Inc. |
02451492 |
Under Review |
SN |
| 64 |
KYPROLIS – 60 mg/vial |
Amgen Canada Inc. |
02451034 |
Does Not Trigger Investigation |
SN |
| 65 |
LENVIMA – 10 mg/day |
Eisai Limited |
02450321 |
Under Review |
SN |
| 66 |
LENVIMA 10/10 – 20 mg/day |
Eisai Limited |
02450305 |
Within Guidelines |
MI-P |
| 67 |
LENVIMA 10/10/4 – 24 mg/day |
Eisai Limited |
02450291 |
Within Guidelines |
MI-P |
| 68 |
LENVIMA 10/4 – 14 mg/day |
Eisai Limited |
02450313 |
Within Guidelines |
MI-P |
| 69 |
LYNPARZA – 50 mg/capsule |
AstraZeneca Canada Inc. |
02454408 |
Subject to Investigation |
MI-P |
| 70 |
MENJUGATE LIQUID – 1 N.A./dose |
GlaxoSmithKline Inc. |
02440709 |
Under Review |
SN |
| 71 |
NATESTO – 5.5 mg/actuation |
Acerus Pharmaceuticals SRL |
02450550 |
Does Not Trigger Investigation |
SN |
| 72 |
NEUPOGEN – 0.6 mg/milliliter |
Amgen Canada Inc. |
02420104 |
Within Guidelines |
SN |
| 73 |
NEUPOGEN – 0.6 mg/milliliter |
Amgen Canada Inc. |
02420112 |
Within Guidelines |
SN |
| 74 |
NINLARO – 2.3 mg/capsule |
Takeda Canada Inc. |
02456796 |
Under Review |
SN |
| 75 |
NINLARO – 3 mg/capsule |
Takeda Canada Inc. |
02456818 |
Under Review |
SN |
| 76 |
NINLARO – 4 mg/capsule |
Takeda Canada Inc. |
02456826 |
Under Review |
SN |
| 77 |
NUWIQ – 250 IU/vial |
Octapharma Canada Inc. |
02432951 |
Does Not Trigger Investigation |
SN |
| 78 |
NUWIQ – 500 IU/vial |
Octapharma Canada Inc. |
02432978 |
Does Not Trigger Investigation |
SN |
| 79 |
NUWIQ – 1000 IU/vial |
Octapharma Canada Inc. |
02432986 |
Under Review |
SN |
| 80 |
NUWIQ – 2000 IU/vial |
Octapharma Canada Inc. |
02432994 |
Under Review |
SN |
| 81 |
ORKAMBI 200/125 – 325 mg/tablet |
Vertex Pharmaceuticals Canada Inc. |
02451379 |
Within Guidelines |
MI-P |
| 82 |
PRECEDEX – 4 mcg/milliliter |
Hospira Healthcare Corporation (Canada) |
02437147 |
Within Guidelines |
SN |
| 83 |
PREGVIT – 1 N.A./tablet |
Duchesnay Inc. |
02451573 |
Under Review |
SN |
| 84 |
PREGVIT FOLIC 5 – 1 N.A./tablet |
Duchesnay Inc. |
02451581 |
Under Review |
SN |
| 85 |
RITUXAN SC – 120 mg/milliliter |
Hoffmann-La Roche Limited |
02457350 |
Within Guidelines |
SN |
| 86 |
SIGNIFOR LAR – 20 mg/vial |
Novartis Pharmaceuticals Canada Inc. |
02437252 |
Within Guidelines |
SN |
| 87 |
SOMAVERT – 25 mg/vial |
Pfizer Canada Inc. |
02448831 |
Within Guidelines |
SN |
| 88 |
SOMAVERT – 30 mg/vial |
Pfizer Canada Inc. |
02448858 |
Within Guidelines |
SN |
| 89 |
STRENSIQ – 18 mg/milliliter |
Alexion Pharmaceuticals Inc. |
02444615 |
Under Review |
B |
| 90 |
STRENSIQ – 80 mg/milliliter |
Alexion Pharmaceuticals Inc. |
02444658 |
Under Review |
B |
| 91 |
SUNVEPRA – 100 mg/capsule |
Bristol-Myers Squibb Canada Co. |
02452294 |
Under Review |
SN |
| 92 |
SYNAGIS – 100 mg/vial |
AbbVie |
02438364 |
Under Review |
SN |
| 93 |
SYNAGIS – 50 mg/vial |
AbbVie |
02438372 |
Under Review |
SN |
| 94 |
SYNJARDY 12.5/1000 – 1012.5 mg/tablet |
Boehringer Ingelheim (Canada) Ltd. |
02456621 |
Under Review |
SN |
| 95 |
SYNJARDY 12.5/500 – 512.5 mg/tablet |
Boehringer Ingelheim (Canada) Ltd. |
02456605 |
Under Review |
SN |
| 96 |
SYNJARDY 12.5/850 – 862.5 mg/tablet |
Boehringer Ingelheim (Canada) Ltd. |
02456613 |
Under Review |
SN |
| 97 |
SYNJARDY 5/1000 – 1005 mg/tablet |
Boehringer Ingelheim (Canada) Ltd. |
02456591 |
Under Review |
SN |
| 98 |
SYNJARDY 5/500 – 505 mg/tablet |
Boehringer Ingelheim (Canada) Ltd. |
02456575 |
Under Review |
SN |
| 99 |
SYNJARDY 5/850 – 855 mg/tablet |
Boehringer Ingelheim (Canada) Ltd. |
02456583 |
Under Review |
SN |
| 100 |
TACTUPUMP FORTE – 70 g/pump |
Galderma Canada Inc. |
02446235 |
Under Review |
SN |
| 101 |
TAGRISSO – 40 mg/tablet |
AstraZeneca Canada Inc. |
02456214 |
Within Guidelines |
MI-P |
| 102 |
TAGRISSO – 80 mg/tablet |
AstraZeneca Canada Inc. |
02456222 |
Within Guidelines |
MI-P |
| 103 |
TALTZ – 80 mg/milliliter |
Eli Lilly Canada Inc. |
02455102 |
Under Review |
SN |
| 104 |
TALTZ – 80 mg/milliliter |
Eli Lilly Canada Inc. |
02455110 |
Under Review |
SN |
| 105 |
TIVICAY – 50 mg/tablet |
ViiV Healthcare ULC |
02414945 |
Within Guidelines |
SN |
| 106 |
TRANSLARNA – 1000 mg/pouch |
PTC Therapeutics International Limited |
|
Within Guidelines |
SN |
| 107 |
TRULICITY – 0.75 mg/pen |
Eli Lilly Canada Inc. |
02448599 |
Within Guidelines |
SN |
| 108 |
UPTRAVI – 200 mcg/tablet |
Actelion Pharmaceuticals Canada Inc. |
02451158 |
Subject to Investigation |
SN |
| 109 |
UPTRAVI – 400 mcg/tablet |
Actelion Pharmaceuticals Canada Inc. |
02451166 |
Does Not Trigger Investigation |
SN |
| 110 |
UPTRAVI – 600 mcg/tablet |
Actelion Pharmaceuticals Canada Inc. |
02451174 |
Within Guidelines |
SN |
| 111 |
UPTRAVI – 800 mcg/tablet |
Actelion Pharmaceuticals Canada Inc. |
02451182 |
Subject to Investigation |
SN |
| 112 |
UPTRAVI – 1000 mcg/tablet |
Actelion Pharmaceuticals Canada Inc. |
02451190 |
Does Not Trigger Investigation |
SN |
| 113 |
UPTRAVI – 1200 mcg/tablet |
Actelion Pharmaceuticals Canada Inc. |
02451204 |
Within Guidelines |
SN |
| 114 |
UPTRAVI – 1400 mcg/tablet |
Actelion Pharmaceuticals Canada Inc. |
02451212 |
Does Not Trigger Investigation |
SN |
| 115 |
UPTRAVI – 1600 mcg/tablet |
Actelion Pharmaceuticals Canada Inc. |
02451220 |
Subject to Investigation |
SN |
| 116 |
VENCLEXTA – 10 mg/tablet |
AbbVie |
02458039 |
Under Review |
SN |
| 117 |
VENCLEXTA – 50 mg/tablet |
AbbVie |
02458047 |
Under Review |
SN |
| 118 |
VENCLEXTA – 100 mg/tablet |
AbbVie |
02458055 |
Under Review |
SN |
| 119 |
VENCLEXTA 10/50/100 – 1 N.A./kit |
AbbVie |
02458063 |
Under Review |
SN |
| 120 |
VIACORAM 14/10 – 24 mg/tablet |
Servier Canada Inc. |
02451557 |
Within Guidelines |
SN |
| 121 |
VIACORAM 3.5/2.5 – 6 mg/tablet |
Servier Canada Inc. |
02451530 |
Within Guidelines |
SN |
| 122 |
VIACORAM 7/5 – 12 mg/tablet |
Servier Canada Inc. |
02451549 |
Within Guidelines |
SN |
| 123 |
VIBATIV – 750 mg/vial |
Pendopharm, Division of Pharmascience Inc. |
02330725 |
Subject to Investigation |
SN |
| 124 |
XIGDUO 5/1000 – 1005 mg/tablet |
AstraZeneca Canada Inc. |
02449943 |
Subject to Investigation |
SN |
| 125 |
XIGDUO 5/850 – 855 mg/tablet |
AstraZeneca Canada Inc. |
02449935 |
Subject to Investigation |
SN |
| 126 |
ZEPATIER 50/100 – 150 mg/tablet |
Merck Canada Inc. |
02451131 |
Subject to Investigation |
SN |
| 127 |
ZERBAXA 1000/500 – 1500 mg/vial |
Merck Canada Inc. |
02446901 |
Under Review |
SN |
| 128 |
ZYTIGA – 500 mg/tablet |
Janssen Inc. |
02457113 |
Does Not Trigger Investigation |
SN |
*Sold after implementation of new Guidelines in 2010:
- SN Slight or No Improvement
- MI-S Moderate Improvement – Secondary
- MI-P Moderate Improvement – Primary
- SI Substantial Improvement
- B Breakthrough
Sold prior to implementation of new Guidelines in 2010:
Category 1 An existing or comparable dosage form of an existing medicine
Category 2 A non-comparable dosage form of an existing medicine, or the first DIN of a new chemical entity that is a breakthrough or provides a substantial improvement over comparable existing DINs
Category 3 A non-comparable dosage form of an existing medicine, or the first DIN of a new chemical entity that provides moderate, little or no therapeutic advantage over comparable existing DINs
Appendix 3: Research and Development
Table 23. Range of R&D-to-Sales Ratios by Number of Reporting Companies and Total Sales Revenue
| Range: R&D-to-sales ratio |
Number of reporting companies: 2016 |
Sales revenues: 2016 ($millions) |
Share: 2016(%) |
Number of reporting companies: 2015 |
Sales revenues: 2015 ($millions) |
Share: 2015(%) |
| 0% |
30 |
2,204.5 |
10.6 |
32 |
1,999.9 |
10.2 |
| ≤10% |
40 |
16,791.7 |
80.5 |
38 |
15,767.8 |
80.1 |
| >10% |
8 |
1,859.5 |
8.9 |
7 |
1,925.6 |
9.8 |
| Total |
78 |
20,855.7 |
100.0† |
77 |
19,693.3 |
100.0† |
† Values in this column may not add to 100.0 due to rounding
Source: PMPRB
Figure 26. Current R&D Expenditures ($millions) by Type of Research, 1988–2016
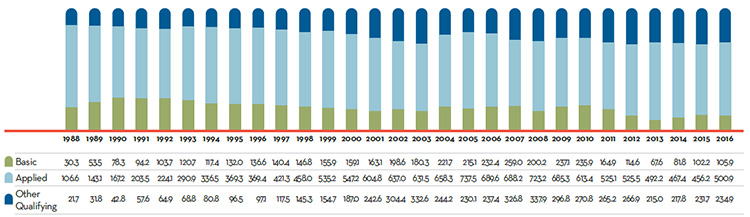
Figure description
This is a vertical stacked bar graph depicting the current R&D expenditures, in millions of dollars, by type of research, based on data collected between 1988 and 2016.
| Year |
Basic |
Applied |
Other Qualifying |
| 1988 |
30.3 |
106.6 |
21.7 |
| 1989 |
53.5 |
143.1 |
31.8 |
| 1990 |
78.3 |
167.2 |
42.8 |
| 1991 |
94.2 |
203.5 |
57.6 |
| 1992 |
103.7 |
224.1 |
64.9 |
| 1993 |
120.7 |
290.9 |
68.8 |
| 1994 |
117.4 |
336.5 |
80.8 |
| 1995 |
132.0 |
369.3 |
96.5 |
| 1996 |
136.6 |
369.4 |
97.1 |
| 1997 |
140.4 |
421.3 |
117.5 |
| 1998 |
146.8 |
458.0 |
145.3 |
| 1999 |
155.9 |
535.2 |
154.7 |
| 2000 |
159.1 |
547.2 |
187.0 |
| 2001 |
163.1 |
604.8 |
242.6 |
| 2002 |
198.6 |
637.0 |
304.4 |
| 2003 |
180.3 |
631.5 |
332.6 |
| 2004 |
221.7 |
658.3 |
244.2 |
| 2005 |
215.1 |
737.5 |
230.1 |
| 2006 |
232.4 |
689.6 |
237.4 |
| 2007 |
259.0 |
688.2 |
326.8 |
| 2008 |
200.2 |
723.2 |
337.9 |
| 2009 |
237.1 |
685.3 |
296.8 |
| 2010 |
235.9 |
613.4 |
270.8 |
| 2011 |
164.9 |
525.1 |
265.2 |
| 2012 |
114.6 |
525.5 |
266.9 |
| 2013 |
67.6 |
492.2 |
215.0 |
| 2014 |
81.8 |
467.4 |
217.8 |
| 2015 |
102.2 |
456.2 |
231.7 |
| 2016 |
105.9 |
500.9 |
234.9 |
Table 24. Ratios of R&D Expenditures to Sales Revenue by Reporting Patentee1, 2016 and 2015
| Company |
R&D-to-Sales Ratio (%) 2016 |
R&D-to-Sales Ratio (%) 2015 |
MIP-to-Cdn Price Ratio (%) – 5 country limit
(List price in Canada compared to International list price) |
Canadian share of sales to PMPRB7 (2016) |
Canadian share of sales to OECD (2016) |
| AbbVie Corporation 2, 3, 4 |
1.7 |
3.0 |
100 |
2.7 |
2.3 |
| Acerus Pharmaceuticals 5 |
2.2 |
|
|
|
|
| Actelion Pharmaceuticals Canada Inc. 2, 4 |
3.6 |
2.6 |
117 |
3.8 |
2.4 |
| Alcon Canada Inc. |
0.9 |
0.6 |
72 |
|
|
| Alexion Pharmaceuticals Inc. 3 |
0.0 |
0.0 |
93 |
|
|
| Allergan Inc. |
1.2 |
2.4 |
83 |
2.5 |
2.3 |
| Amgen Canada Inc. 2, 3 |
4.7 |
5.3 |
73 |
2.8 |
2.6 |
| Aspen Pharma care Canada Inc. 5 |
0.0 |
|
66 |
1.8 |
0.5 |
| Aspri Pharma Canada Inc. |
0.0 |
0.0 |
|
|
|
| Astellas Pharma Canada Inc. 2, 6 |
2.0 |
3.6 |
345 |
2.5 |
1.6 |
| AstraZeneca Canada Inc. 2, 3 |
6.6 |
4.9 |
86 |
3.4 |
2.6 |
| Baxalta Canada Corp. |
0.0 |
0.0 |
|
|
|
| Baxter Corporation |
0.0 |
0.0 |
100 |
0.6 |
0.4 |
| Bayer Inc. 2 |
5.9 |
5.5 |
97 |
9.1 |
5.1 |
| BGP Pharma ULC. 10 |
0.0 |
0.0 |
54 |
|
|
| Biogen Idec Canada Inc. 3 |
10.2 |
10.8 |
100 |
1.5 |
1.4 |
| BioMarin Canada Inc. 3 |
4.5 |
47.0 |
111 |
|
|
| Biovitrum AB |
0.0 |
0.0 |
93 |
2.7 |
1.9 |
| BioSyent Pharma Inc. |
0.0 |
0.0 |
|
|
|
| Boehringer Ingelheim (Canada) Ltd. 2 |
5.0 |
5.5 |
105 |
2.9 |
2.1 |
| Bracco Diagnostics Canada Inc. |
0.0 |
0.0 |
|
0 |
0.004 |
| Bristol-Myers Squibb Canada 2 |
13.6 |
10.8 |
122 |
2.7 |
2.2 |
| Celgene Inc. 3 |
1.5 |
1.6 |
99 |
0.4 |
0.3 |
| Cipher Pharmaceuticals Inc. 5 |
0.0 |
|
|
|
|
| Correvio (UK) Ltd. (Iroko International LP) |
0.0 |
0.0 |
83 |
|
|
| CSL Behring Canada Inc. |
0.2 |
0.2 |
|
|
|
| Duchesnay Inc. |
2.5 |
14.3 |
|
16.5 |
14.1 |
| Eisai Limited. 3 |
8.9 |
1.2 |
107 |
0.7 |
0.3 |
| Eli Lilly Canada Inc. (includes Provel Animal Health Division) 2, 3 |
6.7 |
3.3 |
85 |
2.2 |
1.8 |
| EMD Serono Canada Inc. 2 |
0.0 |
0.0 |
73 |
|
|
| Ferring Pharmaceuticals Inc. 2 |
0.0 |
0.0 |
90 |
3.4 |
2.5 |
| Galderma Canada Inc. |
0.0 |
0.0 |
53 |
|
|
| Gilead Sciences Canada, Inc. 2 |
16.4 |
16.2 |
103 |
2.3 |
1.8 |
| GlaxoSmithKline Inc. 2 |
5.6 |
5.8 |
76 |
3.4 |
2.6 |
| Grifols Canada Ltd. (Talecris Biotherapeutics Ltd.) 3 |
0.0 |
0.0 |
|
0.01 |
0.007 |
| Hoffmann-La Roche Ltd. Canada 2, 3 |
5.6 |
5.1 |
85 |
2.7 |
2.1 |
| Hospira Healthcare Corp. |
0.0 |
0.0 |
76 |
|
|
| Ipsen Biopharmaceuticals Inc. 3, 5 |
0.1 |
|
95 |
0.2 |
0.1 |
| Janssen Inc. 2, 3 |
3.6 |
3.4 |
97 |
7.4 |
6.0 |
| Jazz Pharmaceuticals 11 |
16.5 |
1.4 |
202 |
0.02 |
0.023 |
| Johnson & Johnson Inc. |
0.0 |
0.0 |
|
|
|
| Johnson & Johnson Medical Products |
0.3 |
0.0 |
|
|
|
| Lantheus MI Canada Inc. |
0.0 |
0.0 |
|
|
|
| LEO Pharma Inc. 2 |
0.1 |
1.6 |
30 |
11.0 |
7.9 |
| Lundbeck Canada Inc. |
0.0 |
2.0 |
82 |
7.1 |
5.6 |
| Lupin Pharma Canada Limited |
0.0 |
0.4 |
103 |
0.07 |
0.06 |
| McNeil Consumer Healthcare Canada |
2.7 |
3.8 |
|
|
|
| Meda Pharmaceuticals Ltd. |
0.0 |
0.0 |
30 |
|
|
| Medexus Inc. 5 |
0.0 |
|
|
|
|
| Merck Canada Inc. 2, 3 |
2.9 |
2.2 |
85 |
3.2 |
2.5 |
| Merus Labs |
0.0 |
0.0 |
|
29.1 |
16.5 |
| Merz Pharma Canada Ltd. |
2.0 |
1.3 |
90 |
1.4 |
1.1 |
| Novartis Pharmaceuticals Canada Inc. 2, 3 |
3.6 |
3.9 |
87 |
5.6 |
4.0 |
| Novo Nordisk Canada Inc. 2, 3 |
1.1 |
2.0 |
88 |
1.8 |
1.6 |
| Octapharma Canada Inc. |
6.6 |
0.1 |
|
|
|
| Otsuka Canada Pharmaceutical Inc. (OCPI) 2 |
1.3 |
4.5 |
110 |
1.1 |
0.5 |
| Paladin Labs Inc. 2 |
0.2 |
0.2 |
57 |
|
|
| Pfizer Canada Inc. 2, 3 |
1.0 |
0.9 |
95 |
2.8 |
2.1 |
| Pharmascience Inc. 5 |
8.4 |
|
|
|
|
| Purdue Pharma 2 |
4.8 |
3.9 |
159 |
|
|
| PTC Therapeutics International Ltd. |
130.5 |
0.0 |
|
|
|
| Ranbaxy Pharmaceuticals Canada Inc. |
0.0 |
0.0 |
|
|
|
| Sanofi Canada Inc. 2, 3, 8 |
1.6 |
2.1 |
82 |
1.9 |
1.5 |
| Sanofi Pasteur Ltd. 2, 3, 7 |
68.0 |
80.1 |
|
|
|
| Seattle Genetics Inc. |
9.8 |
7.2 |
95 |
|
|
| Servier Canada Inc. 2 |
2.1 |
2.6 |
|
14.6 |
8.7 |
| Shire Canada Inc. 2, 3 |
0.0 |
0.0 |
100 |
1.9 |
1.7 |
| Shire Human Genetic Therapies 2, 3 |
0.0 |
0.0 |
105 |
|
|
| Sunovion Pharmaceuticals Canada Inc. 2 |
0.0 |
0.0 |
100 |
|
|
| Takeda Canada Inc. 2, 3 |
0.0 |
0.0 |
83 |
2.8 |
1.5 |
| Theratechnologic Inc. 2 |
0.0 |
0.0 |
|
|
|
| Teva Canada Innovation 3 |
0.2 |
0.1 |
102 |
|
|
| Tribute Pharma Canada Inc. |
0.0 |
0.0 |
|
|
|
| UCB Canada Inc. 3 |
16.9 |
1.0 |
94 |
1.4 |
1.1 |
| Valeant Canada Ltd. 3, 9 |
3.1 |
5.0 |
60 |
2.9 |
2.6 |
| Valneva Austria GmbH. 3, 5 |
0.0 |
|
63 |
80.8 |
41.4 |
| Vertex Pharma Canada Inc. 3 |
47.5 |
72.5 |
112 |
|
|
| VIIV Healthcare ULC. 2 |
0.0 |
0.0 |
111 |
|
|
1 To avoid double counting of sales revenues, revenues from royalties are included in calculating each company’s ratio but not included in calculating industry-wide ratios. Federal and provincial government grants are subtracted from the R&D expenditure in calculating individual R&D-to-sales ratios but are included in calculating industry-wide ratios. Differences between the list of firms filing data on prices and those filing R&D data are due to differences in reporting practices of patentees and their affiliates or licensees. Note as well that some veterinary patentees (i.e., those without revenue from sales of products for human use) are required to file information on R&D expenditure but not price and sales information.
2 Member of Innovative Medicines Canada.
3 Member of BIOTECanada.
4 Spin-off of Abbott’s proprietary products division into a separate legal entity effective Oct. 31, 2012.
5 Not a patentee in 2015.
6 Formerly known as Fujisawa Canada Inc.
7 Formerly known as Aventis Pasteur Ltd.
8 Formerly known as Aventis Pharma Inc.
9 Formerly known as ICN Canada Ltd.
10 “BGP Pharma ULC” to house the former “Abbott” and “Fournier” pharmaceutical brands in Canada.
11 Did not file in 2015.
Table 25. Current R&D Expenditures by Province/Territory, 2016
| Province |
Expenditures: All patentees ($thousands) |
Regional share (%) |
Expenditures: Innovative Medicines Canada ($thousands) |
Regional share (%) |
| Newfoundland |
3,934.16 |
0.468 |
2,765.42 |
0.396 |
| Prince Edward Island |
1.20 |
0.000 |
1.20 |
0.000 |
| Nova Scotia |
8,435.37 |
1.002 |
7,313.19 |
1.047 |
| New Brunswick |
3,601.73 |
0.428 |
3,006.98 |
0.431 |
| Quebec |
272,567.83 |
32.384 |
189,378.29 |
27.125 |
| Ontario |
413,077.27 |
49.079 |
367,247.73 |
52.601 |
| Manitoba |
5,505.25 |
0.654 |
4,322.24 |
0.619 |
| Saskatchewan |
2,123.68 |
0.252 |
1,242.42 |
0.178 |
| Alberta |
92,530.46 |
10.994 |
88,201.18 |
12.633 |
| British Columbia |
39,885.44 |
4.739 |
34,692.47 |
4.969 |
| Territories |
0 |
0.000 |
0 |
0.000 |
| Canada |
841,662.41† |
100.0† |
698,171.13† |
100.0† |
† Values in this row may not add due to rounding.
Source: PMPRB
Table 26. Current R&D Expenditures by Performer and Province/Territory, 2016
| Province |
blank |
Patentees |
Other Companies |
University |
Hospitals |
Others |
| Newfoundland |
$000 |
1,258.32 |
1,373.54 |
472.67 |
410.15 |
419.49 |
| blank |
% |
32.0 |
34.9 |
12.0 |
10.4 |
10.6 |
| Prince Edward Island |
$000 |
0.00 |
0.00 |
0.00 |
0.00 |
1.20 |
| blank |
% |
0.0 |
0.0 |
0.0 |
0.0 |
100.0 |
| Nova Scotia |
$000 |
1,099.45 |
3,258.46 |
990.03 |
1,395.31 |
1,692.13 |
| blank |
% |
13.0 |
38.6 |
11.7 |
16.5 |
20.1 |
| New Brunswick |
$000 |
413.51 |
843.06 |
1,461.15 |
653.51 |
230.51 |
| blank |
% |
11.5 |
23.4 |
40.6 |
18.1 |
6.4 |
| Quebec |
$000 |
107,499.96 |
91,929.10 |
12,862.37 |
16,089.83 |
44,186.56 |
| blank |
% |
39.4 |
33.7 |
4.7 |
5.9 |
16.2 |
| Ontario |
$000 |
193,517.07 |
95,449.65 |
33,239.88 |
45,197.01 |
45,673.66 |
| blank |
% |
46.8 |
23.1 |
8.0 |
10.9 |
11.1 |
| Manitoba |
$000 |
1,238.22 |
1,745.22 |
734.11 |
958.05 |
829.65 |
| blank |
% |
22.5 |
31.7 |
13.3 |
17.4 |
15.1 |
| Saskatchewan |
$000 |
60.56 |
1,073.40 |
841.71 |
39.09 |
108.93 |
| blank |
% |
2.9 |
50.5 |
39.6 |
1.8 |
5.1 |
| Alberta |
$000 |
71,869.40 |
8,362.61 |
4,916.43 |
4,175.13 |
3,206.89 |
| blank |
% |
77.7 |
9.0 |
5.3 |
4.5 |
3.5 |
| British Columbia |
$000 |
17,958.17 |
9,535.94 |
2,695.45 |
4,242.57 |
5,453.31 |
| blank |
% |
45.0 |
23.9 |
6.8 |
10.6 |
13.7 |
| Territories |
$000 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
| blank |
% |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
| Canada |
$000 |
394,914.67 |
213,570.98 |
58,213.78 |
73,160.64 |
101,802.33 |
| blank |
% |
46.9 |
25.4 |
6.9 |
8.7 |
12.1 |
Notes:
- The percentage under each R&D category gives the percentage of all money spent in that category in that province.
- Expenditures as a percentage of total means percentage of R&D expenditures in that province compared to total R&D in Canada.
- Rows and columns may not equal totals due to rounding.
- Current expenditures plus capital expenditures (equipment + depreciation) = total R&D expenditures.
Source: PMPRB